Your Family Physician
Sunday, May 3, 2009
Infertility
Introduction
Prevalence
The most commonly accepted definition of the term infertility is the lack of pregnancy (regardless of cause) after 1 year of unprotected intercourse. Infertility affects approximately 15% of couples of reproductive age. Its prevalence has been stable during the past 50 years, although a shift in etiology and in the age of the patient population has occurred.
Fertility is influenced by the moral attitudes of US society, in which sexual liberation and promiscuity have increased during the last 40 years, and by changing priorities among women. In developed countries, where family planning and professional career development are practiced, some women postpone childbearing until after age 30-40 years. Improvements in fertility treatment have made it possible for many patients with severe male-factor infertility to have a family. These new and advanced technologies include in vitro fertilization (IVF), intracytoplasmic sperm injection (ICSI), and other related procedures in Assisted Reproductive Technologies (ART).
Fertility is defined as the capacity to reproduce or the state of being fertile. This term should be differentiated from fecundability, which is the probability of achieving a pregnancy each month, and fecundity, which is the ability to achieve a live birth within one menstrual cycle. The fecundability rate in the general population is rather constant and is approximately 0.22/mo (Maruani, 1983). The estimated fecundity rate is 0.15-0.18/mo, representing a cumulative pregnancy rate of 90%/y (Trussell, 1985).
For excellent patient education resources, visit eMedicine's Pregnancy and Reproduction Center and Women's Health Center. Also, see eMedicine's patient education articles Infertility, In Vitro Fertilization, Amenorrhea, Menopause, Ectopic Pregnancy, and Miscarriage.
Etiology of Infertility
The reproduction process requires the interaction and integrity of the female and male reproductive tracts, which allows for (1) the release of a normal preovulatory oocyte, (2) the production of adequate spermatozoa, (3) the normal transport of the gametes to the ampullary portion of the fallopian tubes (where fertilization occurs), and (4) the subsequent transport of the cleaving embryo up to the endometrial cavity for its normal implantation and further development.
The origin of infertility is similarly due to male or female factors; the causes are multiple. Female factors account for 32% of infertility. Male factors account for 18.8% of infertility. Male and female factors combined cause 18.5% of fertility. The etiology is unknown in 11.1%, and other causes are identified in 5.6%.
Those with an unknown etiology can also be categorized as a normal infertile couple (NIC), indicating that all findings from standard tests used to evaluate the patients are normal. In NICs, the actual cause for infertility cannot be detected because it may be at the oocyte/sperm level or it may be due to the quality of the embryo or to any disruption at the implantation site level. In the future, ascribing the mutation or the absence of a specific gene as the cause of infertility may be possible in this patient population.
Other etiologic factors have been associated with an increased risk of infertility. These include pelvic inflammatory disease (PID); endometriosis; environmental and occupational factors; toxic effects related to tobacco, marijuana, or other drugs; exercise; inadequate diet associated with extreme weight loss or gain; and advanced age.
Pelvic inflammatory disease
PID has been associated with gonorrhea infection for more than a century (Brand, 1985). While gonorrhea still plays an important role in tubal disease, it has been surpassed by chlamydia. Westrom reported a 21% incidence of infertility in a group of Swedish women who were diagnosed with PID, which was confirmed by laparoscopy findings (Westrom, 1975). The rate of damage to the fallopian tubes increases with subsequent PID episodes, from 34% for the first episode to 54% in women with second and third episodes (Westrom, 1980).
PID can be diagnosed clinically and confirmed by results from cervical culture and serologic antibody assays for gonorrhea and chlamydia (Moller, 1985; Tjiam, 1985; Robertson, 1987). In many instances, a patient never recalls having had an acute PID episode; however, years later, the incidental finding of tubal obstruction after hysterosalpingogram (HSG) or laparoscopy may be the only indication of previous PID.
Endometriosis
Endometriosis remains an enigmatic pathologic disease that affects women during their reproductive years. The incidence increases with patient age and low parity, and, although controversial, endometriosis appears to affect more patients in middle and high socioeconomic classes. The evolution of the disease is unpredictable. Pelvic pain and reproductive failure are the 2 major complaints of patients with endometriosis (Strathy, 1982; Balasch, 1996). Although a gene defect has not yet been identified for endometriosis, a genetic link seems probable based on the observation of chromosomal defects in endometriotic tissue and the observation of a 7-fold increased risk of endometriosis in patients with a family history of the disease (Simpson, 1980; Shin, 1997; Kennedy, 1998; Kosugi, 1999).
Endometriosis lesions vary from microscopic to macroscopic. Classic endometriosis appears as black pigments, ie, powder-burn lesions that affect the surface of the peritoneum of the bladder, ovary, fallopian tubes, cul-de-sac, and bowel. Nonclassic endometriosis appears as nonpigments, ie, red, tan, white lesions or vesicles. The final diagnosis should be confirmed by demonstrating endometrial stroma and glands in biopsy tissue (Jansen, 1986; Martin, 1989).
The incidence of endometriosis in primary and secondary infertility varies according to the population studied. Mahmood reported an incidence rate of 26% and 13%, respectively (Mahmood, 1991). Severe endometriosis with damage to the fallopian tubes and ovaries due to adhesions or the presence of endometriomas is an obvious cause of infertility. The hypothesis that minimal and mild endometriosis cause infertility is controversial. Several studies failed to prove an increased pregnancy rate after treatment or expectant therapy (Schenken, 1982; Seibel, 1982; Fedele and Parazzini, 1992; Fedele and Bianchi, Fertil Steril, 1992).
Minimal and mild endometriosis is hypothesized to reduce fertility by the following mechanisms (Dmowski, 1981; Mathur, 1982; Muscato, 1982; Halme, 1984; Chacho, 1986; Gleicher, 1987; Mori, 1992; Hemmings, 1993; Akoum, 1995; Ryan, 1995; Garzetti, 1996; Harada, 1997; Braun, 1998; Koninckx, 1998):
- Increased peritoneal macrophages that increase phagocytosis of the sperm (Muscato, 1982; Halme, 1984; Chacho, 1986)
- Decreased sperm binding to the zona pellucida
- Proliferation of peritoneal lymphocytes
- Increased cytokinin levels
- Increased immunoglobulin production
- Embryo toxic serum
- Defective natural killer activity
Endometriosis has been associated with ovulatory disorders such as luteal phase deficiency (LPD), oligo-ovulation, and luteinized unruptured follicle (LUF) syndrome (Mahmood, 1991). Although pelvic pain appears to be a common symptom of endometriosis, in some patients, endometriosis is an incidental finding discovered during diagnostic laparoscopy for evaluation of infertility (Cornillie, 1990; Koninckx, 1991; Fedele, Obstet Gynecol, 1992; Vercellini, 1996).
Environmental and occupational factors
Concerns are currently increasing regarding the impact of environmental factors and subsequent repercussions on infertility. Published semen analysis reports from 1985 confirm a 20% decrease of sperm concentration compared with reports published in the 1960s (US Congress, 1985).
Many other factors, such as excessive heat exposure, microwave radiation, ultrasonography, and other health hazards, are more controversial as real infertility-inducing factors (Thomas, 1990). Excessive radiation damages the germinal cells. Exposure to lead, other heavy metals, and pesticides has also been associated with male infertility (Buiatti, 1984; Bryant, 1989).
Toxic effects related to tobacco, marijuana, and other drugs
Smoking has been associated with infertility in both males and females (Baird, 1985; Howe, 1985; Mueller, 1989). In experimental animals, nicotine and polycyclic aromatic hydrocarbons block spermatogenesis and decrease testicular size. In women, tobacco alters the cervical mucus and the cilial epithelium and affects gamete transport (Stillman, 1986; Phipps, 1987).
Marijuana and its metabolite, delta-9-tetrahydrocannabinol, inhibit the secretion of luteinizing hormone (LH) and follicle-stimulating hormone (FSH), thus inducing ovulatory disorders and LPD in women (Smith, 1987). Marijuana use affects males by decreasing the sperm count and the quality of the sperm.
Heroin, cocaine, and crack cocaine use induces similar effects but places the user at increased risk for PID and HIV infection associated with sexual promiscuity.
In women, the effects of alcohol are related more to severe consequences for the fetus. Nevertheless, chronic alcoholism is related to ovulatory disorders and, therefore, interferes with fertility. Alcohol use by males interferes with the synthesis of testosterone and has an impact on sperm concentration. Alcoholism may delay the sexual response and may be conducive to impotence.
Exercise
Exercise should be encouraged as part of normal activities. However, compulsive exercise is deleterious, especially for long-distance runners. Jogging stimulates the secretion of endorphins, and excessive secretion of endorphins interferes with the normal production of FSH and LH, in turn inducing ovulatory disorders and LPD, which accounts for lack of embryo implantation and first-trimester miscarriages (Baker, 1981; Green, 1986). In males, exercise has been associated with oligospermia.
Inadequate diet associated with extreme weight loss or gain
Obesity is becoming a major health issue in the United States. Obesity has an impact on infertility only when the female patient's weight reaches extremes. Although weight loss associated with anorexia nervosa or bulimia induces hypothalamic amenorrhea, a low FSH level, and low LH secretion, weight gain is tolerated better. Also, the disruption of the hypothalamic-pituitary-ovarian axis is affected in association with other endocrine disorders such as polycystic ovarian disease (PCOD), adrenal hyperplasia, and hypothyroidism.
Advanced age
The prevalence of infertility rises dramatically as age increases (US Congress, 1988). Furthermore, fertility decreases with marriage duration because of the decrease in the frequency of intercourse or the use of contraception. Studies report that among Mormons, fertility appears to be stable until age 36 years, declines slightly until age 40 years, and is followed by a sharp decline after age 42 years (Trussell, 1985; Menken, 1986).
Similar conclusions can be drawn from the experience of IVF programs, as has been duplicated in multiple IVF studies worldwide. Chromosomal abnormalities and poor oocyte quality are 2 examples of causes of poor embryonic quality, low implantation rate, increased miscarriage, and low birth rates (Romeu, 1987). Analysis from donor oocyte programs in which the oocytes of younger patients (aged 21-30 y) are used has shown that the pregnancy rate in older recipients is comparable to the pregnancy rate of younger patients undergoing ART (Navot, 1994).
Aging also affects male fertility. Testosterone levels decrease, gonadotropin levels increase, sperm concentration and semen volume change, and libido decreases. In addition, the incidence of birth defects increases. Age affects female fertility dramatically, but males are not affected as much; anecdotal reports exist of men fathering children when older than 80 years.
General Guidance On Evaluation Of Infertility
Infertility is a problem that involves both partners equally. The consultation is incomplete if only the woman is evaluated. Anxiety is very common, and many couples seek consultation after a few months of unprotected intercourse. Diagnostic testing is unnecessary if the couple has not attempted to conceive for at least 1 year, unless they have a history of a male factor, endometriosis, a tubal factor, diethylstilbestrol (DES) exposure, PID, or pelvic surgery. A brief explanation of the physiology of reproduction and reassurance are enough to lessen the anxiety of the couple.
History
The patients (ie, the couple) should provide a copy of their previous medical records and should complete a medical history questionnaire to be submitted in advance of the initial consultation (see Image 105 for the Johns Hopkins "Husband and Wife Medical History Packet"). Obtain a detailed medical history regarding the type of infertility (primary or secondary) and its duration.
- Obtain a history of previous pregnancies and their outcomes; interval between pregnancies; and detailed information about pregnancy loss, duration of pregnancy, human chorionic gonadotropin (hCG) level, ultrasound data, and the presence or absence of a fetal heartbeat.
- During the history of previous infertility evaluation and treatment, specific questions should address the issues of frequency of intercourse, use of lubricants (eg, K-Y gel) that could be spermicidal, use of vaginal douches after intercourse, and the presence of any sexual dysfunction such as anorgasmia or dyspareunia.
- Question female patients about their menstrual history, frequency, and patterns since menarche. A history of weight changes, hirsutism, frontal balding, and acne should also be addressed.
- Ask male patients about previous spermiogram results, history of impotence, premature ejaculation, change in libido, history of testicular trauma, previous relationships, history of any previous pregnancy, and the existence of offspring from previous partners.
- Ask the couple about their history of sexually transmitted diseases (STDs); surgical contraception (eg, vasectomy, tubal ligation); lifestyle; consumption of alcohol, tobacco, and recreational drugs (amount and frequency); occupation; and physical activities.
- Ask the couple whether they are currently under medical treatment, the reason, and whether they have a history of allergies.
- A complete review of systems may be helpful to identify any endocrinological or immunological problem that may be associated with infertility.
Physical
A physical examination should be completed. Some health insurance carriers designate or include an obstetrician/gynecologist as a patient's primary care physician.
- Routine records of blood pressure, pulse rate, and temperature (if applicable) are needed.
- Measure height and weight to calculate the body mass index, and measure arm span when indicated.
- Perform an eye examination to establish the presence of exophthalmos, which can be associated with hyperthyroidism.
- The presence of epicanthus, lower implantation of the ears and hairline, and webbed neck can be associated with chromosomic abnormalities.
- Carefully evaluate the thyroid gland to exclude gland enlargement or thyroid nodules.
- Perform a breast examination to evaluate breast development and to seek abnormal masses or secretions, especially galactorrhea. Take the opportunity to educate patients about breast self-examination during the early days of their menstrual cycles.
- The abdominal examination should be directed to the presence of abnormal masses at the hypogastrium level.
- A thorough gynecological examination should include an evaluation of hair distribution, clitoris size, Bartholin glands, labia majora and minora, and any condylomata acuminatum or other lesions that could indicate the existence of venereal disease.
- The inspection of the vaginal mucosa may indicate a deficiency of estrogens or the presence of infection.
- The evaluation of the cervix should include a Papanicolaou test (Pap smear) and cultures for gonorrhea, chlamydia, and Ureaplasma urealyticum.
- Bimanual examination should be performed to establish the direction of the cervix and the size and position of the uterus in order to exclude the presence of uterine fibroids, adnexal masses, tenderness, or pelvic nodules indicative of infection or endometriosis.
- The gynecologist should perform a pelvic ultrasonographic scan. This enables the physician to establish an early diagnosis of adnexal masses; to determine the size and aspect of the ovaries; and to detect the presence of endometrial polyps, submucous fibroids, and hydrosalpinx.
- The examination of the extremities is important to rule out malformation, such as shortness of the fourth finger or cubitus valgus, which can be associated with chromosomal abnormalities and other congenital defects. Examine the skin to establish the presence of acne, hypertrichosis, and hirsutism.
- The urologist usually examines the male partner.
- Attention should be directed to congenital abnormalities of the genital tract (eg, hypospadias, cryptorchid, congenital absence of the vas deferens).
- Testicular size, urethral stenosis, and presence of varicocele are also determined.
- A history of previous inguinal hernia repair can indicate an accidental ligation of the spermatic artery.
Comprehensive Evaluation Of Infertility
Evaluation of infertile couples should be organized and thorough. The findings must be discussed with the couple after completion of the history and physical examination. Diagnostic tests should progress from the simplest (eg, postcoital test [PCT], endometrial biopsy) to the more complex or to the one that implies a major risk to the patient (eg, laparoscopy). The couple will be stressed by their need to seek medical intervention; therefore, to relieve anxiety, emphasize that a complete infertility evaluation is performed according to the woman's menstrual cycle and may take up to 2 menstrual cycles before the factor(s) causing the infertility problem is found.
Evaluation of the male partner
The male partner must submit a semen sample for a comprehensive semen analysis. Previous paternity does not guarantee that his reproductive system has not been affected since the birth of his offspring. The comprehensive semen analysis must be performed in a certified andrology laboratory, and the semen sample, preferably, should be collected at the same andrology laboratory that will conduct the test. However, if the sample must be collected at home, it must be collected in a sterile plastic container and delivered to the andrology laboratory at body temperature not later than 30 minutes after ejaculation.
Some patients cannot produce a semen sample through masturbation. Therefore, the sample can be collected through intercourse, using a special nonspermicidal condom provided by the andrology laboratory. To optimize results, the semen sample should be collected after a period of 3 days but no more than 5 days of sexual abstinence.
Semen analysis
The basic semen analysis assesses the characteristics of sperm concentration, motility, morphology, and viability. The World Health Organization's semen analysis parameters (with the variable and the corresponding reference range) are as follows (World Health Organization, 1992):
- Volume - 2-5 mL
- pH - 7.2-7.8
- Sperm concentration - 20 million or greater
- Motility - 50%, forward progression
- Morphology - Normal sperm (50% or greater)
- White blood cells - Fewer than 1 million cells/µL
Morphology has become an important parameter to evaluate the quality of the sperm and to assess the fertilization capability. Kruger reported a new classification based on strict sperm morphology after fixing and staining the sperm (Kruger, 1986). Using the Kruger criteria, sperm morphology must be greater than 14% to be considered normal. Morphology of less than 4% is associated with severe infertility and is an indication for ART/ICSI.
Specific biochemistry analyses relevant to the accessory sex gland functions can be performed using the semen sample. These include fructose from the seminal vesicles, zinc and acid phosphatase from the prostate gland, and alpha-glucosidases and carnitine from the epididymis (Zeneveld, 1990).
Sperm agglutination indirectly indicates the presence of sperm antibodies. The immunobead test can be performed either directly on the sperm or indirectly on the sperm and blood. Surface antibodies against immunoglobulin A (IgA) or immunoglobulin G (IgG) may be present. The antibodies can be specific for the head or for the tail of the sperm (Clarke, 1985; Clarke, 1988; Barratt, 1992). IgA sperm antibodies interfere with the sperm-oocyte interaction and account for a lack of fertilization, whereas IgG sperm antibodies are more responsible for decreasing sperm motility. Sperm antibodies are associated with infection (ie, orchitis), testicular trauma, and a history of vasectomy.
Interpretation of semen analysis
Spermatogenesis takes approximately 72 days. Abnormal semen analysis results can be attributed to various unknown reasons (eg, short period of sexual abstinence, incomplete collection, poor sexual stimulus); therefore, repeating the semen analysis at least 1 month later is important before a diagnosis is made. Explaining to the patient the normal fluctuation that can occur between semen samples is also important. Avoid a hasty diagnosis that may create unnecessary anxiety for the couple.
Azoospermia indicates absence of sperm that could be related to congenital absence or bilateral obstruction of the vas deferens or ejaculatory ducts, history of spermatogenesis arrest, Sertoli cell syndrome, or postvasectomy.
Oligozoospermia indicates a concentration of fewer than 20 million sperm/mL and could be associated with ejaculatory dysfunction such as retrograde ejaculation.
Asthenozoospermia indicates sperm motility of less than 50%. Extreme temperatures and delayed analysis after sperm collection are among the factors that decrease sperm motility.
Teratospermia indicates an increased number of abnormal sperm morphology at the head, neck, or tail level.
Hypospermia indicates a decrease of semen volume to less than 2 mL per ejaculation.
Hyperspermia indicates an increase of sperm volume to more than 8 mL per ejaculation.
Sperm function tests
After ART, a proliferation of different sperm tests have been developed to evaluate and predict the sperm fecundability, including (1) the acrosome reaction test with fluorescent lectins or antibodies, (2) computer assessment of the sperm head, (3) computer motility assessment, (4) hemizona-binding assay, (5) hamster penetration test, and (6) human sperm-zona penetration assay (Margalioth, 1986; Franken and Burkman, 1989; Franken and Oehninger, 1989; Oehninger, 1989). Numerous publications describe the positive and negative aspects of these tests. The tests are diagnostic tools, and the results are subject to many variations in their interpretation, which render them of more academic interest than of practical therapeutic value.
Evaluation of the female partner
Several congenital or acquired conditions affect female reproductive function. These conditions alter either the anatomy or the normal physiology of reproduction and may impair the transport of the gametes or embryo(s) and/or interfere with implantation and embryo/fetal development.
A complete evaluation of the female reproductive tract must include cervical, uterine, endometrial, tubal, peritoneal, and ovarian factors.
Cervical factors
The uterine cervix plays a pivotal role in the transport and capacitation of the sperm after intercourse. The cervical factor accounts for 5-10% of infertility. Cervical mucus production, amount, and characteristics change according to the estrogen concentration during the late follicular phase.
At the beginning of the menstrual cycle, cervical mucus is scanty, viscous, and very cellular. The mucus forms a netlike structure that does not allow the passage of sperm. Mucus secretion increases during the mid follicular phase and reaches its maximum approximately 24-48 hours before ovulation.
The water and salt concentration increases, changing the physical characteristics of the mucus. It becomes thin, watery, alkaline, acellular, and elastic (spinnbarkheit) because of the increased concentration of sodium chloride, despite a fernlike pattern when the mucus is allowed to dry on a cover slide under the microscope (see Images 1-3).
At this point, the mucus organizes itself, forming multiple microchannels so the spermatozoa can travel through. During this journey, the spermatozoa simultaneously undergo activation and capacitation (Overstreet, 1986). In addition, the mucus acts as a filter for abnormal spermatozoa and cellular debris present in the semen.
Cervical-factor infertility refers to abnormalities of the mucus-sperm interaction. The PCT is also known as the Sims-Huhner test (Giner, 1974; Hull, 1982; Portuondo, 1982; Overstreet, 1986). The test consists of evaluating the amount of spermatozoa and its motility within the cervical mucus during the preovulatory period. The couple is asked to have intercourse, without the use of lubricants, 8-12 hours before the test. Although several articles have shown a higher sperm concentration 2-3 hours after intercourse, a longer interval of 8-12 hours provides the opportunity to evaluate sperm endurance.
The mucus characteristics that determine a positive PCT test result are a volume of 0.3-1 mL, spinnbarkheit greater than 10 cm, a ferning pattern, occasional cellularity, and a sperm count of 10-20 per high-power field (eg, 45X motility forward progression).
The timing of the PCT is crucial for the interpretation of results. If the PCT is scheduled too early or 24 hours after ovulation, a false-negative finding is the most likely result. Therefore, performing the test around the time of ovulation is recommended. If the patient has a 28-day menstrual cycle, the test should be performed on approximately menstrual cycle day 13-14. If the patient has menstrual irregularities, using a urinary LH kit to predict the preovulatory LH surge is advised in order to have a reliable PCT result. Changes in the basal body temperature (BBT) chart indicate ovulation. Although a BBT chart (see Image 59) taken during the menstrual cycle has been used for decades, the results from analysis are always retrospective and are misleading in approximately 20% of cases (Lenton, 1977).
An abnormal PCT result may indicate (1) poor timing, (2) hostile cervical mucus related to cervicitis (eg, bacteria/yeast), (3) low pH, (4) sperm antibodies, (5) poor technique during intercourse (eg, premature ejaculation), (6) undiagnosed male factor infertility, and/or (7) anatomical defects (eg, due to DES exposure in utero, cervical cone). An abnormal PCT result must be repeated in subsequent cycles before a final diagnosis is made. Note that an abnormal PCT result does not preclude the possibility of a spontaneous pregnancy; therefore, be cautious to avoid a premature final diagnosis.
Uterine factors
The uterus is the final destination for the embryo and the place where the fetus develops until delivery. Therefore, uterine factors may be associated with primary infertility or with pregnancy wastage and premature delivery. Uterine factors can be congenital or acquired. They may affect the endometrium or the myometrium and are responsible for 2-5% of infertility cases. Other problems affect the development and function of the endometrium.
- Congenital defects
- The normal development of the müllerian ducts accounts for the normal anatomic configuration of the uterus, fallopian tubes, cervix, and upper vagina. The full spectrum of congenital/müllerian abnormalities varies from total absence of the uterus and vagina (ie, Rokitansky-Küster-Hauser syndrome) to minor defects such as arcuate uterus and vaginal septa (transverse or longitudinal).
- The most common uterine malformations observed during the past 40 years are drug induced. From late in the 1950s until the early 1970s, DES was used as a treatment modality in patients with a history of recurrent miscarriages. Years later, DES was found to be responsible for inducing malformations of the uterine cervix, irregularities of the endometrial cavity (eg, T-shaped uterus), malfunction of the fallopian tubes, menstrual irregularities, and the development of clear cell carcinoma of the vagina (DeCherney, 1981). In many instances, the role or implications of those anomalies and the etiology of infertility are unclear.
- In 1988, the American Fertility Society (AFS) established a new classification of müllerian anomalies. The purpose of this classification was to gather prospective clinical information, to determine its relevance, and to generate future recommendations for patient care. The relationship between müllerian anomalies and infertility is not clear except when an absolute absence of the uterus, cervix, vagina, or a combination of these absences occurs.
- Premature delivery has been associated with cervical incompetence, unicorn uterus associated with a blind horn, and septate uterus, which could be responsible for implantation problems and first-trimester miscarriages.
- Acquired defects
- Endometritis associated with a traumatic delivery, dilatation and curettage, intrauterine device, or any instrumentation (eg, myomectomy, hysteroscopy) of the endometrial cavity may create intrauterine adhesions or synechiae (ie, Asherman syndrome), with partial or total obliteration of the endometrial cavity.
- Placental polyps may develop from placental remains.
- Intrauterine and submucous fibroids cause distortion of the cavity; compromise the blood supply; and are responsible for a lack of embryo implantation, early miscarriages, premature delivery, and abruptio placentae.
- Diagnosis
- Some defects can be detected during the pelvic examination. These include absence of the vagina and uterus, vaginal septum, and the presence of fibroids.
- Detection of most defects requires ancillary studies such as HSG, pelvic ultrasonography, hysterosonogram, and MRI.
- Operative procedures, such as laparoscopy and hysteroscopy, are often necessary for confirmation of the final diagnosis.
- Hysterosalpingogram
- The HSG is the most frequently used diagnostic tool to evaluate the endometrial cavity. Some have tried to displace the role of HSG in the evaluation of infertility; however, a meticulous and well-executed procedure, performed under fluoroscopy, provides accurate information about the (1) endocervical canal; (2) diameter and configuration of the internal os; (3) endometrial cavity; (4) uterine/tubal junction (cornual ostium); (5) diameter, location, and direction of the fallopian tubes; (6) status of the fimbriae; and (7) spill into the endometrial cavity. Furthermore, the HSG provides indirect evidence of pelvic adhesions and uterine, ovarian, or adnexal masses (Hunt, 1990).
- The HSG should be performed during the early follicular phase. At this phase, the endometrium is thin, the HSG provides better information and delineation of minor defects, and the possibility of accidental irradiation to the fetus if the patient is pregnant is eliminated.
- Carefully disinfect the cervix with a povidone-iodine solution (Betadine) to avoid the transfer of bacteria inside the reproductive tract during the procedure. A breakaway vaginal speculum should be used to facilitate its removal before the injection of the radiopaque medium. Use a single-tooth tenaculum, applied to the anterior cervical lip, for traction of the uterus and to correct any anteroflexion or retroflexion of the uterus that interferes with a good view of the endometrial cavity. A Jarcho-type metal cannula with a plastic adjustable acorn or a balloon HSG catheter is used for the injection of the radiocontrast media. The use of water-based contrast media is preferable to oil-based media in order to avoid the risks of oil embolism and granuloma formation. Images 4-30 provide further information.
- Image 4 - Normal HSG finding with bilateral spillage
- Image 5 - Antero-retroflexion uterus
- Image 6 - Anteroflexion or retroflexion of the uterus
- Image 7 - Bilateral cornual obstruction
- Image 8 - Salpingitis isthmica nodosa
- Image 9 - Hydrosalpinx
- Image 10 - Tubal obstruction post–bilateral tubal ligation
- Image 11 - Tubal obstruction; Pomeroy bilateral tubal ligation
- Image 12 - Intravasation of the radiocontrast medium due to myoma
- Image 13 - Extravasation or lymphatic penetration of the radiocontrast medium
- Image 14 - Irregular cavity due to intramural fibroids intruding into the cavity
- Image 15 - Submucous fibroid
- Image 16 - Intrauterine synechiae
- Image 17 - Synechiae
- Image 18 - Endometrial polyp
- Image 19 - Placental polyp
- Image 20 - T-shaped uterus
- Image 21 - Fundal and right fibroid tubal obstruction
- Image 22 - Unicornuate uterus
- Images 23-27 - Bicornuate uteruses
- Image 28 - Didelphys uterus
- Image 29 - Double vagina
- Image 30 - Irregular endometrial cavity after myomectomy
- Ultrasonography
- Traditionally, obstetricians have used ultrasonography more frequently. In the 1980s, pelvic ultrasonography became an important tool in the evaluation and monitoring of infertile patients, especially during ovulation induction. Furthermore, pelvic ultrasonography should be part of the routine gynecologic evaluation because it allows a more precise evaluation of the position of the uterus within the pelvis and provides more information about its size and irregularities. Pelvic sonograms also help in the early detection of uterine fibroids and endometrial polyps and help demonstrate the presence of ovarian cysts, adnexal masses, and endometriomas. Ultrasonography helps in the diagnosis of anovulation, polycystic ovaries, and persistent corpus luteum cysts.
- Some common ultrasonographic findings are depicted in images 31-48, as follows:
- Image 31 - Sonogram (sagittal view of the uterus)
- Image 32 - Sonogram (sagittal view of the uterus; 3-laminar endometrial pattern)
- Image 33 - Sonogram (sagittal view of normal uterine cavity)
- Image 34 - Sonogram (transverse view of the uterus)
- Image 35 - Intramural fibroid
- Image 36 - Submucous myoma
- Image 37 - Endometrial polyp
- Image 38 - Ovarian cyst
- Image 39 - Dermoid cyst
- Image 40 - Endometrioma
- Image 41 - Corpus luteum
- Image 42 - Preovulatory follicle
- Image 43 - Multiple follicles during ovulation induction with human menopause gonadotropin (hMG)
- Image 44 - Polycystic ovary
- Image 45 - Preovulatory follicle in polycystic ovary during clomiphene citrate (CC) ovulation induction
- Image 46 - Uterine septum
- Image 47 - Uterus didelphys
- Image 48 - Sonohysterogram
- Magnetic resonance imaging
- As a diagnostic tool, the use of MRI has increased in recent years, although it should be limited to those patients in whom a definitive diagnosis cannot be ascertained based on conventional HSG, ultrasonography, and hysteroscopy findings.
- MRI is useful for delineating complex pelvic masses and for helping in the diagnosis of such conditions as congenital malformation related to cryptomenorrhea and absence of the cervix (Fedele, 1989).
- Hysteroscopy
- Hysteroscopy is a method of direct visualization of the endometrial cavity. The instrument used has evolved from the historical cystoscope and is based on the same principles (Pantaleoni, 1869; Rubin, 1925; Edstrom, 1970; Guerrero, 1972). The technology has changed substantially and now uses optical devices, videocamera-enhanced images, and television monitors, which allow more efficient participation and coordination of other members of the operating room team.
- The use of glycine and sorbitol solutions, different from the classic Hyskon, administered under constant pressure using an automatic pump, improves imaging resolution and is less risky to the patient. The diameter of the device has become smaller, making it more user friendly; thus, the procedure can be performed in the physician's office using local anesthesia (ie, paracervical block).
- Carbon dioxide hysteroscopy is for diagnostic purposes only and requires a constant flow of carbon dioxide. It does not require cervical dilation and allows a rather easy evaluation of the endometrial cavity (Lindeman, 1972).
- The operative hysteroscope has been designed based on the resectoscope principle (Neuwirth, 1976). It allows both the diagnosis and treatment of endometrial pathology. The design of refined instruments (eg, scissors, cautery loops, lasers) facilitated the treatment of pathologies such as uterine synechiae, endometrial polyps, and submucous myoma and the removal of foreign bodies (eg, intrauterine devices). In combination with specially designed catheters, it can be used to perform tubal cannulation (Goldrath, 1981; DeCherney, 1987; Vancaillie, 1989).
- Images 49-54 show some of the common hysteroscopy findings.
Endometrial factors
The endometrial lining constantly responds to the different hormonal secretions that occur during the menstrual cycle or to the exogenous administration of estrogen and progesterone. In the 1950s, Novack and Noyes published their findings on the microscopic changes of the endometrium throughout the menstrual cycle and established the criteria for endometrial dating (Noyes, 1950).
Jones first described the LPD and its association with recurrent pregnancy loss (RPL). LPD diagnosis is based on the lack of correlation between (1) endometrial development, diagnosed using premenstrual endometrial biopsy, and (2) the onset of the immediate menstrual cycle (Jones, 1949). To fulfill the diagnostic criteria, more than 2 days' difference must exist between the endometrial date and the beginning of the next menstrual period; furthermore, the same findings should be repeated in 2 consecutive menstrual cycles (Jones, 1977).
The serum progesterone level is not a good parameter for diagnosing LPD unless several serum determinations are performed on different days throughout the luteal phase to calculate the total amount of progesterone under the curve (Jones, 1974).
Images 55-58 show the endometrial changes during the menstrual cycle according to Novak and Noyes.
- Image 55 - Proliferative endometrium
- Image 56 - Early secretory endometrium
- Image 57 - Mid secretory endometrium
- Image 58 - Late secretory endometrium
LPD is associated with ovulatory dysfunction that is related to the following 3 factors. The first is, at the time of ovulation, follicle size is small. The second is when the preovulatory LH surge is rather blunt. Although the LH level is high enough to induce resumption of myosis of the oocyte, it might not be enough to induce follicular rupture and normal corpus luteum function (Soules, 1989). The third is when follicle size development is normal (ie, 23-24 mm in diameter), the LH surge prior to ovulation is normal, and serum progesterone levels are normal. However, the progesterone receptor at the endometrium level may require more progesterone because of decreased sensitivity; therefore, these patients require a greater amount of progesterone to respond adequately (Saracoglu, 1985).
Tubal factors
The fallopian tubes play an important role in reproduction. After ovulation, the fimbriae pick up the oocyte from the peritoneal fluid that has accumulated in the cul-de-sac. The epithelial cilia transport the oocyte up to the ampulla. The capacitated spermatozoa are transported from the endometrium through the cornual section and advanced through the fallopian tube down into the ampulla, where fertilization occurs. The embryo initiates its early cleaving stages and is propelled upward to arrive at the endometrial cavity at the blastocyst stage (ie, 96-120 h after ovulation).
Abnormalities or damage to the fallopian tube interferes with fertility and is responsible for abnormal implantation (eg, ectopic pregnancy). Obstruction of the distal end of the fallopian tubes accounts for accumulation of the normally secreted tubal fluid, creating distention of the tube with subsequent damage of the epithelial cilia.
Other tubal factors associated with infertility are either congenital or acquired. Congenital absence of the fallopian tube(s) can be due to spontaneous torsion in utero followed by necrosis and reabsorption. Elective tubal ligation and salpingectomy are acquired causes.
The 2 most frequent tests used for diagnosis of tubal pathology are HSG and laparoscopy (see Hysterosalpingogram and Laparoscopy).
Peritoneal factors
The uterus, ovaries, and fallopian tubes share the same space within the peritoneal cavity. The cul-de-sac is the reservoir for the peritoneal fluid that accumulates around the time of ovulation. The oocyte is released from the follicle and floats in the peritoneal fluid. The fimbriae of the fallopian tube pick up the oocyte within minutes after ovulation and transport it up to the ampullary portion, where fertilization occurs. This finely orchestrated mechanism is important for the normal reproductive process.
Anatomical defects and/or physiologic dysfunctions of the peritoneal cavity, including infection, adhesions, and adnexal masses, account for infertility. PID, peritoneal adhesions secondary to previous pelvic surgery, endometriosis, and ovarian cyst rupture all compromise the motility in the fallopian tubes or produce blockage of the fimbriae with development of hydrosalpinx. Large myomas, pelvic masses, or blockage of the cul-de-sac interferes with the accumulation of peritoneal fluid and interferes with the normal oocyte pickup mechanism. Periovarian adhesions that encapsulate the ovary interfere with the normal oocyte release at ovulation, becoming a mechanical factor for infertility.
- Laparoscopy
- The laparoscope is one of the greatest developments in gynecologic instrumentation. Its origin dates to the pioneering work of Jacobaeus in 1910 (Jacobaeus, 1910). The laparoscope was first used to visualize the pelvic cavity. The procedure was abandoned in the 1930s because of fatal complications.
- In the 1950s, a new generation of laparoscope was developed using a fiberoptic technique; later, safer electrocautery techniques resurrected the application and use of operative laparoscopy, especially for sterilization purposes and for diagnosis of ectopic pregnancy (Frangenheim, 1964). In 1970, Semm advanced the field of operative laparoscopy with the development of numerous accessory instruments (Semm, 1979; Feste, 1985). Semm opened the doors to new surgical applications and forever changed the traditional way of practicing gynecologic surgery (Gomel, 1983).
- Laparoscopy is indicated as the last test in the evaluation of infertility because of the risks, the need for anesthesia, and the operative cost. The only exception is when a known medical history directs attention to a pelvic factor as the cause of infertility.
- Laparoscopy is contraindicated in patients with probable bowel obstruction (ileus) and bowel distention, cardiopulmonary disease, or shock due to internal bleeding. Because of the risk of bowel perforation, uterine and pelvic vessel injury, and bladder trauma, a skilled and experienced surgeon must perform the procedure. Relative contraindications include massive obesity, large abdominal mass or advanced pregnancy, severe pelvic adhesions, and peritonitis.
- Images 60-82 show laparoscopic findings associated with infertility.
- Image 60 - Healthy uterus, ovaries, and fallopian tubes
- Image 61 - Bicornuate uterus
- Image 62 - Bicornuate uterus and ectopic pregnancy
- Image 63 - Uterine fibroids
- Image 64 - Hydrosalpinx
- Image 65 - Peritubal and ovarian adhesions
- Images 66-67 - Frozen pelvis
- Images 68-69 - Pelvic endometriosis
- Images 70-71 - Endometrioma
- Image 72 - Tubal ligation
- Image 73 - Uterus (tubal ligation)
- Image 74 - Bicornuate uterus (ectopic pregnancy)
- Images 75-76 - Ectopic pregnancy
- Image 77 - Paratubal cyst
- Images 78-79 - Septate uterus
- Image 80 - Septate uterus and pedunculated fibroid
- Images 81-82 - Polycystic ovaries
Ovarian factors
Oogenesis occurs in the ovary from the first trimester of embryonic life and is completed by 28-30 weeks of gestation. By then, approximately 7 million oocytes are present. They are arrested at the prophase stage of the first meiosis division. Subsequently, the number of oocytes decreases because of a continuous process of atresia. At birth, the pool of oocytes is reduced to approximately 2 million. By menarche, approximately 500,000 oocytes are present. Those oocytes are used throughout the reproductive years until menopause.
The ovulatory process is initiated once the hypothalamus-pituitary-ovarian axis matures and FSH and LH, under the regulation of gonadotropin-releasing hormone (GnRH), acquire their normal secretory patterns. From the cohort of follicles available each month, only a single oocyte is selected, establishes dominance, and develops to the preovulatory stage. During follicular development, the granulosa cells secrete increasing amounts of estradiol (E2), which, initially, through down-regulation, decreases the secretion of FSH. Later, through a positive feedback mechanism, E2 participates in the LH surge that triggers the ovulatory process, induces the resumption of meiosis by the oocyte, and stimulates the formation of the corpus luteum and subsequent progesterone secretion.
Ovulatory dysfunction is defined as an alteration in the frequency and duration of the menstrual cycle. A normal menstrual cycle lasts 25-35 days, with an average of 28 days. Failure to ovulate is the most common ovulatory problem. Absence of ovulation can be caused by primary amenorrhea or premature menopause, indicative of the depletion of oocytes or absence of the ovaries.
Oligomenorrhea (irregular or delayed ovulation) is related to dysfunction of the hypothalamus-pituitary-ovarian axis; the most common cause is PCOD (Goldzieher, 1981; Conway, 1989).
Primary amenorrhea is the absence of a spontaneous menstrual period by age 16 years or after 3 years of pubarche and telarche (Frisch, 1971; Knobil, 1988). Primary amenorrhea is generally related to gonadal development failure, as in Turner syndrome, in which the karyotype 45,X indicates an absence of the X chromosome. These patients present with sexual infantilism associated with short stature, webbed neck, and cubitus valgus. Streak gonads replace their ovaries, but they have a small uterus and normal fallopian tubes and vagina. This condition is associated with elevation of FSH and LH in serum (menopausal stage) and low estrogen levels.
Other chromosome abnormalities include 46,XX, which is associated with partial deletions of the short or long arm of one of the X chromosomes, and mosaicism (eg, X/XXX; X/XX/XXX; pure gonadal dysgenesis; 46,XX; 46,XY).
Primary amenorrhea also occurs in patients with hypothalamic failure secondary to inadequate GnRH synthesis, neurotransmitter defects, or isolated gonadotropin insufficiency. Other entities associated with primary amenorrhea include congenital absence of the uterus, vagina, or hymen (cryptomenorrhea) and enzyme deficiencies such as 17-alpha hydroxylase deficiency and 17,20-desmolase deficiency.
Secondary amenorrhea that is not related to a pregnancy is the absence of ovulation for more than 6 months. This condition is related to a severe dysfunction of the endocrine system and can be related to thyroid, adrenal, and pituitary disorders, including tumors. However, more frequently, secondary amenorrhea is related to premature ovarian failure.
- Classification of premature ovarian failure
- Ovarian follicle depletion
- Pure gonadal dysgenesis
- Idiopathic
- Congenital ovarian torsion
- Turner syndrome - Mosaicism variant
- Fragile X chromosome
- Galactosemia
- Autoimmune
- Viral oophoritis
- Idiopathic
- Ovarian follicle dysfunction
- 17-alpha hydroxylase deficiency
- 17,20-desmolase deficiency
- Cholesterol desmolase deficiency
- Gonadotropin-receptor blocking immunoglobulins
- Antibodies to gonadotropins
- Idiopathic - Resistant ovary syndrome
- Ovarian follicle depletion
Oligomenorrhea is a dysfunction of the hypothalamus-pituitary-ovarian axis and is the most common ovulatory disorder associated with infertility. Patients with this disorder present with a history of irregular menstrual cycles that fluctuate from 35 days to 2-5 months, sometimes associated with a history of dysfunctional uterine bleeding or prolonged periods of breakthrough bleeding. Patients may have symptoms of hyperandrogenism, acne, hirsutism, and baldness. Obesity is frequently associated and aggravates the prognosis. Although these patients are not sterile, their fertility is decreased, and the obstetrical outcome is poor because of an increased history of pregnancy losses.
In patients with PCOD, the gonadotropin assay reveals a normal to slightly low FSH concentration and a slight or very elevated LH level. Prolactin, thyroid, and adrenal hormones can be elevated if a combination of glandular dysfunction (pluriglandular syndrome) is present. Pelvic ultrasonographic findings confirm the presence of enlarged ovaries with multiple small (<10 href="http://emedicine.medscape.com/article/274143-media">Image 44).
A monophasic BBT is associated with anovulatory cycles, although this is not a reliable finding in 20% of patients. A determination of serum progesterone level below 2 ng, an endometrial biopsy finding that shows a proliferative pattern, and pelvic ultrasonography results that show absence of a follicle and/or a corpus luteum are better criteria for diagnosing lack of ovulation.
Infertility Treatment
A consultation with the infertile couple once the evaluation has been completed is imperative. Outline a treatment plan according to the diagnosis, duration of infertility, and the woman's age. If pregnancy has not been established within a reasonable time, consider further evaluation or a different treatment plan. In many instances, patients are not well advised; therefore, their expectations are unrealistic, and the lack of success exacerbates their anxiety and frustration levels.
Treatment of cervical factors
An abnormal PCT result attributable to chronic cervicitis may be treated with antibiotics. Treatment of reduced secretion of cervical mucus due to destruction of the endocervical glands by previous cervical conization, freezing, or laser vaporization responds poorly to low-dose estrogen therapy. The easiest and most successful treatment is intrauterine insemination (IUI) (Te Velde, 1989). Similar treatments apply when the abnormal PCT result is related to oligospermia or hypospermia and to ejaculatory disorders such as impotence, hypospadias, or retrograde ejaculation (Moghissi, 1977; Nachtigall, 1979; Brassesco, 1988). Patients with azoospermia that is not amenable to IVF/ICSI treatment benefit from artificial insemination (AI) with donor sperm (Goss, 1975).
AI can be performed by depositing the sperm at the cervical level (cervical insemination [CI]) (Nachtigall, 1979) or inside the endometrial cavity (IUI). CI has almost been abandoned because of its low success and has been relegated only to cases in which the sperm count is normal, such as in AI using donor sperm.
For IUI, IVF, and ICSI procedures, the removal of certain components of the ejaculate (ie, seminal fluid, excess cellular debris, leukocytes, morphologically abnormal sperm) with the retention of the motile fraction of sperm is desirable.
For most specimens, the greatest recovery of the motile portion results from separation via centrifugal filtration through a discontinuous density gradient system. However, for certain very poor specimens with low original concentrations of motile sperm, the use of the gradient system results in such a negligible recovery as to render it useless. The recourse for these specimens is to remove the seminal fluid by successive media washes.
A small number of specimens have acceptable original concentrations of motile sperm but poor recoveries with the gradient system. These specimens benefit most from layering a washed pellet of sperm with nutrient media and allowing the motile fraction to swim up into the media before being separated (Kerin, 1989).
After sperm preparation, the spermatozoa are enhanced in motility and become activated and ready to fertilize an oocyte. Currently, no reliable technique allows the separation of the X- and Y-bearing spermatozoa. IUI is performed during a natural cycle or after ovulation induction with CC or hMGs. The procedure is performed 30-34 hours after the spontaneous LH surge or 40-44 hours after the administration of 10,000 U of hCG (Silverberg, 1992). The sperm is delivered into the endometrial cavity using an IUI cannula. After injection of the sperm, the patient remains in the recumbent position for 10 minutes. Prophylactic antibiotics are not necessary unless a medical indication exists (eg, history of mitral valve prolapse).
The average pregnancy rate achieved after a natural-cycle IUI is 8%. The rate increases to 10-12% after CC ovulation induction and to 12-15% per cycle after hMG/hCG ovulation induction. Of the successful pregnancies, 85% are achieved within the first 4 cycles of IUIs.
Homologous insemination refers to the use of sperm from the patient's partner. Heterologous or therapeutic insemination (TI), formerly called artificial insemination by donor sperm, refers to the use of frozen sperm that has been quarantined for at least 6 months (Goss, 1975). Thereafter, the specimen is ready to use once the donor has undergone the necessary screening tests required by the tissue bank, the US Food and Drug Administration (FDA), and the American Society for Reproductive Medicine (ASRM) (American Fertility Society, 1991). The source of the sperm can be either anonymous or from a designated donor previously accepted by the infertile couple. A cumulative pregnancy rate of 80% is achieved during the first 6 cycles of TI.
Treatment of uterine factors
Until IVF became available, a patient with congenital absence of the uterus and vagina (Rokitansky-Küster-Hauser syndrome) had no chance to have a biologic child. Today, it is feasible by using a surrogate mother or gestational carrier. The treatment for patients with congenital absence of uterus and vagina consists of the creation of a neovagina for their normal sexual function (McIndoe, 1938). Once patients desire to have children, they proceed with stimulation of the ovaries, oocyte aspiration, and IVF, but the embryos are transferred to a gestational carrier (see In vitro fertilization).
The treatment of uterine malformations depends on the severity of the problem. Fertility is not an issue for some patients affected by DES, and they remain undiagnosed until they have an abnormal Papanicolaou test (Pap smear) result. Those who do have fertility problems are treated according to the following guidelines (Kaufman, 1977; Haney, 1979; Barnes, 1980):
- Chronic cervical factor of absence of mucus - Intrauterine insemination
- Cervical incompetence - Cerclage
- Damage/absence of fallopian tubes (ectopic) - In vitro fertilization
Unicornuate uterus
A unicornuate uterus remains undetected unless fertility is compromised. Patients with this type of uterus can have a normal term of pregnancy. Most problems are related to premature labor and pregnancy loss. No treatment is available for this condition, and the patient eventually delivers a child after several pregnancy losses (DeCherney, 1986). In conjunction with a unicornuate uterus, 15% of patients are born without a kidney or with a pelvic kidney. Most patients are asymptotic; therefore, an intravenous pyelogram must be performed.
Bicornuate uterus
A bicornuate uterus causes only minimal problems with infertility (if any). A bicornuate uterus can be associated with a history of recurrent miscarriages, and its repair is indicated only if other etiologies for the miscarriage have been excluded (see Surgical intervention).
Arcuate uterus
In general, an arcuate uterus does not cause infertility. Whether it should be corrected in cases of primary infertility is debatable.
Septate uterus
The hypothesis that a uterine septum can cause infertility is controversial. It is difficult to advise surgery in cases of primary infertility. The basis for removal of the septum is related to the deficiency in blood supply in case the implantation of the embryo occurs at the septum level.
Surgical interventionUterine anomalies can be corrected through operative hysteroscopy under general anesthesia or conscious sedation (DeCherney, 1986). Ideally, perform the procedure during the early follicular phase and under laparoscopic surveillance to decrease the risk of uterine perforation. Furthermore, the laparoscopy helps in the differential diagnosis between a septate uterus and a bicornuate uterus. A bicornuate uterus is characterized by the presence of an indentation at the fundus.
The 2 techniques are the Strassman metroplasty and the Jones metroplasty. The Strassman metroplasty consists of performing an incision at the fundus of the uterus between both cornual areas and closing the defect with an anteroposterior suture. The Jones metroplasty consists of resecting the septum using an anteroposterior wedge incision and closing the defect in the same direction (Adamson, 1992; Jones, 1992; Khalifa, 1993) (see Images 83-84).
Uterine synechiae
Uterine synechiae are corrected using operative hysteroscopy. The surgery is performed during the early follicular phase. A good distention of the endometrial cavity is necessary to perform the procedure with minimal risk of uterine perforation. Once the defect is removed, leaving an intrauterine stent for 7 days is advisable to prevent a recurrence of adhesions. The patient should receive prophylactic antibiotics and uterine relaxants (eg, ibuprofen) during these 7 days to prevent infection and stent expulsion, respectively. The patient should be prescribed a high dose of E2 (5 mg qd for 21 d) followed by medroxyprogesterone (10 mg for 10 d). A postoperative HSG should be performed 2 months later. In many instances, more than one hysteroscopy is required for total resection.
Endometrial polyps
Endometrial polyps are removed through operative hysteroscopy associated with a dilatation and curettage, if necessary. An HSG follow-up procedure is not necessary. To prevent further polyp development associated with anovulation, the patient should have withdrawal bleeding at least every 6 weeks.
Myoma treatment
In general, small and asymptomatic myomas do not require treatment, but the patient should be periodically monitored. The fibroids should be treated if they are associated with hypermenorrhea or menometrorrhagia or if they are causes of infertility. Three modalities are used to treat myomas: medical treatment, surgical treatment, and embolization.
- Medical treatment is a temporary treatment, ideally used for patients who are close to menopause and/or candidates with great risk during major surgery. However, medical treatment can be used to reduce the myoma size prior to its removal. GnRH analog ([GnRHa], leuprolide acetate, nafarelin acetate, goserelin acetate) causes down-regulation of the pituitary, inducing chemical menopause after injections of 3.75 mg intramuscularly every 4 weeks for a period of up to 6 months (Andreyko, 1988; Schlaff, 1989; Chipato, 1991; Adamson, 1992). Disadvantages of this treatment are symptoms of menopause, osteoporosis, and recurrence of the myomas after discontinuation of the treatment.
- Surgical treatment of myomas is indicated in cases of hypermenorrhea, menometrorrhagia, and when the myoma is implicated in recurrent miscarriages or in interfering with embryo implantation. Three surgical techniques are described: conventional laparotomy, operative laparoscopy, and operative hysteroscopy.
- Laparotomy: This technique is indicated for large myomas, for submucous myomas larger than 3 cm in diameter, or for myomas that, regardless of being submucous, have a portion of the myoma that compromises the myometrium so that a complete resection through the hysteroscopy is not feasible.
- Operative laparoscopy: This technique is indicated for pedunculated and superficial intramural myomas. Use this technique for myomas with a diameter less than 6 cm (Dubuisson, 1991). Several cases have been reported of uterine rupture during pregnancy because the reconstruction of the uterus after laparoscopic myomectomy was not as good as a myomectomy performed using laparotomy (Harris, 1992).
- Operative hysteroscopy: The removal of a submucous fibroid using hysteroscopy should be limited to small fibroids (<3>
- For myoma embolization, uterine fibroid embolization (UFE) consists of catheterization of the uterine artery and the injection of microbeads of polyvinyl alcohol to selectively occlude the circulation of the fibroid. The procedure is performed by the interventional radiologist and requires overnight admission for the patient (Hurst, 2000).
- Indications: Indications for UFE include (1) patients with history of uterine bleeding related to the fibroid who are poor candidates for major surgery, (2) patients who are not interested in reproduction, and (3) patients who want to preserve their menstrual function as a sign of youth or because of a misconception regarding femininity.
- Results: Following the embolization, patients may experience lower abdomen pain for approximately 1 week. The reduction of the fibroid depends on its size and the success of blocking its blood supply, and several weeks may elapse before symptoms (vaginal bleeding) subside (Ravina, 1995).
- Complications: Although complications appear minimal (perhaps underreported), cases of pelvic infection subsequent to necrosis of the tissue have been reported. In 1-2% of patients, embolization compromises the ovarian blood supply, resulting in undesirable menopause (Vedantham, 1997). This potential complication is a major consideration in patients who have this type of procedure because of the interest in future pregnancies.
- Long-term effects: Long-term effects are unknown because UFE has only been in clinical practice since 1991.
Treatment of endometrial factors
The treatment of LPD should be oriented to treat the underlying factor; therefore, having a precise diagnosis of the ovulatory dysfunction is crucial.
Patients who have a small follicle size at the time of ovulation should be treated with CC (50 mg qd for 5 d, starting on the second menstrual cycle d). Monitor follicular development using pelvic ultrasonography starting on the 10th menstrual cycle day and continue until the day of ovulation. Ideally, the follicle should be ovulated when the diameter is 23-24 mm.
Blunt preovulatory LH surge is associated with LUF syndrome. Administer hCG (10,000 U) during a natural menstrual cycle when the follicle reaches 23-24 mm in diameter.
Patients with normal follicle development, normal LH surge, and corpus luteum detected by ultrasonography, in whom the endometrial biopsy result still lags behind the normal date, should be treated with exogenous progesterone (eg, vaginal supp 50 mg qd), micronized progesterone (200 mg q6h), or oral progesterone (200 mg q8h). Start progesterone supplementation 48 hours after ovulation.
Document the response to treatment of LPD by repeating the endometrial biopsy during the first cycle of treatment. The patient should continue the progesterone treatment for 2 weeks and should not discontinue the progesterone unless a pregnancy test result is negative. If the patient conceives during the treatment, continue progesterone until the 10th week of pregnancy. By this time, the placenta takes over the endocrine control of the pregnancy. Of the patients in whom the LPD treatment is adequate, 70% conceive during the first 6 months of treatment. If pregnancy has not occurred during that interim, further fertility evaluation is required to exclude other associated factors that interfere with the success of the treatment. Remember that other lethal factors account for RPL (eg, chromosome abnormalities), and the patient will have another miscarriage regardless of a corrected LPD.
Treatment of tubal and peritoneal factors
The treatment of tubal-factor infertility underwent major changes, especially during the last quarter of the century when microsurgery became available (Winston, Fertil Steril, 1980; Gomel, 1992). Tubal reconstruction was the only hope for those patients before ART became available.
Because of the intimate relationship between the fallopian tubes and the other pelvic organs and because, in the great majority of the cases, peritoneal pathology involves tubal pathology, the treatments of these factors are discussed together.
Tubal and peritoneal factor infertility treatment requires a good surgeon who is skilled in currently available techniques (Rock, 1984). The patient's age and the severity of the tubal pathology play important roles in the selection of patients, as do any other infertility issues such as the presence of endometriosis and severe pelvic adhesions. Before surgery, review the HSG films and the results of previous laparoscopy, if available, to decide on the type of surgical technique that is required and to explain to the patient the expected degree of success and risks involved with the procedure.
Tubal obstruction and lysis of adhesions can be corrected through laparotomy, operative laparoscopy, and, in special circumstances, through operative hysteroscopy and tubal cannulation.
Laparotomy is indicated in patients with severe pelvic adhesions that compromise the bowel, ovaries, and tubes, with obliteration of the cul-de-sac (Ismajovich, 1984). The aim of the procedure is to correct what is necessary to allow the normal transport of the gametes; complete restoration of the anatomy is not intended (Caspi, 1979). Lysis of adhesions should be meticulous, using hydrodissection and fine instruments. Avoid blunt dissection. Constant irrigation with Ringer lactate solution and heparin prevents fibrin formation. Meticulous hemostasis is imperative (Rock, 1978; Jones, 1983; Murphy, 1992).
Operative laparoscopy was reintroduced into the surgical armamentarium in the 1950s; however, in the 1970s, Semm developed different procedures and operative instruments that currently allow for the outpatient surgical treatment of multiple tuboperitoneal pathologies (Semm, 1979). Electrocautery, endocoagulation, lasers, and ultrasonography scalpels facilitate the performance of operations that otherwise required a laparotomy a few years ago.
Operative hysteroscopy associated with tubal cannulation is helpful to treat cornual obstruction.
Treat fimbrial phimosis and periadnexal disease with laparoscopy (Gomel, 1977; Mettler, 1979; Daniel, 1984; Donnez, 1987; Schlaff, 1991). The pregnancy rate after salpingolysis is 50-60% during the first year after treatment. Fimbrioplasty for fimbria agglutination or phimosis without destruction of the cilial epithelium is equally successful. The incidence rate of ectopic pregnancy after surgery is in the range of 5%.
Treatment of hydrosalpinx (distal tubal obstruction) with salpingostomy can be performed through microsurgery or operative laparoscopy. No difference in the pregnancy rate occurs if a skillful microsurgeon or laparoscopist performs the salpingostomy. The success of the procedure is related to the diameter of the hydrosalpinx and to the damage to the cilial epithelium. If the cilial epithelium has been destroyed, the outcome of the procedure is poor, and it is better to perform a salpingectomy in preparation for future IVF. The pregnancy rate fluctuates from 20-35%, and the expected ectopic pregnancy rate is as high as 20%.
Treatment of cornual obstruction (proximal) should start by confirming the diagnosis. In many cases, cornual obstruction diagnosed based on HSG findings can be a false obstruction caused by a simple cornual spasm. Before performing a tubocornual anastomosis, the patient must have a diagnostic laparoscopy associated with tubal cannulation by hysteroscopy (Confino, 1988; Novy, 1988; Confino, 1990). If one tube remains open, anastomosis is not needed because pregnancy can be achieved in 50% of cases. The success rate of tubocornual anastomosis fluctuates from 20-58%. The ectopic pregnancy rate is 5-7%. If the obstruction is caused by salpingitis isthmica nodosa or fibrosis, the best results are achieved through IVF (Schoysman, 1984).
Surgical preparation for IVF
The surgeon should be as conservative as possible but should bear in mind that the patient is better served with a single well-functioning fallopian tube than 2 defective tubes, which elicits an increased risk for ectopic pregnancy or recurrence of pelvic adhesions. If the fallopian tubes are beyond repair, bilateral salpingectomy with destruction of the cornual area is recommended in preparation for IVF.
Tubal obstruction due to elective sterilization is better repaired with microsurgery, although the modern tendency is to perform the anastomosis using operative laparoscopy (Winston, Clin Obstet Gynecol, 1980; Jansen, 1986; Rock, 1987). In either event, knowing in advance what type of tubal ligation technique was used is important. Unfortunately, tubal cauterization destroys a great deal of tissue, so that the amount of fallopian tube remaining is often not long enough to facilitate a successful reanastomosis.
Before anastomosis, evaluate the patient using HSG and laparoscopy findings to measure how far proximal and distal fragments of the fallopian tubes remain from the tubal ligation. To have a successful reanastomosis, the final tube should measure at least 4.5 cm. If fimbriectomy was performed, no treatment is available other than IVF. The best candidates for tubal reanastomosis are patients who had tubal ligation by the method of fallopian ring, Hulka clip, or Pomeroy (Rock, 1987). The pregnancy rate following a tubal reanastomosis performed by a skillful surgeon fluctuates from 70-80%. The ectopic pregnancy rate is approximately 7%.
Treatment of endometriosis
Endometriosis treatment may be divided according to the severity of the disease and patient needs. Four alternatives are currently available to treat endometriosis: expectant therapy, surgical intervention, medical treatment, and combined therapy.
- Expectant therapy
- Expectant therapy should be based on a complete workup with diagnosis of very early stages of the disease (minimal) in patients without clinical symptoms, ie, an incidental finding (Olive, 1989). A second-look laparoscopy is required for follow-up observation within 6-18 months.
- Considering the unpredictability of the disease and its tendency to advance over time, this approach is worrisome (Thomas, 1987).
- Surgical treatment
- Surgical treatment should be directed at destroying the disease using electrocoagulation, laser vaporization, or endocoagulation (Vancaillie, 1989). Perform removal of the endometriomas through ovarian cystectomy, lysis of adhesions, and uterine suspension to complete the treatment and to prevent postoperative adhesions.
- Laparotomy was the criterion standard for surgical treatment. Most surgical treatment for endometriosis is currently performed through operative laparoscopy (Hasson, 1979; Keye, 1987; Olive, 1989). Laparotomy has been relegated to the treatment of severe disease or if a need for hysterectomy arises (Olive, 1989).
- Medical treatment
- Medical treatment is directed toward suppressing estrogen production by the ovary. Different modalities of treatment are available. Depending on the therapeutic agent and the duration of treatment, endometriosis can be treated with oral contraceptives, progestins, androgens, or GnRH agonists.
- The progestins that can be used and the doses are as follows:
- Medroxyprogesterone acetate (eg, Provera 40-60 mg PO qd, Depo-Provera 200 mg IM q2wk)
- Megestrol acetate (eg, Megace 20-40 mg PO qd)
- Norethindrone acetate (eg, Aygestin 15 mg PO qd) (Johnston, 1976; Hasson, 1979; Hull, 1987; Keye, 1987; Telimaa, 1987; Olive, 1989)
- The androgens used are 17-ethinyl testosterone derivatives (eg, Danazol 400-800 mg PO qd) (Dickey, 1984; Henzl, 1988; Bayer, 1989)
- The GnRH agonists used are as follows:
- Leuprolide acetate (eg, Lupron 3.75 mg IM q4wk or 11.25 mg IM every 3 mo)
- Nafarelin acetate (eg, Synarel 400 mcg IN qd)
- Goserelin acetate (eg, Zoladex 3.6 mg SC q4wk or 10.8 mg SC every 3 mo)
- GnRHa therapy can be administered along with cyclic or continuous progestins or with cyclic or continuous estrogen and progestins in cases of severe hot flashes (Henzl, 1988; Olive, 1989).
- Combined therapy
- Medical and surgical treatments are usually combined for the treatment of severe endometriosis. No consensus exists as to whether the medical treatment should precede surgery or vice versa (Wheeler, 1981; Ronnberg, 1984; Buttram, 1985). Those who prefer medical treatment first argue that the size of the endometriosis decreases; therefore, surgery will be easier and shorter. Those who prefer surgery first argue that because the size of endometriosis decreases, lesions that cannot be observed during surgery may be present; therefore, the operation is less than ideal and is associated with an increased chance for early recurrence.
- Regardless of the treatment approach, establish a 6- to 12-month interval during which a spontaneous pregnancy is expected to occur. Otherwise, a second-look laparoscopy is indicated for further evaluation of recurrent endometriosis and to exclude any other infertility factor.
- A more proactive approach now exists. Ovulation induction and IUI are used after completion of the treatment in hopes of expediting the establishment of a pregnancy before relapse of the disease (Chaffkin, 1991; Dodson, 1991; Fedele and Bianchi, Fertil Steril, 1992).
Treatment of ovarian factors
Ovulation induction is the treatment for infertile patients who still have oocytes within the ovaries but in whom a dysfunction of the hypothalamic-pituitary-ovarian axis exists. The ovulation induction agents used include CC, hMG, hCG, recombinant FSH, and recombinant LH.
Clomiphene citrate (Clomid, Serophene)
The chemical formula for CC is 2-[p -(2-chloro-1,2-difhenylvinyl) phenoxy] triethylamine dihydrogen citrate. CC is a nonsteroidal estrogen capable of interacting with estrogen receptor–binding proteins in a manner similar to estrogen but in a more prolonged way (Clark, 1974; Clark 1981). Therefore, CC behaves similar to an antiestrogen.
CC has been in clinical use since the early 1960s. Its mechanism of action is still not well understood, but it competes for the estrogen receptor at the hypothalamus, pituitary, and ovarian levels. Because of the action at the estrogen-receptor level within the hypothalamus, CC alleviates the negative feedback effect exerted by endogenous estrogens (Tobias, 1981; Adashi, 1984; Kokia, 1990). As a result, CC normalizes the GnRH release; therefore, the secretion of FSH and LH is capable of normalized follicular recruitment, selection, and development to reestablish the normal process of ovulation (Tobias, 1981; Miyake, 1983).
The standard dose of CC is 50 mg PO qd for 5 days, starting on the fifth menstrual cycle day or after progestin-induced bleeding. As an antiestrogen, CC requires that the patient have some circulating estrogen levels; otherwise, the patient will not respond to the treatment. The CC response is monitored using pelvic ultrasonography starting on the 12th menstrual cycle day. The follicle should develop to a diameter of 23-24 mm before a spontaneous LH surge occurs.
BBT can be used to observe the thermogenic shift (temperature rises 0.5°F above the basal level) induced by the early secretion of progesterone. The only disadvantage with BBT is that in many instances, the shift does not occur in a clear way, and the patient misses the time of ovulation. While BBT is an inexpensive way to monitor ovulation, it is often impractical.
Urinary monitoring of the LH surge (eg, with an LH Predictor Kit) can be a substitute for BBT. The patient should start monitoring the urinary LH secretion daily starting on the 12th menstrual cycle day. Ovulation usually occurs within the 32-40 hours after the indicative color change. Serum LH determination is more precise, especially when performed in combination with pelvic ultrasonography. A postovulatory ultrasonography should be performed during the first CC cycle to exclude the presence of LUF syndrome.
Because of the antiestrogenic effect, CC may desiccate the cervical mucus, creating an iatrogenic cervical factor that can be responsible for the lack of pregnancy in a patient who has otherwise ovulated (Shirai, 1972). Therefore, a PCT must be performed during the first CC cycle and every time the doses of CC are increased. Other adverse effects associated with CC are hot flashes, scotomas, dryness of the vagina, headache, and ovarian hyperstimulation, which, although rare, has been reported in patients who are sensitive to CC (Southam, 1962; Scommegna, 1969). Whether the use of CC increases the risk of ovarian cancer is unknown, although 2 articles illustrate a potential risk (Rossing, 1994). Other authorities disagree with this assumption.
The principal indications for CC use are in patients with oligomenorrhea, especially PCOD, and for patients with slight menstrual irregularities. CC is indicated in the treatment of patients with LPD due to small-size follicle development (van Hall, 1969; Garcia, 1977). Its use has been extended to ART.
The use of CC is contraindicated in cases of ovarian cyst, pregnancy, and liver disease. Its use is controversial in patients with a history of breast cancer.
Human menopause gonadotropins
Crowe discovered that the gonads were under the control of the anterior hypophysis (Crowe, 1966). Zondek and Aschheim discovered that FSH and LH were responsible for the development of the gonads in immature animals and confirmed Crowe's work (Mettler, 1979; Daniel, 1984; Schlaff, 1991). In the 1930s, ovulation induction was attempted by using gonadotropins from a mare, but its use was discontinued because of the development of antibodies (Cole, 1930; Leethem, 1948).
Borth et al demonstrated the effect of FSH and LH extracted from menopausal urine (Borth, Experientia, 1954; Borth, Human Pituitary Gonadotropins, 1961). Gemzell reported the first ovulation induction using human pituitary gonadotropin in 1958, and the first pregnancy was reported in 1960 (Gemzell 1958; Gemzell, 1960). Lunenfeld reported preliminary results using hMG; however, in 1963, it was definitely established as a real ovulation induction agent (Lunenfeld, 1963; Domini, 1964; Spadoni, 1974).
hMG (Repronex) contains 75 U of FSH and 75 U of LH per mL, although the concentration may vary among batches (ranges from FSH at 60-90 U and LH at 60-120 U). In the 1980s, a pure form of FSH became available. Bravelle contains 75 U of FSH. The new generations of available gonadotropins are produced by genetically engineered mammalian cells (ie, Chinese hamster ovary cells), in which the gene coding for the alpha and beta FSH subunits has been inserted (Gonal F and Follistim) (Devroey, 1992). Recombinant LH is currently undergoing clinical trials.
The administration of hMG and its derivatives should be under the direct supervision of a reproductive endocrinologist. An ultrasonography unit and an endocrine laboratory capable of performing daily determinations of E2, FSH, and LH are necessary (Cabau, 1981; Garcia, 1983; Hackeloer, 1984; Lequin, 1986).
Multiple adverse effects and complications may occur during the use of the gonadotropins, to include (1) multiple pregnancy (24-33%); (2) ectopic pregnancy (5-8%); (3) miscarriages (15-21%); (4) ovarian torsion and rupture; and (5) ovarian hyperstimulation syndrome (OHSS), which is the most severe (Schenker, 1979; Brown, 1986). Whittemore et al, using a large combined data set derived from case-controlled studies in the United States, showed that the increase of ovarian cancer associated with infertility might be due to the use of fertility drugs (Whittemore, 1992).
OHSS is an iatrogenic condition that occurs in patients undergoing ovulation induction with hMG or controlled ovarian hyperstimulation (COH) for ART. The incidence rate fluctuates from 0.1-2%. The pathophysiology of the disease is not well understood, but a massive extravascular accumulation of fluid occurs that is associated with a severe depletion of the intravascular volume responsible for dehydration, hemoconcentration, and electrolyte imbalance (ie, hyponatremia, hyperkalemia) (Tollan, 1990). OHSS can be mild, moderate, or severe (Golan, 1989).
Mild OHSS is characterized by ovarian enlargement (up to 5 cm in diameter), minimal ascites, and weight gain of less than 10 lb. Moderate OHSS is characterized by ovarian enlargement (5-12 cm in diameter) moderate ascites, nausea, vomiting, abdominal discomfort, and weight gain greater than 10 lb. Severe OHSS is characterized by easily palpable ovaries, severe ascites, nausea, vomiting, diarrhea, shortness of breath, hydrothorax, peripheral edema, oliguria, hemoconcentration (eg, hematocrit level >48% and hemoglobin level >16 g), and creatinine level greater 1.6 mg/dL. Renal failure and thrombosis can occur if the patient is not treated correctly (Whelan, 2000).
Some patients have a greater risk of developing OHSS. They usually are young patients with a history of oligo-ovulation who responded with elevated E2 levels (3000 pc/mL) and multiple follicles (>15) and patients in whom the ovulation has been triggered by the administration of exogenous hCG (Navot, 1988; Asch 1991).
OHSS usually has 2 phases. The first phase develops between the second and seventh day after ovulation, and the second phase only occurs if the patient becomes pregnant. The first phase is self-limited, and the symptoms subside by the end of the luteal phase and by the beginning of the next menstruation (Lyons, 1994).
Patients with mild and moderate OHSS are treated at home with bedrest and strict control of fluid intake and output. If weight gain greater than 2 lb occurs, evaluate the patient to determine if hospitalization is required.
Patients with severe OHSS must be hospitalized and confined to bed, with strict control of fluid intake and output. Intravenous fluids (ie, isotonic sodium chloride solution) must be administered until hemodilution is achieved; at this point, diuresis must be forced with intravenous furosemide. If the urinary output remains low, albumin 25% (50 mL/h IV for 4 h) has been effective in promoting diuresis. Perform abdominal paracentesis if the patient becomes uncomfortable. Thoracentesis is indicated in cases of pleural effusion. Because of the risk of thrombosis, heparin (5000 U SC q12h) is recommended (Whelan, 2000).
hMG and its derivatives are indicated for ovulation induction in patients with primary amenorrhea due to hypopituitarism and in patients with secondary amenorrhea who did not respond to CC ovulation induction. For the past 20 years, hMG and its derivatives have been the first choice for COH in ART.
Gonadotropin-releasing hormone
Synthetic GnRH (eg, Factrel, Gonadorelin, Lutrepulse) has a chemical composition similar to native GnRH and is indicated for patients with hypothalamic dysfunction, especially those who do not respond to CC (Zacur, 1985; Schriock, 1986). This drug is administered in a pulsatile fashion every 60-120 minutes, intravenously or subcutaneously using a delivery pump. The starting dose is 5 mcg per pulse intravenously or 5-25 mcg subcutaneously. The administration on GnRH should be extended throughout the luteal phase, or this should be supplemented with the administration of exogenous hCG (Homburg, 1989).
Monitoring folliculogenesis is simpler than using hMG. Because OHSS does not occur, the patient's response is slow. In most cases, only one follicle is recruited and develops until ovulation. A urinary LH kit is a practical way to monitor these patients. Pelvic ultrasonography can be used once a week until the dominant follicle is detected; once this occurs, ultrasonography can be used more frequently until ovulation occurs. Determination of serum E2 and LH levels can also be performed (Hopkins, 1989).
Treatment of primary amenorrhea
hMG is the treatment of choice for patients with primary amenorrhea due to hypopituitarism. Generally, their response to hMG ovulation induction is too brisk or delayed, and no way exists to predict whether the patient will respond easily. The risk of OHSS and multiple pregnancy is heightened; therefore, hMG should be started at the minimal dose (75 IU SC qd for 7 d). On the seventh day, E2 measurement and ultrasonography are performed. If the E2 level is below 100 pc/mL and the sonogram shows small follicular development, hMG is increased to 150 IU/d for an additional 5 days. However, if the E2 level is greater than 100 pc/mL and the follicles are 10 mm in diameter, hMG should be continued at the same dose. Once the follicular diameter reaches 18 mm and the E2 level is below 2000 pc/mL, ovulation is triggered by the administration of hCG (10,000 IU IM).
The ideal response is one in which only 2-3 follicles develop. If the response is exaggerated, with more than 5 sizable follicles (18 mm in diameter), and the E2 level is greater than 2500 pc/mL, cancelling the ovulation is better in order to avoid the risk of OHSS and a high order of multiple pregnancy. In current practice, an alternative for patients with more than 5 sizable follicles is to convert the treatment to IVF.
Treatment of secondary amenorrhea and oligo-ovulation
Once the diagnosis is established and any other endocrinopathy has been excluded, the ovulation induction agent of choice depends on the estrogen levels of the patient. In hypoestrogenic patients (ie, E2 <20>
PCOD is the most frequent indication for ovulation induction. CC is the drug of choice. Restrict the treatment to 4 ovulatory cycles because 85% of patients conceive by the fourth ovulatory cycle (Garcia, 1977). If pregnancy is not achieved, further evaluation is required to exclude other factors that may be associated with infertility and may interfere with the success of CC therapy. If ovulation does not occur with the 50-mg dose, the CC dose must be increased in subsequent cycles to 100 mg for 5 days. If ovulation still does not occur, modify the CC regimen to 50 mg for 10 days. Although the total CC dose is the same, the follicular stimulation is different. The maximum recommended dosage is 150 mg/d for 5 days. CC failure may indicate either lack of ovulation or lack of pregnancy.
Lack of ovulation
Patients with anovulation who did not ovulate after several cycles of CC at different doses of treatment are deemed clomiphene-resistant. This situation can be related to the presence of other endocrine disorders such as hyperprolactinemia, congenital adrenal hyperplasia, adrenal tumors, Cushing syndrome, thyroid dysfunction, and extreme obesity. Therefore, this problem must be corrected first or concomitantly to obtain an ovulatory response. Although obesity is well known to be associated with insulin resistance, women with PCOD are even more resistant to insulin than other obese patients.
A subgroup of patients has PCOD with hyperinsulinism, hyperandrogenism associated with acanthosis nigricans, and resistance to CC (Stuart, 1986; Dunaif, 1989). This group is amenable to metformin treatment (Nestler, 1998; Kolodziejczyk, 2000). Metformin improves insulin sensitivity and decreases hepatic gluconeogenesis and, therefore, reduces hyperinsulinism, basal and stimulated LH levels, and free testosterone concentration. Consequently, the patient with PCOD becomes responsive to CC ovulation induction.
Adverse effects of metformin include GI intolerance, nausea, vomiting, and abdominal cramps. Weight loss has also been observed. Therefore, patients must build up tolerance. The initial dose is 500 mg PO qd for 7 days, then 500 mg bid for another 7 days, and, finally, 500 mg tid. Start CC at the initial dose of 50 mg/d for 5 days. In many instances, patients can ovulate while on metformin treatment. Therefore, pelvic ultrasonography is required before CC is initiated (Nestler, 1998).
Pure FSH treatment for ovulation induction is another alternative for patients with PCOD who are clomiphene-resistant. Start pure FSH at 37.5 IU/d SC. The dosage is increased by 37.5 IU every 5 days until follicle development is detectable based on an elevation of the E2 levels and the presence of follicle development on sonograms. Using this small amount of FSH, the patient generally develops 1-2 follicles, decreasing the risk for multiple pregnancy and eliminating the risk of OHSS.
Lack of pregnancy
Lack of pregnancy can be related to disruption of the cervical mucus, inadequate follicular development (small follicle [LPD]), presence of LUF syndrome, progesterone deficiency, and premature administration of hCG to trigger ovulation. Fifty percent of patients who ovulate after taking CC have shown LPD diagnosed using endometrial biopsy (Garcia, 1977).
Some authorities believe that ovulation occurs 5 days after the last dose of CC. In the authors' experience, ovulation may occur any time from the 10th to the 23rd day of the menstrual cycle or between the first and the 14th days after the last tablet of CC is administered. Thus, pelvic ultrasonography has become an important tool for monitoring ovulation induction.
The quality of the cervical mucus can be improved with the administration of a small dose of estrogens, or the problem can be bypassed by IUI (Bateman, 1990; Kokia, 1990). If the follicle is smaller than 23-24 mm at the time of ovulation, a better size can be obtained by starting the CC therapy on the second day of the menstrual cycle.
LUF syndrome can be prevented by the administration of hCG (10,000 IU IM) once the follicle reaches 23-24 mm in diameter. Progesterone deficiency can be corrected by the administration of progesterone during the luteal phase, starting 48 hours after ovulation (Garcia, 1977).
Patients with hyperprolactinemia need a thorough evaluation to exclude a pituitary microadenoma. The patient must be treated with bromocriptine (Parlodel) at an initial dose of 2.5 mg PO qd and increased to 5 mg once tolerance is built. If the GI symptoms persist, the medication can be administered intravaginally. Patients with adrenal hyperplasia and other disorders must be treated with prednisone. Patients with hyperthyroidism or hypothyroidism must be treated accordingly (Lisse, 1980; Lobo, 1982). Weight reduction should be part of the treatment because it helps the patient's response to ovulation induction (Shepard, 1979).
Male factors
Asthenospermia associated with varicocele is treated with varicocelectomy or with embolization of the spermatic veins. The initial result of the procedure is not detected before 3 months because spermatogenesis takes 72 days. If no improvement occurs, depending on the amount of functional sperm recovery after the sperm wash, the decision must be made to proceed with either IUI or IVF.
Oligospermia is the most frequent cause of male infertility. Its treatment depends on the etiologic factor, but, in many instances, the underlying cause remains unknown. CI or IUI is the treatment of choice if more than 2 million sperm are recovered after the sperm wash (Nachtigall, 1979; Kerin, 1989) (see Treatment of cervical factors).
Patients whose reproductive tract and FSH, LH, and testosterone levels are determined to be normal can be treated empirically with cycles of CC (25 mg PO qd for 25 d, followed by a 5-d break for at least 6-12 mo). A spermiogram control study is performed after 3 cycles of therapy. Improvement in the sperm count is a good sign, and the treatment should be continued. Checking testosterone levels is advisable because an elevation above the reference range has a negative feedback effect on sperm production. Depending on the sperm count, the couple is advised to have intercourse near the time of ovulation or to proceed with IUI.
Azoospermia treatment depends on the etiology. In patients with obstructive azoospermia and normal gonadotropin levels, sperm can be obtained through microsurgical epididymal sperm aspiration or testicular biopsy (Silber, 1988). Fertilization of the oocytes is performed using IVF/ICSI. In patients with nonobstructive azoospermia, retrograde ejaculation can be the etiologic factor (Brassesco, 1988). The treatment consists of recovering sperm from a urine sample collected immediately after ejaculation.
Alkalinizing the urine before the procedure is necessary. The night before the procedure, the patient must take 2 tablespoons of sodium bicarbonate. The bladder must be emptied 1 hour prior to sperm collection, and a second dose of sodium bicarbonate is taken along with 16 ounces of fluid. A urine sample must be collected immediately after ejaculation. The urine specimen must be centrifuged immediately. The sediment is suspended in a buffer solution, and the recovered sperm is processed using the sperm-wash technique before it can be used for IUI.
Normal infertile couple
The prognosis for the NIC is poor and unpredictable. Only 5-10% of NICs eventually achieve a pregnancy within 5 years. Empirical treatment with COH followed by IUI has improved the pregnancy rate in those patients (Fisch, 1989). If pregnancy does not occur during the first 4 IUI cycles, other alternatives include IVF or any of the associated ART procedures.
Recurrent pregnancy loss
RPL or habitual abortion is defined as 3 or more consecutive spontaneous miscarriages. They occur before the 20th week of pregnancy or when the fetus weighs less than 500 g. In the past, the obstetrical outcome after 3 spontaneous miscarriages was very grim, and the chances of these patients having a live birth were only 15-25%. This prognosis has improved over the years. Currently, the chance of another pregnancy loss after 3 consecutive miscarriages is estimated to be 30-45%. The chances of having a live birth are 55-60%; however, if a previous live birth has occurred, the rate increases to up to 70% (Daya, 1996).
Although the incidence of spontaneous miscarriage is very high in healthy persons, the great majority of pregnancy losses are unrecognized because they may occur before the onset of the menstrual period. Others are classified as menstrual irregularities, and only those that occur after a positive pregnancy test result or those that are identified after the trophoblastic tissue has been passed are defined as miscarriages; the rate is 12% (Knudsen, 1991).
Several causes are associated with or responsible for pregnancy loss, including anatomical defects, infections, environmental factors, endocrine disorders, genetics, immunologic problems, and thrombophilia.
Anatomical factors
Congenital abnormalities of the müllerian ducts are associated with miscarriages, particularly septate uterus and bicornuate uterus with a blind horn. Patients who were exposed to DES in utero and were born with a T-shaped uterus and patients with cervical incompetence have a greater risk of RPL (Tidey, 1991). The treatment for septate uterus (see Hysteroscopy) is the removal of the septum through hysteroscopy. Patients with a blind uterine horn must have it removed, and patients with an incompetent cervix require cerclage during the first trimester of pregnancy.
Intrauterine synechiae are associated with RPL through unknown mechanisms, although the compromise of the endometrial vascularization produced by the synechiae could be the reason for poor embryo implantation and subsequent miscarriage (Schnorr, 1991).
Infectious factors
The role of infectious diseases in the etiology of RPL is not clear (Kimball, 1979). Toxoplasmosis is not responsible for miscarriage during acute episodes. Nevertheless, spontaneous abortion has been described in pregnant patients with seroconversion of Toxoplasma gondii antibody. Chronic toxoplasmosis is not associated with habitual abortion.
Listeria monocytogenes is responsible for recurrent abortions in cattle and is an established cause of human pregnancy loss during the second and third trimesters. The pregnancy loss results from the hematogenous spread of enterically acquired infection from ingestion of unpasteurized dairy products. Listerial colonization of the genital tract is unlikely, and multiple studies have failed to demonstrate a chronic infection (Rabau, 1963).
U urealyticum plays a role in miscarriages in animals, but, in humans, its role is controversial. According to many reports, U urealyticum has been cultured from the endocervix and from products of conception (Knudsen, 1967). Patients with positive culture findings appear to have some fever after the miscarriage. Drawing any conclusion is difficult based on the fact that U urealyticum can be isolated from 35-75% of the lower genital tracts of patients who are sexually active (Stray-Pedersen, 1978). Further, few studies have checked the incidence of U urealyticum infection after spontaneous full-term pregnancy deliveries. Despite of the lack of solid information, customary practice is to treat patients prophylactically with doxycycline (100 mg bid for 10 d).
Cytomegalovirus (CMV) and group B streptococci are not associated with RPL. Although CMV has been isolated from placentas and fetuses, most of the pathology is associated with premature rupture of membranes, preterm delivery, and neonatal infection.
Environmental factors
Coffee consumption has been reported to be associated with a low fertility rate and increased risk of pregnancy loss. Other published reports cannot support this hypothesis (Srisuphan, 1986; Mills, 1993). Patients who are heavy smokers (>10 cigarettes per d) have an increased incidence of pregnancy loss, premature deliveries, and babies of low birth weight. Also, anesthetic gases and exposure to tetrachloroethylene (used in dry cleaning) have been implicated in early pregnancy loss.
Microwave radiation, video terminals, hot tubs, waterbeds, and electric blankets do not cause pregnancy loss.
Endocrine factors
Subclinical thyroid dysfunction or mild diabetes is not associated with pregnancy loss. Patients with PCOD have an increase in first-trimester miscarriages. Chronic LH elevation has been suggested as the etiology of these losses; nevertheless, suppression of LH secretion with a GnRH agonist prior to ovulation induction shows no improvement in the miscarriage rate. The authors' experience with patients with PCOD undergoing IVF has failed to show an increase in the miscarriage rate. LPD is most likely responsible for these losses. Garcia et al reported that LPD was present in 50% of patients with PCOD who conceived after CC ovulation induction and subsequently miscarried (Garcia, 1977); however, when these patients were supplemented with progesterone in subsequent pregnancies, the miscarriage rate was similar to that of the general population.
Genetic factors
Chromosome abnormalities are the most frequent causes of spontaneous miscarriage. The incidence increases with age, but, in general, the fetal chromosomal abnormalities vary. RPL occurs in 3-8% of couples in whom one of the partners carries a balance translocation (De Braekeleer, 1990). Nothing can be done to improve or avoid this problem, except to reassure the patients that they have a good chance of having a healthy child. In the worst-case scenario, the child could have the same balance translocation.
The incidence of an abnormal karyotype is slight in the general population; therefore, a complete evaluation for RPL must be performed before checking the couple's chromosomes (Simpson, 1981).
Autoimmune factors
Patients with a history of autoimmune diseases such as lupus have an increased incidence of miscarriages. When tested, these patients have positive results for lupus anticoagulant and anticardiolipin antibodies (Out, 1992). Lupus anticoagulant antibodies are not exclusive to lupus erythematosus. The antiphospholipid antibodies are directed against platelets and the vascular endothelium, causing thrombosis. These antibodies block prostacyclin formation, which results in unbalanced thromboxane activity and vasoconstriction before the thrombosis. This thrombosis may be the cause of placental insufficiency and, therefore, pregnancy loss.
Patients with RPL must be screened for activated partial thromboplastin time, lupus anticoagulant, and anticardiolipin antibody (Thiagarajan, 1980). If results are positive, the treatment is low-dose aspirin (80 mg PO qd) and low-dose heparin once the pregnancy is established (Cowchock, 1992; Pollard, 1992). Some patients develop toxemia. The future of patients who have no symptoms of lupus but who are positive for antiphospholipid antibodies is not clear; thus, they must be informed about the results and the uncertainty of the prognosis.
Assisted Reproductive Technologies
The first successful human IVF attempt resulted in the 1978 delivery of Louise Brown in England and is considered the beginning of a new era for the treatment of infertility (Steptoe, 1978).
Since 1981, the understanding of the ovulatory process has been revolutionized, hopelessly infertile patients have seen their dreams fulfilled, and success has shown that patients who were considered sterile in the past are now capable of having children.
IVF indications have departed from the narrow scope of tubal infertility to other indications that were almost impossible to overcome, including infertility related to oligospermia and obstructive azoospermia.
In vitro fertilization
IVF consists of retrieving a preovulatory oocyte from the ovary and fertilizing it with sperm in the laboratory, with subsequent embryo transfer (replacement) within the endometrial cavity. Biologists and veterinarians have used IVF for several decades in the laboratory for applications such as animal husbandry and cattle breeding. Lack of understanding of human embryo development and special metabolic needs accounted for the delay in achieving success. The pioneering work of Edwards and Steptoe has been duplicated worldwide, and IVF is now recognized as an established treatment for infertility.
Indications
Absence of the fallopian tubes and severe pelvic adhesions were the absolute indications for IVF, but they have been broadened. Patients with a history of endometriosis unsuccessfully treated medically or surgically can undergo IVF. Patients with some malformation of the uterus related to DES exposure during pregnancy are candidates. Patients with husbands who have severe oligospermia or a history of obstructive azoospermia are also candidates for IVF. Finally, patients with an unknown etiology of infertility (ie, NICs) may undergo IVF.
Procedure
IVF consists of retrieving a preovulatory oocyte from the ovary and fertilizing it with sperm in the laboratory, with subsequent embryo transfer (replacement) within the endometrial cavity. The following steps are required during an IVF cycle:
- Ovarian stimulation
- Follicular aspiration
- Oocyte classification
- Sperm preparation
- Oocyte insemination
- Embryo culture
- Embryo transfer
Ovarian stimulation for IVF
Although the world's first IVF pregnancy, which occurred in England, followed the fertilization of a single oocyte obtained during a spontaneous menstrual cycle, IVF success is related to the patient's age and the number of embryos transferred into the endometrial cavity, among other factors (Garcia, 1981; Romeu, 1987; Murdock, 1988). The number of embryos obtained in any given IVF cycle depends on the number of oocytes obtained after ovulation induction, follicular aspiration, and fertilization.
Several protocols are available for ovarian stimulation, and all are based on the principles of follicular recruitment, selection, and dominance.
- Clomiphene citrate
- Today, CC is used infrequently in IVF. A clomiphene-only protocol consists of 5-7 days of treatment, with doses of 50-150 mg starting on the second menstrual cycle day (Marrs, 1984). The ovarian response is monitored using pelvic ultrasonography and determinations of serum E2 and LH levels. When the follicle diameter reaches 18 mm, LH must be measured every 3-4 hours to detect a premature LH surge that generally occurs when the follicle reaches 23-24 mm in diameter (Punnonen, 1988). Oocyte retrieval must be performed within 24-26 hours after the LH surge.
- The advantages of the CC protocol include low cost and an almost nonexistent risk of OHSS.
- The major disadvantages include (1) low oocyte yield (1-2 per cycle), (2) frequent LH surges that lead to oocyte retrieval at any time of the day, (3) high cancellation rate (25-50%), and (4) low pregnancy rate.
- Clomiphene citrate and human menopausal gonadotropins
- This combination has the advantage of increasing the number of recruited follicles. The doses of CC are similar, but the hMG (150 IU qd) is administered for a period of 2-7 days after the CC (Diamond, 1986). Frequent pelvic ultrasonography and daily determinations of E2 levels are required, as are frequent determinations of LH once the follicular diameter reaches 15 mm. Remember that the follicle is smaller with this protocol. The follicle completes its development at 17-18 mm; therefore, hCGs (10,000 IU IM) must be administered at this time to complete the oocyte maturation. Oocyte aspiration should be performed 35 hours after the hCG injection.
- The advantage of the CC/hMG protocol is an increase in the number of recruited follicles. The disadvantages of the protocol are premature luteinization, spontaneous LH surge (20-50%), and high cancellation rate (15-50%).
- Human menopausal gonadotropins
- The administration of hMG for ovarian stimulation in a healthy anovulatory patient has been one of the groundbreaking discoveries in IVF (Garcia, Fertil Steril, 1983, 167-73; Garcia, Fertil Steril, 1983, 174-9). The use of hMG has evolved with the introduction of new technologies and the generation of pure FSH gonadotropins that can be used subcutaneously (Edelstein, 1990). Furthermore, recombinant FSH and LH gonadotropins are currently available (Weatherbee, 1999). hMGs are as follows:
- Human menopausal gonadotropin (Repronex) - FSH of 75 IU and LH of 75 IU
- Pure FSH (Bravelle) - FSH of 75 IU and LH of less than 1 IU
- Recombinant FSH (Gonadal F, Follistim) - FSH of 75 IU and LH of 0 IU
- Recombinant LH (in clinical trials) - FSH of 0 IU and LH of 75 IU
- The doses of gonadotropins vary from 150-450 IU/d, depending on the patient's age and history of previous ovulatory response. In general, patients are started on the second or third menstrual cycle day. The response is monitored using daily serum E2 determination and later using pelvic ultrasonography (Garcia, Fertil Steril, 1983, 174-9).
- Once most of the follicles reach 17-18 mm in diameter, the gonadotropins are discontinued. hCG (10,000 IU) is administered that evening, and oocyte retrieval is performed 35 hours later. With the use of gonadotropins, ovulation induction appears to be more predictable. However, a spontaneous LH surge occurs in approximately 25% of cycles, which is one of the reasons for cancellation.
- Regardless of the use of gonadotropins, a group of patients, known as poor responders, develops only 2-3 follicles, and their oocytes are of poor quality (Schoolcraft, 1997). The possibility of a successful pregnancy in this group of patients is seriously compromised. In general, these patient responses are related to poor ovarian reserve (POR) (Scott, 1989).
- Patient response, which is defined by an elevated baseline FSH level (>10 mIU/mL), a low E2 level (<20>100 pg) associated with early follicle recruitment, characterize POR. Some patients respond with premature luteinization due to elevated tonic LH levels and subsequent premature elevation of progesterone that compromise oocyte quality. Analysis of each ovarian stimulation cycle is important to modify future stimulations and to increase the patient's response.
- The administration of hMG for ovarian stimulation in a healthy anovulatory patient has been one of the groundbreaking discoveries in IVF (Garcia, Fertil Steril, 1983, 167-73; Garcia, Fertil Steril, 1983, 174-9). The use of hMG has evolved with the introduction of new technologies and the generation of pure FSH gonadotropins that can be used subcutaneously (Edelstein, 1990). Furthermore, recombinant FSH and LH gonadotropins are currently available (Weatherbee, 1999). hMGs are as follows:
- GnRHa agonists
- Initially, GnRHa agonists were used in IVF patients who had a history of premature luteinization, spontaneous LH surge, and an ovarian stimulation response that was less than acceptable or ideal (Meldrum, 1989). The GnRHa agonists can be used in 2 protocols known as the flare-up protocol and the luteal-phase protocol (Garcia, 1990; Padilla, 1991; Hughes, 1992; Marci, 2001).
- The flare-up protocol has the advantage of using the transitory elevation of FSH (agonist effect) that occurs during the first 4 days of the follicular phase. This elevation helps in the follicular recruitment process; after 5 days of GnRHa administration, the pituitary gland undergoes down-regulation, which prevents premature luteinization and the spontaneous LH surge. The administration of gonadotropins must be initiated on the fifth menstrual cycle day (Garcia, 1990).
- The modified flare-up protocol is the administration of birth control pills for 4-21 days during the preceding IVF menstrual cycle. A microdose of GnRHa agonist leuprolide acetate is administered twice daily subcutaneously starting on day 1 of the menstrual cycle, and the human menopause gonadotropin (recombinant FSH) begins on day 4 of the menstrual cycle.
- In the luteal-phase protocol, GnRHa is started on the 17th or 21st menstrual cycle day. By the onset of the menstrual cycle, the phenomenon of pituitary down-regulation is in effect, so the administration of the gonadotropins begins on the second day of bleeding (Hughes, 1992; Marci, 2001).
- The adjunctive use of GnRHa in ovarian stimulation leads to an overall improvement in IVF. This accounts for the increase in the number, quality, and synchronization of the oocytes recovered per cycle and improves the fertilization rate, the number of embryos, and the pregnancy rate (Garcia, 1990). Furthermore, because more embryos are available than the number used in fresh embryo transfer, an opportunity exists to cryopreserve the excessive number of embryos for future embryo transfer(s).
- Potential risks and disadvantages with the use of GnRHa include (1) increased requirements of gonadotropins, (2) increased costs due to additional days of therapy, (3) the risk of OHSS due to excessively high E2 levels, and (4) an increased rate of multiple pregnancies.
- Initially, GnRHa agonists were used in IVF patients who had a history of premature luteinization, spontaneous LH surge, and an ovarian stimulation response that was less than acceptable or ideal (Meldrum, 1989). The GnRHa agonists can be used in 2 protocols known as the flare-up protocol and the luteal-phase protocol (Garcia, 1990; Padilla, 1991; Hughes, 1992; Marci, 2001).
- GnRHa antagonists
- The GnRHa antagonists are the latest generation of GnRHa that block LH secretion without a flare-up effect (North American Ganirelix Study Group, 1999). The GnRHa antagonists (eg, cetrorelix [Cetrotide], ganirelix [Antagon]) are administered (1) as a single dose on the eighth menstrual cycle day; (2) in small amounts over 4 days, starting on the seventh menstrual cycle day; (3) when the largest follicle reaches a diameter of 14 mm; or (5) when the LH levels in serum are greater than 10 mIU/mL (Olivennes, 1995).
- GnRHa antagonists have the advantage of blocking the LH surge at the periovulatory period; therefore, premature luteinization or spontaneous LH surge does not occur. Because the pituitary gland is not down-regulated at the beginning of the menstrual cycle, smaller amounts of gonadotropins are required to stimulate ovulation. Another advantage with this protocol is the prevention of OHSS, especially in patients with elevated E2 levels (>3000 pg/mL) or more than 15 follicles during the stimulation. Because the half-life of the GnRHa antagonist is short, it is possible to elicit the preovulatory LH surge by the administration of leuprolide acetate and avoid the long-term effects of the hCG injection that are responsible for triggering the ovarian hyperstimulation.
Follicular aspiration
Oocyte aspiration is undertaken 35-36 hours after hCG administration. Originally, all aspirations were performed using laparoscopy (Jones, 1982). This requires general anesthesia, a skillful laparoscopist, major discomfort for the patient, and the risks involved with the laparoscopy. Oocyte aspiration in patients with severe pelvic adhesions is extremely difficult with most patients and virtually impossible with others (see Image 85).
Follicular aspirations under ultrasonographic guidance were first performed transabdominally, then transurethrally, and, finally, transvaginally (Lenz, 1981). The transvaginal route became the preferred procedure for most IVF programs. The procedure consists of placing the patient in the dorsal lithotomy position for the transvaginal oocyte aspiration. The bladder is catheterized, and the vaginal wall is prepared with povidone iodine. A 5- to 7-MHz ultrasonographic probe is inserted in the vagina to localize the ovaries and the follicles. A 17-gauge needle is subsequently passed through the vaginal fornix into the ovaries. Image 86 illustrates how the follicular fluid is aspirated. The fluid is delivered to the IVF laboratory, which, ideally, is located adjacent to the operating room.
General anesthesia is not required for ultrasonographic-guided oocyte aspiration. Heavy sedation is suitable for this procedure. The risk of bowel injury is almost zero and allows for oocyte retrieval in patients with a frozen pelvis in whom laparoscopic retrieval is not possible. Furthermore, it avoids the use of carbon dioxide pneumoperitoneum. The major risks are infection and damage to the pelvic vessel, but they should not be a problem if the procedure is carefully performed (Howe, 1988).
Oocyte classification
The classification of the oocyte is a crucial step for success with IVF (Elder, 1992). The follicular fluid is scanned under either a dissecting microscope or an inverted microscope. The oocytes are graded according to the appearance of the corona-cumulus complex. The presence of a polar body (metaphase II stage) and/or germinal vesicle (prophase stage) is a determining factor for the preincubation time prior to the insemination. Other oocytes are degenerated (atretic and fractured zona). The last category constitutes fewer than 15% of the total oocytes obtained (see Images 87-91).
Sperm preparation and oocyte insemination
A semen sample is obtained after a 3- to 5-day period of sexual abstinence immediately prior to the oocyte retrieval. For IUI, IVF, and ICSI procedures, the removal of certain components of the ejaculate (ie, seminal fluid, excess cellular debris, leukocytes, morphologically abnormal sperm) with the retention of the motile fraction of sperm is desirable.
For most specimens, the greatest recovery of the motile portion results from separation via centrifugation through a discontinuous density gradient system. However, for certain very poor specimens with low original concentrations of motile sperm, the use of the gradient system results in such a negligible recovery that it is rendered useless. The recourse for these specimens is to simply remove the seminal fluid through successive media washes.
Furthermore, a small number of specimens have acceptable original concentrations of motile sperm but poor recoveries with the gradient system. These specimens benefit most by layering a washed pellet of sperm with nutrient media and allowing the motile fraction to swim up into the media before being separated (Kerin, 1989).
The sperm are incubated for 60 minutes in an atmosphere of 5% carbon dioxide in air. Finally, the supernatant containing the motile fraction of sperm is removed. Sperm concentration and motility are determined. A final concentration of 50,000 motile sperm per mL of insemination media is added to the oocytes.
Embryo culture
The inseminated oocytes are incubated in an atmosphere of 5% carbon dioxide in air with 98% humidity. Ideally, the presence of 2 pronuclei and the extrusion of a second polar body are the criteria required to ascertain fertilization, which should occur approximately 18 hours after insemination (see Image 92). The presence of 3 or more pronuclei can be related to polyspermia or to the retention of the second polar body. This occurs in 9% of all fertilized oocytes (see Image 93). Mechanical dispersion of the cumulus is required when the oocytes are heavily covered by the cumulus; with this, the number of pronuclei can be visualized.
The fertilized oocytes (embryos) are transferred into growth media and placed in the incubator. No further evaluation is performed over the next 24 hours. A 4- to 8-cell stage preembryo is observed approximately 36-48 hours after insemination. A 10- to 16-cell embryo is observed after 48-72 hours. The morula or blastocyst stage is observed after 96-120 hours. Although the goal is to observe the embryos as described, pregnancy has been achieved from embryos with slow or delayed cleavage. Embryos are classified according to symmetry, presence of fragments, clarity, and number of blastomeres (see Images 94-100) (Elder, 1992).
Embryo transfer
Transcervical transfer under transabdominal ultrasonography is the most common method used for embryo transfer. The procedure is usually performed within 42-72 hours after oocyte insemination. However, pregnancies have been reported after transfer performed at the 2-pronuclei stage. Lately, a tendency to delay the transfer until the embryo reaches the blastocyst stage has been noted (Testart, 1986; Scott, 1998; Schoolcraft, 1999; Shapiro, 2000).
Several catheters have been designed for embryo transfer (see Image 101). All are equally suitable for embryo transfer, and their use is a matter of physician preference rather than a specific catheter being synonymous with a greater pregnancy rate. The embryos should be loaded with 30 µL of culture media. The catheter is advanced up to the fundus of the endometrial cavity, then withdrawn slightly. The embryos are ejected with a small amount of air (see Image 102). Subsequent to the embryo transfer, the patient must be on bedrest for 30-60 minutes.
Management of the luteal phase
Progesterone supplementation during the luteal phase is started 72 hours after oocyte retrieval (Garcia, 1981). The exogenous progesterone is administered because of concerns that superovulation and the aspiration of granulosa cells at the time of oocyte retrieval may induce an abnormal endocrine milieu (Soliman, 1994). Recent publications support the benefits of supporting the luteal phase with exogenous progesterone. Several preparations are available; for example, natural progesterone in oil base is administered intramuscularly and vaginal progesterone suppositories and capsules of micronized progesterone are used intravaginally or sublingually (Fanchin, 2001).
Normally, progesterone supplementation is continued for approximately 2 weeks. If the pregnancy test result is positive, progesterone is continued until the eighth week of pregnancy.
IVF-related procedures
The following ART procedures have been used as alternatives to IVF:
- Gamete intrafallopian transfer
- Zygote intrafallopian transfer
- Assisted fertilization techniques
- Partial zona dissection
- Subzonal sperm injection
- Intracytoplasmic sperm injection
- Assisted hatching
- Embryo cryopreservation
Gamete intrafallopian transfer
In 1985, Asch et al described the gamete intrafallopian transfer (GIFT) procedure. The procedure consists of ovarian stimulation, monitored follicular development, and oocyte aspiration similar to IVF. It differs in that the patient must have at least 1 normal-appearing and patent fallopian tube.
On the day of oocyte retrieval, the male provides a semen sample 2 hours before the procedure in order to perform the sperm wash and capacitation. Once the oocytes have been classified, a laparoscopy or a minilaparotomy is performed. The oocytes, along with 150,000 sperm, are loaded into a special catheter in the laboratory under a microscope. The loaded catheter is brought to the operating room, where the laparoscopist is waiting. The injection of the gametes into the fallopian tube requires that the fimbria be gently picked up with an atraumatic grasping forceps. The ostium has already been identified, so in a minimal amount of time, the tip of the catheter is passed through the ostium and advanced up to the ampulla of the fallopian tube(s), where the gametes are released. Fertilization should occur inside the fallopian tube.
It appears that this procedure is more physiologic. Nevertheless, a major drawback of the GIFT procedure is that it does not allow for visual confirmation of fertilization because it occurs inside the body. Also, if pregnancy does not occur, no method exists to determine whether the cause was lack of fertilization or lack of implantation. Additionally, General anesthesia is required. Finally, with the GIFT procedure, a laparoscopy or minilaparotomy is required, which implies additional expense.
Zygote intrafallopian transfer
The zygote intrafallopian transfer (ZIFT) procedure is a combination of IVF and GIFT (Devroey, 1986). Fertilization occurs in the IVF laboratory. However, the preembryo is transferred into the fallopian tube via laparoscopy at the 2-pronuclei stage or 24 hours after oocyte retrieval. One variant of the ZIFT procedure is the transfer of the cleaved preembryos at 48 hours (tubal embryo transfer) (Balmaceda, 1988). This procedure requires that the patient be taken to the operating room twice, which represents additional expense and doubles the risk for complications.
Assisted fertilization techniques
Major problems in IVF arise when not enough sperm (<1>
Assisted fertilization techniques include partial zona dissection (PZD), subzonal sperm injection (SUZI), ICSI, and assisted hatching (AH).
PZD consists of creating a small opening at the zona pellucida level either mechanically or using low-pH solutions (Tyrode) to digest a small portion of the zona (Cohen and Malter, 1988). The intent is for the sperm to take advantage of this weak spot to make contact with the oocyte membrane so that fertilization will occur. The rate of fertilization per oocyte is approximately 20-25%, the incidence rate of polyspermia is elevated (50%), and the pregnancy rate is only 5% per cycle. To succeed with PZD requires the recovery of approximately 500,000 sperm after the sperm wash.
The SUZI procedure is indicated in patients for whom conventional IVF or PZD does not work (Laws-King, 1987). The procedure consists of suspending the oocyte in a sucrose medium. By osmosis, the oocyte is dehydrated; therefore, the perivitelline space is enlarged. Next, 3-5 spermatozoa are injected into the perivitelline space using a microneedle. Fertilization occurs in approximately 15-20% of the oocytes, the pregnancy rate is less than 3%, and the incidence of polyspermia is 50%.
PZD and SUZI are obsolete and have been replaced by the ICSI procedure. Palermo et al developed the ICSI procedure in Belgium (1992). ICSI has revolutionized the treatment of severe male factor infertility because only a single live sperm is required per oocyte. The sperm can be obtained through masturbation, epididymal aspiration, testicular biopsy, or needle puncture of the testes (Silber, 1988). ICSI gives the opportunity to males with a history of obstructive azoospermia (perhaps due to congenital absence of the vas deferens) to have a biological child.
The sperm is paralyzed by stroking the distal portion of its tail. The oocyte is stripped from the cumulus using a solution of hyaluronidase. To inject the sperm, first the oocyte is stabilized with a micropipette, then, the sperm is loaded, tail first, into a microneedle. The oocyte membrane is pierced with the microneedle, and the oolemma is entered. The spermatozoon is released inside the oolemma, and the microinjected oocyte is kept in the incubator (see Images 103-104).
The fertilization rate varies from 50-75% per oocyte. The current fertilization rate is 95% per patient, and the pregnancy rate is comparable to that of IVF. The risks involved with the procedure are few and include oocyte rupture and damage and/or retention of the second polar body with the presence of 3 pronuclei (Blake, 2000). Currently, an increased risk of congenital malformation and chromosome abnormalities exists with ICSI. However, a few reports claim that no significant difference exists between IVF and ICSI outcome (Cohen, 1992).
Embryo hatching is an obligatory step in the process of embryo implantation. Cohen et al reported that some of the IVF embryos demonstrated a rather thick zona pellucida; this zona thickness may represent an obstacle for the normal embryo hatching, therefore interfering with the implantation. They suggested the use of selective AH for embryos that show a thick zona pellucida. The procedure is performed a couple of hours before the embryo transfer. AH can be performed mechanically, creating a microrent at the zona level, or by chemical digestion of the zona using a Tyrode solution (low pH). Either of the procedures creates a weak spot within the zona, facilitating the break of the zona and the hatching of the embryo. AH is recommended for patients undergoing IVF who are older than 38 years, patients with multiple ART failures, and in all cryopreserved embryos.
Embryo cryopreservation
The use of GnRHa-COH protocols to prevent the LH surge not only decreased the number of canceled cycles but also increased the number of follicles and their synchronization; therefore, more embryos were available than the ideal number to be transferred. Furthermore, the incidence of multiple pregnancy increased, especially the incidence of triplets, quadruplets, and so on. Embryo cryopreservation became an important part of ART to prevent multiple pregnancies, to prevent maternal and fetal complications, and to decrease the cost of ART because patients have the opportunity to achieve a pregnancy from the same IVF cycle (Marrs, 1987; Trounson, 1988; Veeck, 1993).
Embryo cryopreservation can be performed at the 2-pronuclei stage, on cleaved embryos, and at the blastocyst stage (Cohen and DeVane, 1988; Hartshorne, 1991). The embryos are stored in liquid nitrogen for a period previously agreed upon by the IVF program policy and the patient. Most of the IVF programs store the embryos for a period of 3-5 years, and their subsequent disposition should be outlined in the consent form, to include (1) embryo donation to another couple, (2) donating the embryos for research, or (3) disposition of the embryo after thawing.
Frozen embryo transferThe transfer of cryopreserved embryos can be performed during a natural menstrual cycle or during an artificial endometrium-stimulated cycle. No differences in the pregnancy rate occur, provided that the endometrium, during a natural menstrual cycle, demonstrates a 10-mm thick triple-line pattern. The embryos are thawed 24, 48, or 96 hours after ovulation, depending on the stage at which cryopreservation was performed. The thawed embryos are incubated 24 hours before embryo transfer.
The artificially stimulated endometrium requires detection of ovulation during a previous menstrual cycle and down-regulation of the pituitary gland with GnRHa starting on the 21st menstrual cycle day and is followed by increasing doses of estrogen starting with the onset of the menstrual bleeding (Navot, 1986). Estrogen is administered for 2 weeks. At that time, pelvic ultrasonography is performed to determine endometrial thickness. If the above-mentioned criteria are fulfilled, progesterone is added to the estrogen for 48 hours, then the embryos are thawed, cultured, and transferred as previously described. The administration of estrogen and progesterone is continued until the day of the pregnancy test. If the pregnancy test result is positive, the hormonal support must be continued until the 10th week of pregnancy.
Results
A normal term pregnancy is the ultimate goal of IVF; thus, the pregnancy rate is the best indicator for evaluation of a program. Its efficiency can also be determined based on the following parameters:
- The number of patients who canceled because of deficient response to stimulation or spontaneous LH surge
- The number and quality of oocytes obtained at aspiration
- The number of oocytes that fertilized and cleaved properly
- The number of cryopreserved embryos
- The number of embryos transferred
The success of a program is also established by the patient population enrolled in the program, the indication(s) for the procedure, and the patient's age at the time of ovarian stimulation. Some IVF programs enroll only patients with good prognoses in order to maintain a higher pregnancy rate.
In 2002, 115,392 ART procedures were performed in the United States (Centers for Disease Control and Prevention, 2003) as follows:
- In vitro fertilization - 99.1%
- Gamete intrafallopian transfer - 0.2%
- Zygote intrafallopian transfer - 0.5%
- Cryopreserved embryos - 14%
- Donor oocyte - 11%
The primary diagnosis or indications for ART were as follows:
- Tubal factor - 13.6%
- Male factor - 18.8%
- Endometriosis - 6.7%
- Ovulatory dysfunction - 6.0%
- Diminished ovarian reserve - 5.7%
- Unknown factor - 11.1%
- Other factors - 5.6%
- Multiple factors, female only - 12.7%
- Multiple factors, male and female - 18.5%
Success rates were as follows:
- Overall pregnancy rate per initiated cycle - 34.3%
- Live birth rate per initiated cycle - 28.3%
- Live births per oocyte retrieval - 32.6%
- Live births per embryo transfer - 34.8%
No significant differences in the pregnancy rate were noted among IVF (34.0%), GIFT (25.1%), and ZIFT (26.3%). The incidence of miscarriage (spontaneous abortion, 15.0%), stillbirths (0.5%), congenital malformation, or chromosome abnormality is similar to that of the general population. Ectopic pregnancy has been reported after IVF due to migration of the embryo through the cornual ostium. Ectopic pregnancy occurs in approximately 0.7% of cases. In some instances, ectopic pregnancy is associated with heterotopic pregnancy.
The pregnancy rate by patient age was consistent at approximately 45.2% among women younger than 35 years and declined to 37.7% for the group aged 35 to 37 years. Women aged 38-40 years enjoyed a pregnancy rate of 23.5%. The success rate was approximately 23.0% in those older than 40 years. Pregnancy after age 44 years is the exception.
The live birth rate by primary diagnosis was similar in all groups, as follows:
- Male factors - 33.6%
- Endometriosis - 32.4%
- Ovarian factors - 13.9%
- Tubal factors - 30.5%
- Uterine factors -22.9%
- Other causes - 25.7%
- Unexplained - 30.0%
- Ovulatory dysfunction - 33.1%
- Multiple factors, female only - 23.4%
- Multiple factors, female and male - 26.4%
The percentage of ART births that were multiple births was 35.4% (twins, 31.6%; triplets and quadruplets, approximately 3.8%). This compares with a multiple live birth rate of 3% in the general US population.
The pregnancy rate is also related to the number of embryos transferred, as follows:
Live Birth Rates
Open table in new window
Table
| Number of Embryos | Percentage of Cycles | Live Birth Rate, % | Multiple Births Rate, % |
|---|---|---|---|
| 1 | 12.8% | 98.0% | 2.0%* |
| 2 | 39.5% | 66.7% | 32.6% |
| 3 | 37.7% | 61.7% | 32.6% |
| 4 | 32.8% | 62.3% | 32.1% |
| 5+ | 28.9% | 63.2% | 31.3% |
| Number of Embryos | Percentage of Cycles | Live Birth Rate, % | Multiple Births Rate, % |
|---|---|---|---|
| 1 | 12.8% | 98.0% | 2.0%* |
| 2 | 39.5% | 66.7% | 32.6% |
| 3 | 37.7% | 61.7% | 32.6% |
| 4 | 32.8% | 62.3% | 32.1% |
| 5+ | 28.9% | 63.2% | 31.3% |
*Identical twins
The pregnancy rate from frozen preembryos is 20.9%, which is comparable to the 28.8% rate achieved with a fresh embryo transfer.
The live birth rate with ICSI (31.9%) is nearly identical to the live birth rate with conventional IVF, which is 34.0%. Nevertheless, keep in mind that patients who underwent ICSI had almost no chance of succeeding with a pregnancy using conventional IVF or obsolete techniques such as PZD and SUZI.
Donor oocyte
Patients with POR have a rather low chance of overcoming infertility; yet, some of them, along with patients with premature menopause and patients with physiologic menopause, are interested in having a child. The only alternative for these patients is adoption or an oocyte donation (Buster, 1983; Rosenwaks, 1986). Donor oocyte is the counterpart of donor sperm. The source of the oocyte can be anonymous or known (ie, younger relative, designated donor). Ideally, the donor should be aged 21-30 years, although the age can extend to 35 years.
The donor undergoes ovulation induction according to the standard IVF protocol. Meanwhile, the recipient takes increasing doses of estrogens to synchronize her endometrium in preparation for a fresh embryo transfer. This technique is similar to the one described under Frozen embryo transfer (Navot, 1986).
Because oocyte cryopreservation is still in a preliminary stage of development, only fresh oocytes without quarantine are used. However, the donor must be screened for numerous transmissible diseases (eg, HIV, syphilis, CMV, hepatitis, gonorrhea, chlamydia), and a complete physical and gynecological evaluation is performed. The donor patient also undergoes a psychological evaluation and consultation with a genetic counselor. The recipient patient also has a psychological evaluation. The oocyte recipient and her partner are required to have the same kinds of screening tests as the oocyte donor. The legal aspects of the procedure and future offspring must be discussed. A thorough consent form must be signed by all parties involved (American Fertility Society, 1993).
The success of donor oocyte programs surpasses the success of conventional IVF. The risk of multiple pregnancy is very high; thus, the conservative approach of transferring only 2 embryos at the blastocyst stage is highly recommended to the recipient couple.
Donor embryo
Donor embryo is the earliest form of adoption. As stated in the embryo cryopreservation consent form, the couple must sign an advance directive regarding embryo ownership and disposition. Those directives should include statements regarding (1) embryo donation to another couple, (2) donation of the embryos for research, or (3) disposition of the embryos after thawing.
Donor embryo is one option the patient can choose; therefore, those embryos can be donated according to the IVF program policy. Embryo donation programs must follow the regulations established for tissue donors, which require that all screening tests must be performed before embryo cryopreservation, with the tests being repeated at least 6 months later. The same screening tests are required for the recipient couple (Lutjen, 1984).
The donor embryo transfer protocol is identical to the protocol described for cryopreserved embryo transfer (Navot, 1986). The embryos must not be sold as a commodity. This concept has been endorsed by the Society of Assisted Reproductive Technology and the ASRM (American Society for Reproductive Medicine, 2002).
Gestational carriers
Patients who were unable to have a biological child because of absence of the uterus (congenital or acquired) or patients in whom a pregnancy is contraindicated are now able to have a biological child by the use of a gestational carrier or surrogate mother. The gestational carrier program must be designed under the strictest policies because of the medicolegal implications.
The selection or approval of the gestational carrier should be based on the premise that the pregnancy does not imply a risk for the carrier and that all the consent forms have been signed once the carrier and the genetic parents have completed the psychological evaluations and screening tests. The endometrium of the gestational carrier is artificially stimulated through the administration of estrogen and progesterone as described under Frozen embryo transfer (Navot, 1986). A national consensus does not exist regarding the birth certificate for children born under the surrogate mother program. Some states have no requirement to mention the surrogate; in others, a complete adoption process is required.
Multimedia
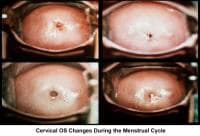 | Media file 1: Infertility. Cervical os changes during the menstrual cycle. Image courtesy of Jairo E. Garcia, MD. |
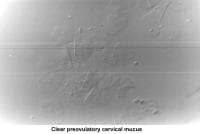 | Media file 2: Infertility. Clear preovulatory cervical mucus. Image courtesy of Jairo E. Garcia, MD. |
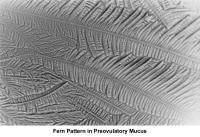 | Media file 3: Infertility. Fern pattern of preovulatory mucus. Image courtesy of Jairo E. Garcia, MD. |
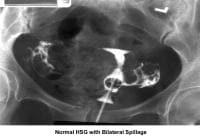 | Media file 4: Infertility. Hysterosalpingogram image demonstrating normal findings with bilateral spillage. Image courtesy of Jairo E. Garcia, MD. |
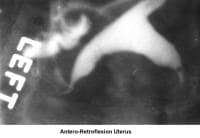 | Media file 5: Infertility. Antero-retroflexion uterus. Image courtesy of Jairo E. Garcia, MD. |
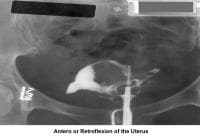 | Media file 6: Infertility. Anteroflexion or retroflexion of the uterus. Image courtesy of Jairo E. Garcia, MD. |
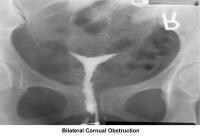 | Media file 7: Infertility. Bilateral cornual obstruction. Image courtesy of Jairo E. Garcia, MD. |
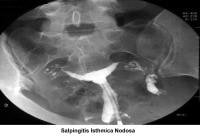 | Media file 8: Infertility. Salpingitis isthmica nodosa. Image courtesy of Jairo E. Garcia, MD. |
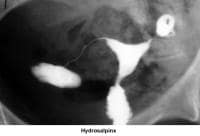 | Media file 9: Infertility. Hydrosalpinx. Image courtesy of Jairo E. Garcia, MD. |
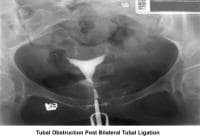 | Media file 10: Infertility. Tubal obstruction post–bilateral tubal ligation. Image courtesy of Jairo E. Garcia, MD. |
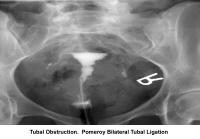 | Media file 11: Infertility. Tubal obstruction: Pomeroy bilateral tubal ligation. Image courtesy of Jairo E. Garcia, MD. |
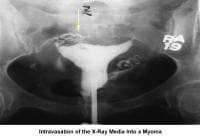 | Media file 12: Infertility. Intravasation of the contrast medium due to myoma. Image courtesy of Jairo E. Garcia, MD. |
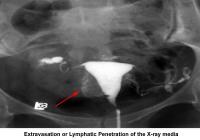 | Media file 13: Infertility. Extravasation or lymphatic penetration of the contrast medium. Image courtesy of Jairo E. Garcia, MD. |
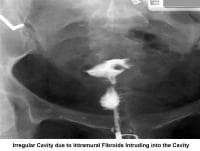 | Media file 14: Infertility. Irregular cavity due to intramural fibroids intruding into the cavity. Image courtesy of Jairo E. Garcia, MD. |
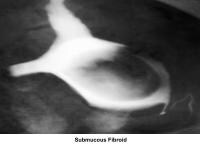 | Media file 15: Infertility. Submucous fibroid. Image courtesy of Jairo E. Garcia, MD. |
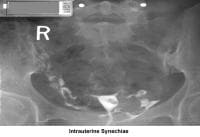 | Media file 16: Infertility. Intrauterine synechiae. Image courtesy of Jairo E. Garcia, MD. |
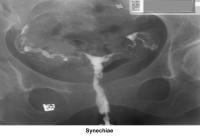 | Media file 17: Infertility. Synechiae. Image courtesy of Jairo E. Garcia, MD. |
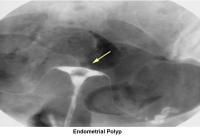 | Media file 18: Infertility. Endometrial polyp. Image courtesy of Jairo E. Garcia, MD. |
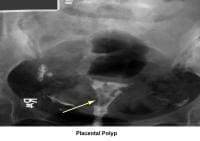 | Media file 19: Infertility. Placental polyp. Image courtesy of Jairo E. Garcia, MD. |
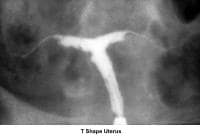 | Media file 20: Infertility. T-shaped uterus. Image courtesy of Jairo E. Garcia, MD. |
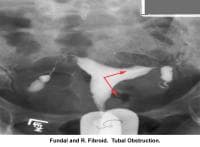 | Media file 21: Infertility. Fundal and right fibroid tubal obstruction. Image courtesy of Jairo E. Garcia, MD. |
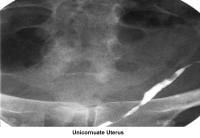 | Media file 22: Infertility. Unicornuate uterus. Image courtesy of Jairo E. Garcia, MD. |
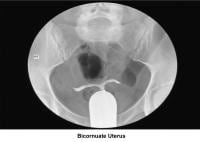 | Media file 23: Infertility. Bicornuate uterus. Image courtesy of Jairo E. Garcia, MD. |
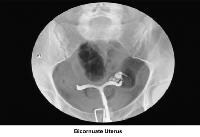 | Media file 24: Infertility. Bicornuate uterus. Image courtesy of Jairo E. Garcia, MD. |
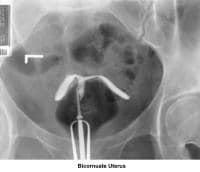 | Media file 25: Infertility. Bicornuate uterus. Image courtesy of Jairo E. Garcia, MD. |
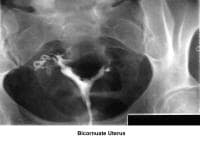 | Media file 26: Infertility. Bicornuate uterus. Image courtesy of Jairo E. Garcia, MD. |
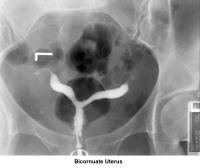 | Media file 27: Infertility. Bicornuate uterus. Image courtesy of Jairo E. Garcia, MD. |
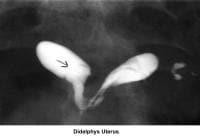 | Media file 28: Infertility. Didelphys uterus. Image courtesy of Jairo E. Garcia, MD. |
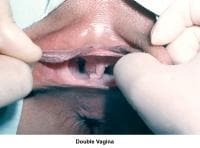 | Media file 29: Infertility. Double vagina. Image courtesy of Jairo E. Garcia, MD. |
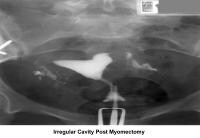 | Media file 30: Infertility. Irregular endometrial cavity after myomectomy. Image courtesy of Jairo E. Garcia, MD. |
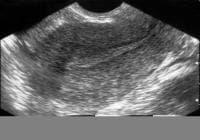 | Media file 31: Infertility. Sonogram: Sagittal view of the uterus. Image courtesy of Jairo E. Garcia, MD. |
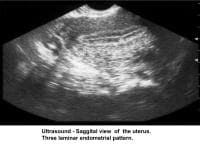 | Media file 32: Infertility. Sonogram: Sagittal view of the uterus. Three-laminar endometrial pattern. Image courtesy of Jairo E. Garcia, MD. |
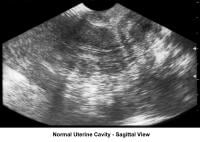 | Media file 33: Infertility. Sonogram: Sagittal view of normal uterine cavity. Image courtesy of Jairo E. Garcia, MD. |
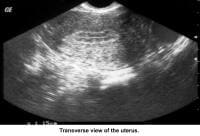 | Media file 34: Infertility. Sonogram: Transverse view of the uterus. Image courtesy of Jairo E. Garcia, MD. |
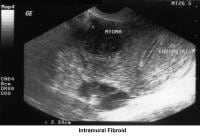 | Media file 35: Infertility. Intramural fibroid. Image courtesy of Jairo E. Garcia, MD. |
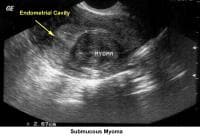 | Media file 36: Infertility. Submucous myoma. Image courtesy of Jairo E. Garcia, MD. |
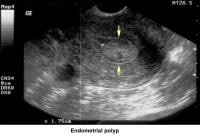 | Media file 37: Infertility. Endometrial polyp. Image courtesy of Jairo E. Garcia, MD. |
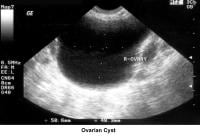 | Media file 38: Infertility. Ovarian cyst. Image courtesy of Jairo E. Garcia, MD. |
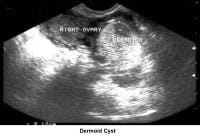 | Media file 39: Infertility. Dermoid cyst. Image courtesy of Jairo E. Garcia, MD. |
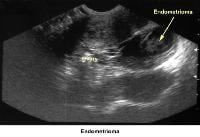 | Media file 40: Infertility. Endometrioma. Image courtesy of Jairo E. Garcia, MD. |
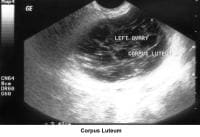 | Media file 41: Infertility. Corpus luteum. Image courtesy of Jairo E. Garcia, MD. |
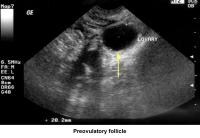 | Media file 42: Infertility. Preovulatory follicle. Image courtesy of Jairo E. Garcia, MD. |
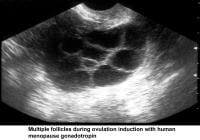 | Media file 43: Infertility. Multiple follicles during ovulation induction with human menopause gonadotropin. Image courtesy of Jairo E. Garcia, MD. |
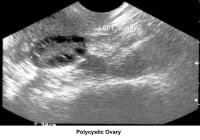 | Media file 44: Infertility. Polycystic ovary. Image courtesy of Jairo E. Garcia, MD. |
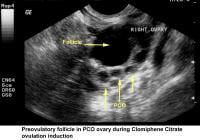 | Media file 45: Infertility. Preovulatory follicle in polycystic ovary during clomiphene citrate ovulation induction. Image courtesy of Jairo E. Garcia, MD. |
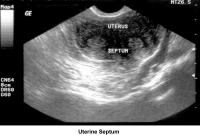 | Media file 46: Infertility. Uterine septum. Image courtesy of Jairo E. Garcia, MD. |
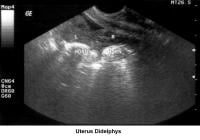 | Media file 47: Infertility. Uterus didelphys. Image courtesy of Jairo E. Garcia, MD. |
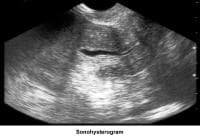 | Media file 48: Infertility. Sonohysterogram. Image courtesy of Jairo E. Garcia, MD. |
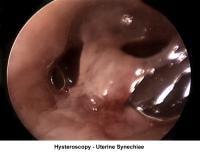 | Media file 49: Infertility. Hysteroscopy - Uterine synechiae. Image courtesy of Jairo E. Garcia, MD. |
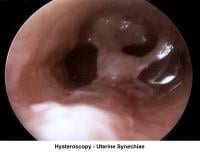 | Media file 50: Infertility. Hysteroscopy - Uterine synechiae. Image courtesy of Jairo E. Garcia, MD. |
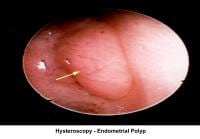 | Media file 51: Infertility. Hysteroscopy - Endometrial polyp. Image courtesy of Jairo E. Garcia, MD. |
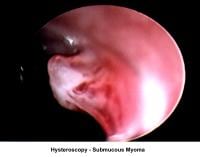 | Media file 52: Infertility. Hysteroscopy - Submucous fibroid. Image courtesy of Jairo E. Garcia, MD. |
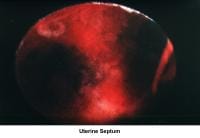 | Media file 53: Infertility. Uterine septum. Image courtesy of Jairo E. Garcia, MD. |
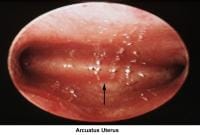 | Media file 54: Infertility. Arcuate uterus. Image courtesy of Jairo E. Garcia, MD. |
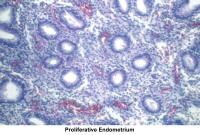 | Media file 55: Infertility. Proliferative endometrium. Image courtesy of Jairo E. Garcia, MD. |
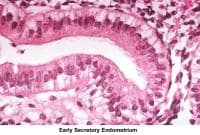 | Media file 56: Infertility. Early secretory endometrium. Image courtesy of Jairo E. Garcia, MD. |
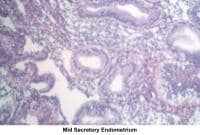 | Media file 57: Infertility. Mid secretory endometrium. Image courtesy of Jairo E. Garcia, MD. |
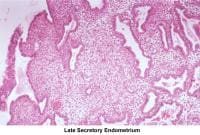 | Media file 58: Infertility. Late secretory endometrium. Image courtesy of Jairo E. Garcia, MD. |
 | Media file 59: Infertility. Basal body temperature chart. Image courtesy of Jairo E. Garcia, MD. |
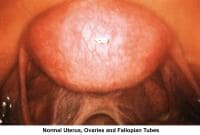 | Media file 60: Infertility. Normal uterus, ovaries, and fallopian tubes. Image courtesy of Jairo E. Garcia, MD. |
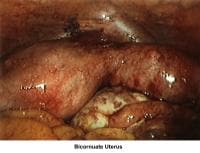 | Media file 61: Infertility. Bicornuate uterus. Image courtesy of Jairo E. Garcia, MD. |
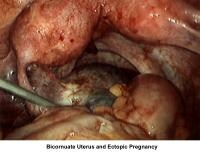 | Media file 62: Infertility. Bicornuate uterus and ectopic pregnancy. Image courtesy of Jairo E. Garcia, MD. |
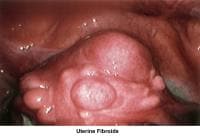 | Media file 63: Infertility. Uterine fibroids. Image courtesy of Jairo E. Garcia, MD. |
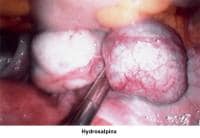 | Media file 64: Infertility. Hydrosalpinx. Image courtesy of Jairo E. Garcia, MD. |
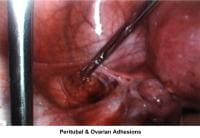 | Media file 65: Infertility. Peritubal and ovarian adhesions. Image courtesy of Jairo E. Garcia, MD. |
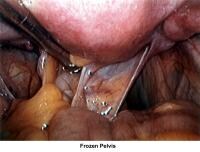 | Media file 66: Infertility. Frozen pelvis. Image courtesy of Jairo E. Garcia, MD. |
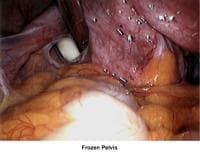 | Media file 67: Infertility. Frozen pelvis. Image courtesy of Jairo E. Garcia, MD. |
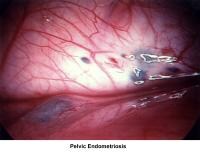 | Media file 68: Infertility. Pelvic endometriosis. Image courtesy of Jairo E. Garcia, MD. |
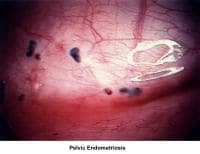 | Media file 69: Infertility. Pelvic endometriosis. Image courtesy of Jairo E. Garcia, MD. |
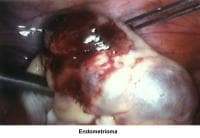 | Media file 70: Infertility. Endometrioma. Image courtesy of Jairo E. Garcia, MD. |
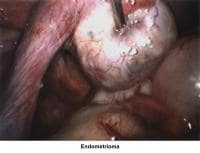 | Media file 71: Infertility. Endometrioma. Image courtesy of Jairo E. Garcia, MD. |
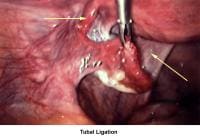 | Media file 72: Infertility. Tubal ligation. Image courtesy of Jairo E. Garcia, MD. |
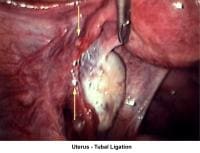 | Media file 73: Infertility. Uterus - Tubal ligation. Image courtesy of Jairo E. Garcia, MD. |
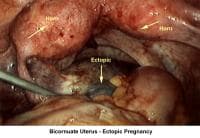 | Media file 74: Infertility. Bicornuate uterus - Ectopic pregnancy. Image courtesy of Jairo E. Garcia, MD. |
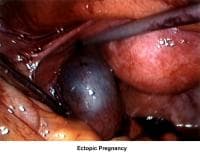 | Media file 75: Infertility. Ectopic pregnancy. Image courtesy of Jairo E. Garcia, MD. |
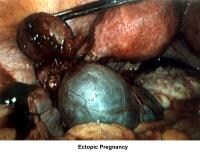 | Media file 76: Infertility. Ectopic pregnancy. Image courtesy of Jairo E. Garcia, MD. |
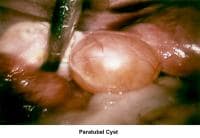 | Media file 77: Infertility. Paratubal cyst. Image courtesy of Jairo E. Garcia, MD. |
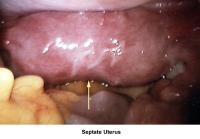 | Media file 78: Infertility. Septate uterus. Image courtesy of Jairo E. Garcia, MD. |
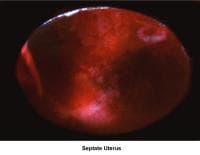 | Media file 79: Infertility. Septate uterus. Image courtesy of Jairo E. Garcia, MD. |
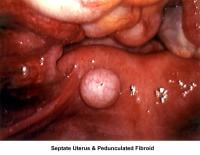 | Media file 80: Infertility. Septate uterus and pedunculated fibroid. Image courtesy of Jairo E. Garcia, MD. |
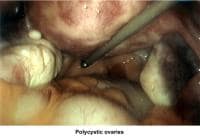 | Media file 81: Infertility. Polycystic ovaries. Image courtesy of Jairo E. Garcia, MD. |
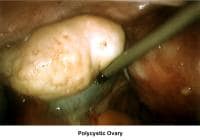 | Media file 82: Infertility. Polycystic ovary. Image courtesy of Jairo E. Garcia, MD. |
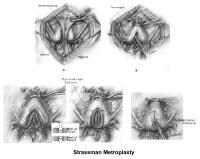 | Media file 83: Infertility. Metroplasty - Strassman technique. Image courtesy of Clifford R. Wheeless, Jr, MD. |
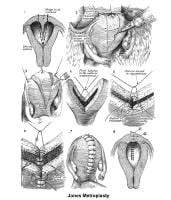 | Media file 84: Infertility. Metroplasty - Jones technique. Image courtesy of Clifford R. Wheeless, Jr, MD. |
 | Media file 85: Infertility. Diagram of oocyte aspiration by laparoscopy. |
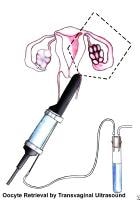 | Media file 86: Infertility. Diagram of oocyte aspiration by transvaginal ultrasonography. |
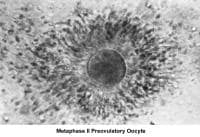 | Media file 87: Infertility. Metaphase II preovulatory oocyte. Image courtesy of Jairo E. Garcia, MD. |
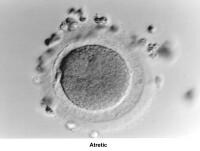 | Media file 88: Infertility. Atretic. Image courtesy of Jairo E. Garcia, MD. |
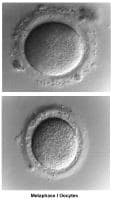 | Media file 89: Infertility. Metaphase I oocytes. Image courtesy of Jairo E. Garcia, MD. |
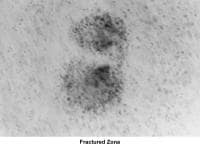 | Media file 90: Infertility. Fractured zona. Image courtesy of Jairo E. Garcia, MD. |
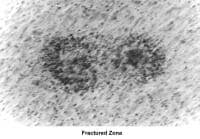 | Media file 91: Infertility. Fractured zona. Image courtesy of Jairo E. Garcia, MD. |
 | Media file 92: Infertility. Two-pronuclei stage embryo - Eighteen hours postinsemination. Image courtesy of Jairo E. Garcia, MD. |
 | Media file 93: Infertility. Polyspermia or second polar body. Image courtesy of Jairo E. Garcia, MD. |
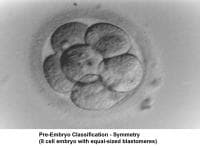 | Media file 94: Infertility. Preembryo classification - Symmetry (8-cell embryo with equal-sized blastomeres). Image courtesy of Jairo E. Garcia, MD. |
 | Media file 95: Infertility. Fragments. Image courtesy of Jairo E. Garcia, MD. |
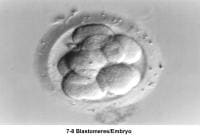 | Media file 96: Infertility. Blastomeres/embryo. Image courtesy of Jairo E. Garcia, MD. |
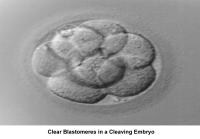 | Media file 97: Infertility. Clear blastomeres in a cleaving embryo. Image courtesy of Jairo E. Garcia, MD. |
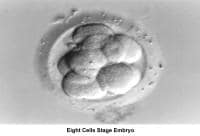 | Media file 98: Infertility. Embryo (8-cell stage). Image courtesy of Jairo E. Garcia, MD. |
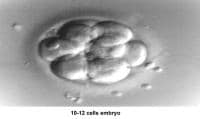 | Media file 99: Infertility. Embryo (10- to 12-cell stage). Image courtesy of Jairo E. Garcia, MD. |
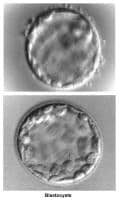 | Media file 100: Infertility. Blastocysts. Image courtesy of Jairo E. Garcia, MD. |
 | Media file 101: Infertility. Catheters for embryo transfer. Image courtesy of Jairo E. Garcia, MD. |
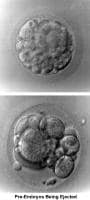 | Media file 102: Infertility. Preembryos being ejected. Image courtesy of Jairo E. Garcia, MD. |
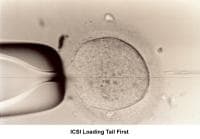 | Media file 103: Infertility. Intracytoplasmic sperm injection loading tail first. Image courtesy of Jairo E. Garcia, MD. |
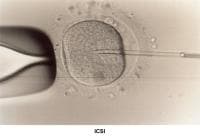 | Media file 104: Infertility. Intracytoplasmic sperm injection. Image courtesy of Jairo E. Garcia, MD. |
 | Media file 105: Infertility. Husband-and-wife medical history packet. |
Keywords
infertility, lack of pregnancy, fertility, in vitro fertilization, IVF, intracytoplasmic sperm injection, ICSI, assisted reproductive technologies, ART, pelvic inflammatory disease, PID, endometriosis, sexually transmitted diseases, gonorrhea, chlamydia, STDs, cryopreservation, sperm donor, sperm morphology, sperm concentration, sperm quality, semen analysis, sperm washing, sperm evaluation, egg evaluation, egg quality, oocyte quality, egg donor, oocyte donor, gamete donation, oocyte donation, sperm donation, conception problems, pregnancy problems, pregnancy complications, pregnancy, normal infertile couple, NIC
endometriosis, Rokitansky-Küster-Hauser syndrome, double vagina, absent vagina, absent uterus, uterine malformation, DES, diethylstilbestrol, sexual history, intrauterine fibroid, submucous fibroid, placental polyps, T-shaped uterus, tubal ligation, premature ovarian failure, luteal phase deficiency, luteal-phase deficiency, luteinized unruptured follicle syndrome, LUF syndrome, recurrent pregnancy loss, recurrent abortion, gamete intrafallopian transfer, gamete intra-fallopian transfer, GIFT, zygote intrafallopian transfer, zygote intra-fallopian transfer, ZIFT, partial zona dissection, PZD, subzonal sperm injection, SUZI, assisted hatching, AH, gestational carrier, surrogate carrier.
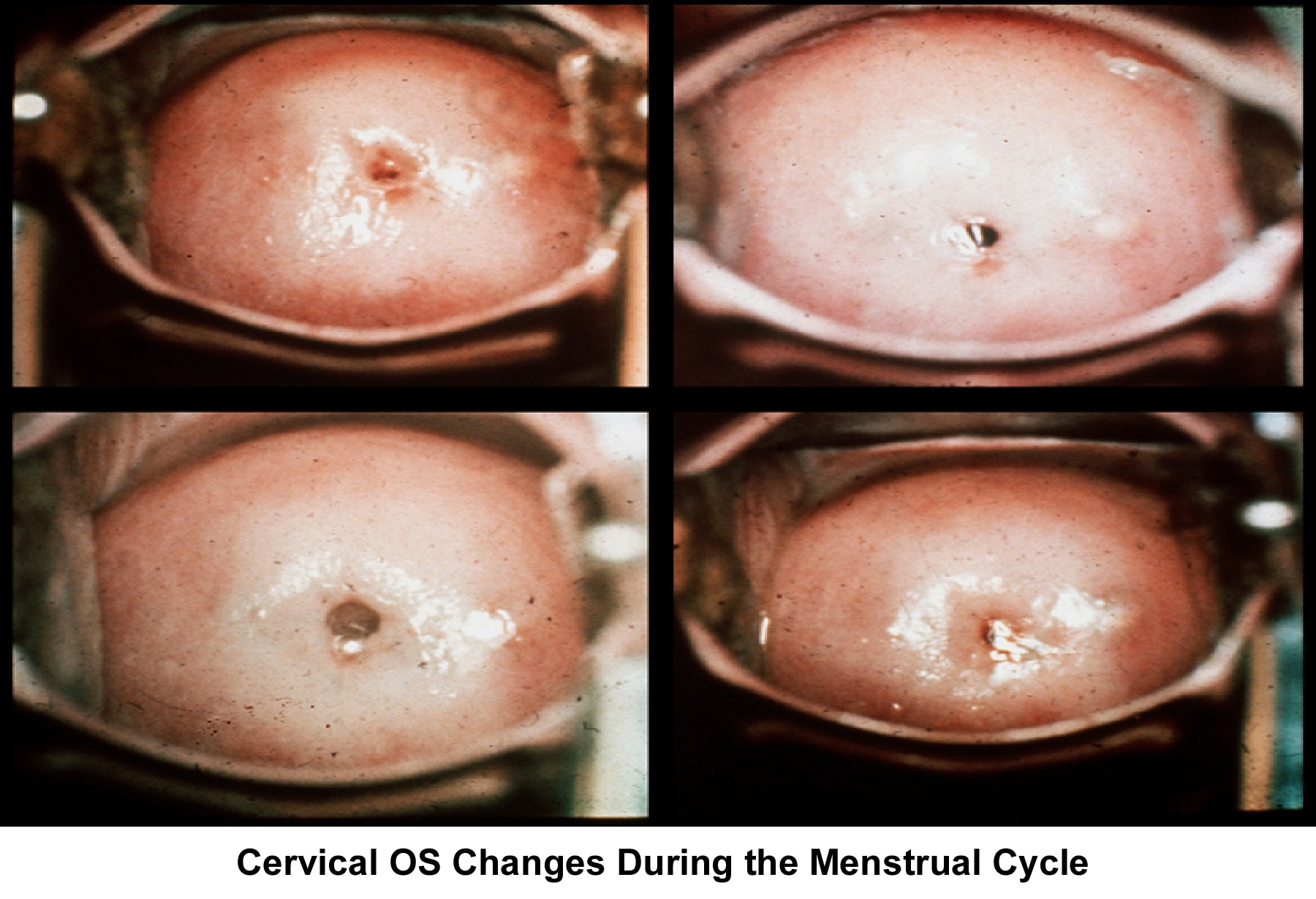
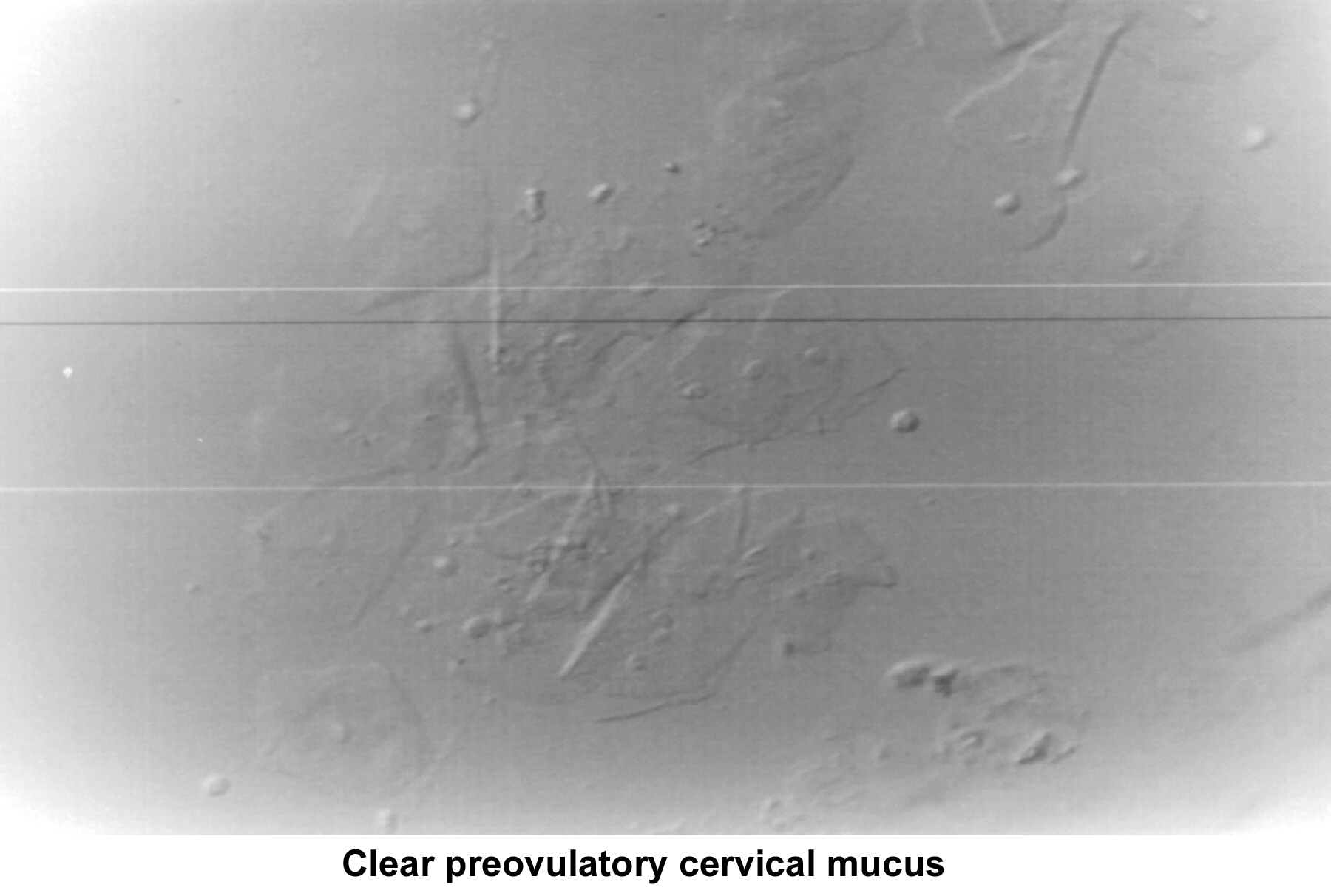
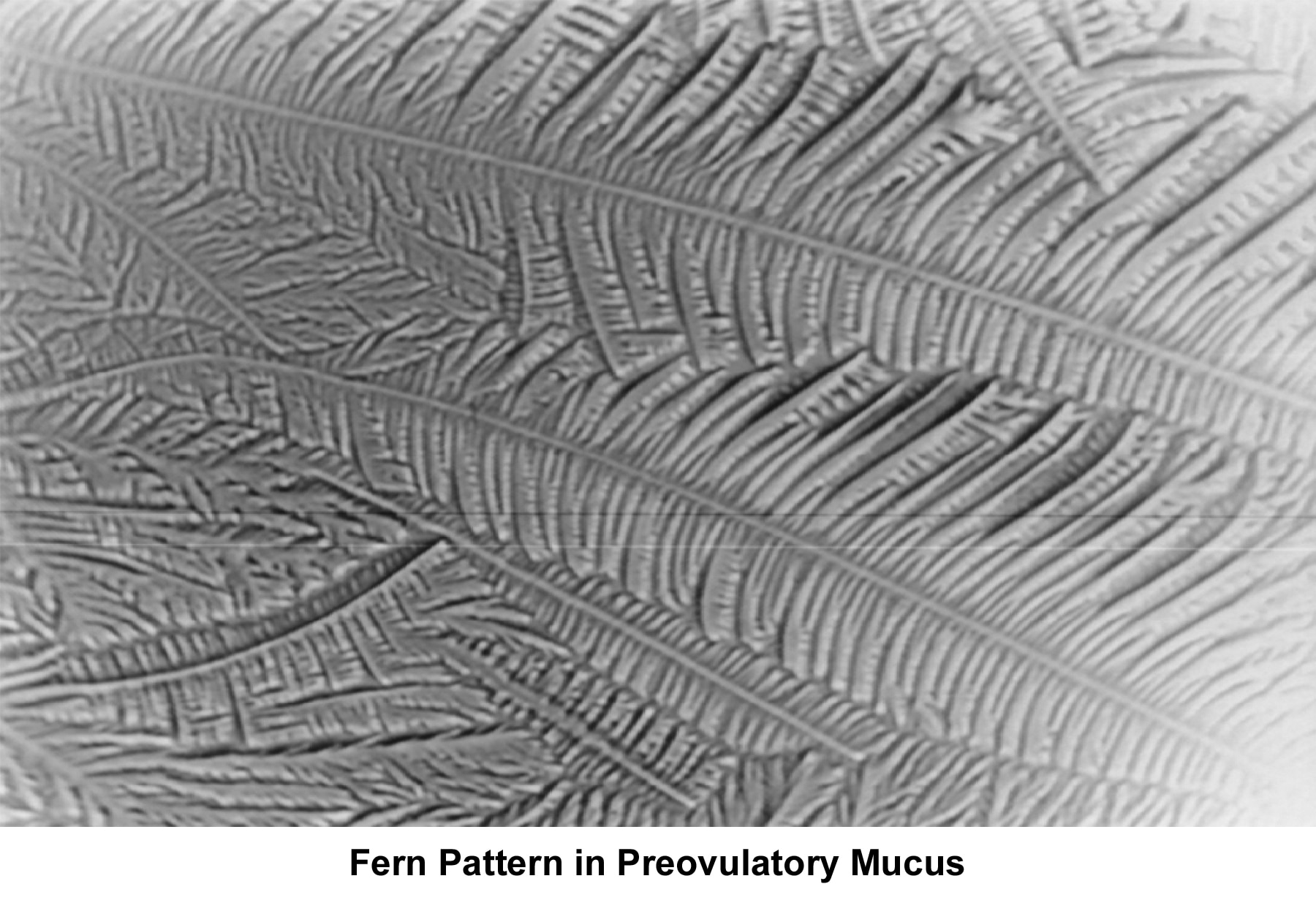
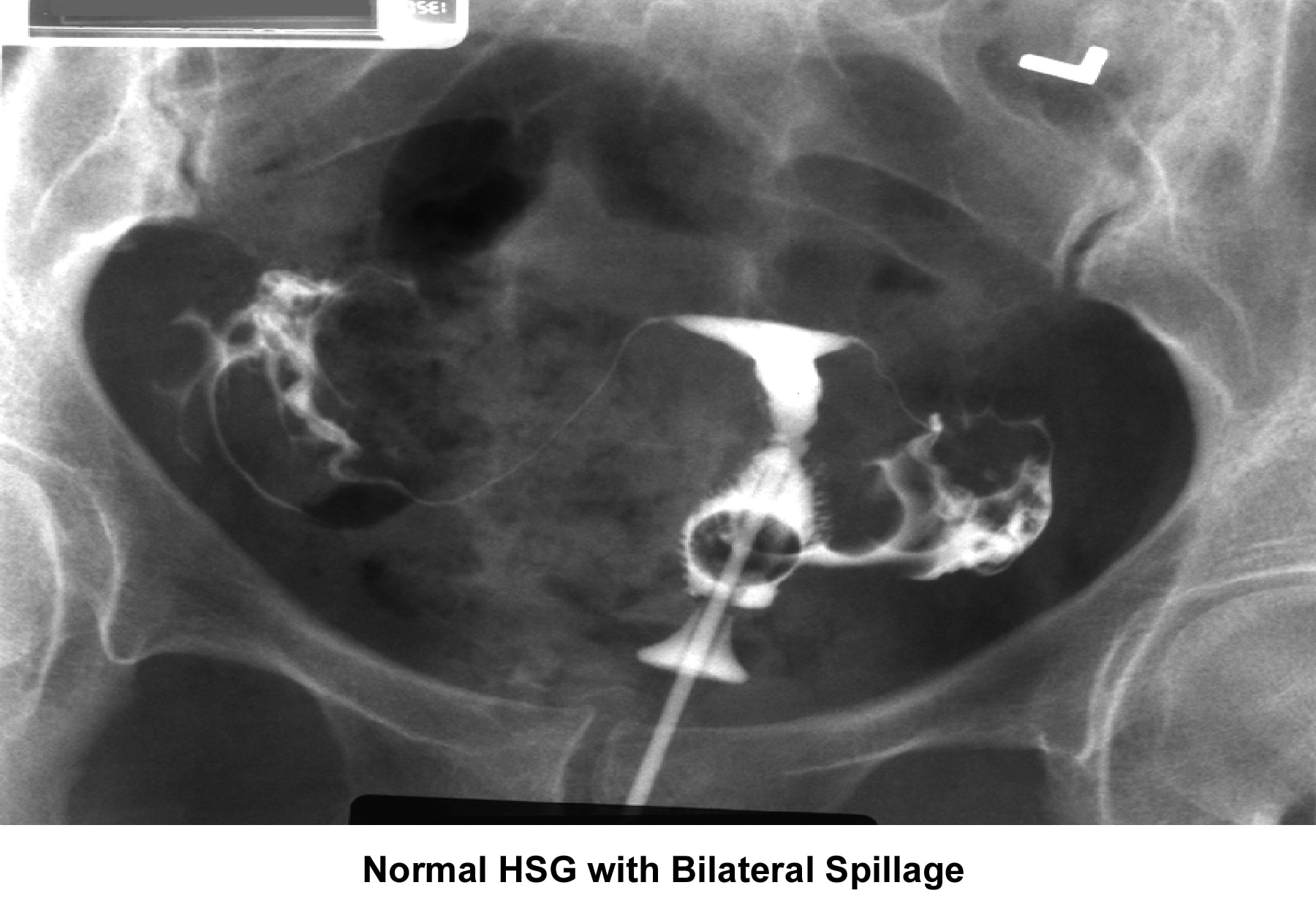
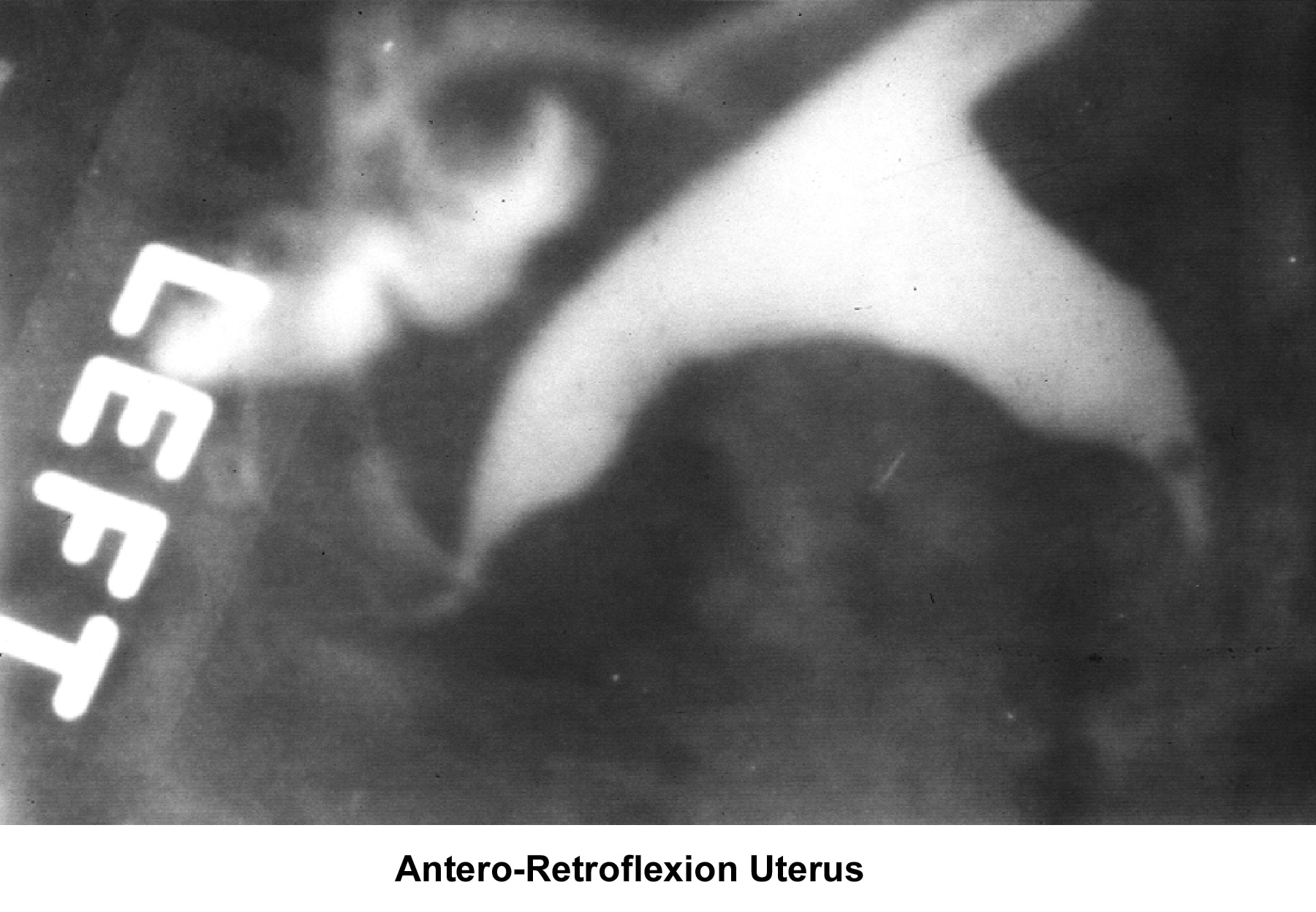
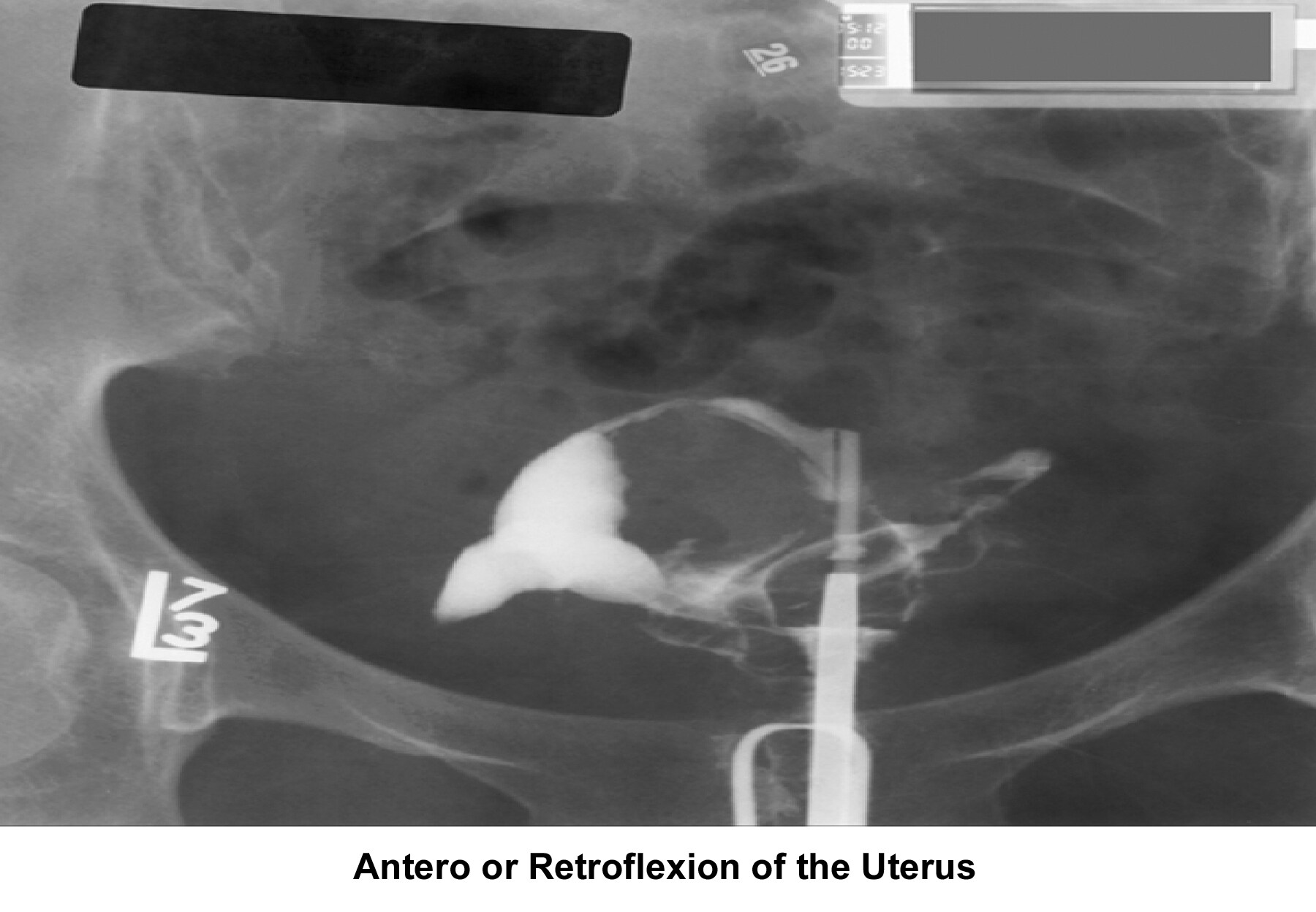
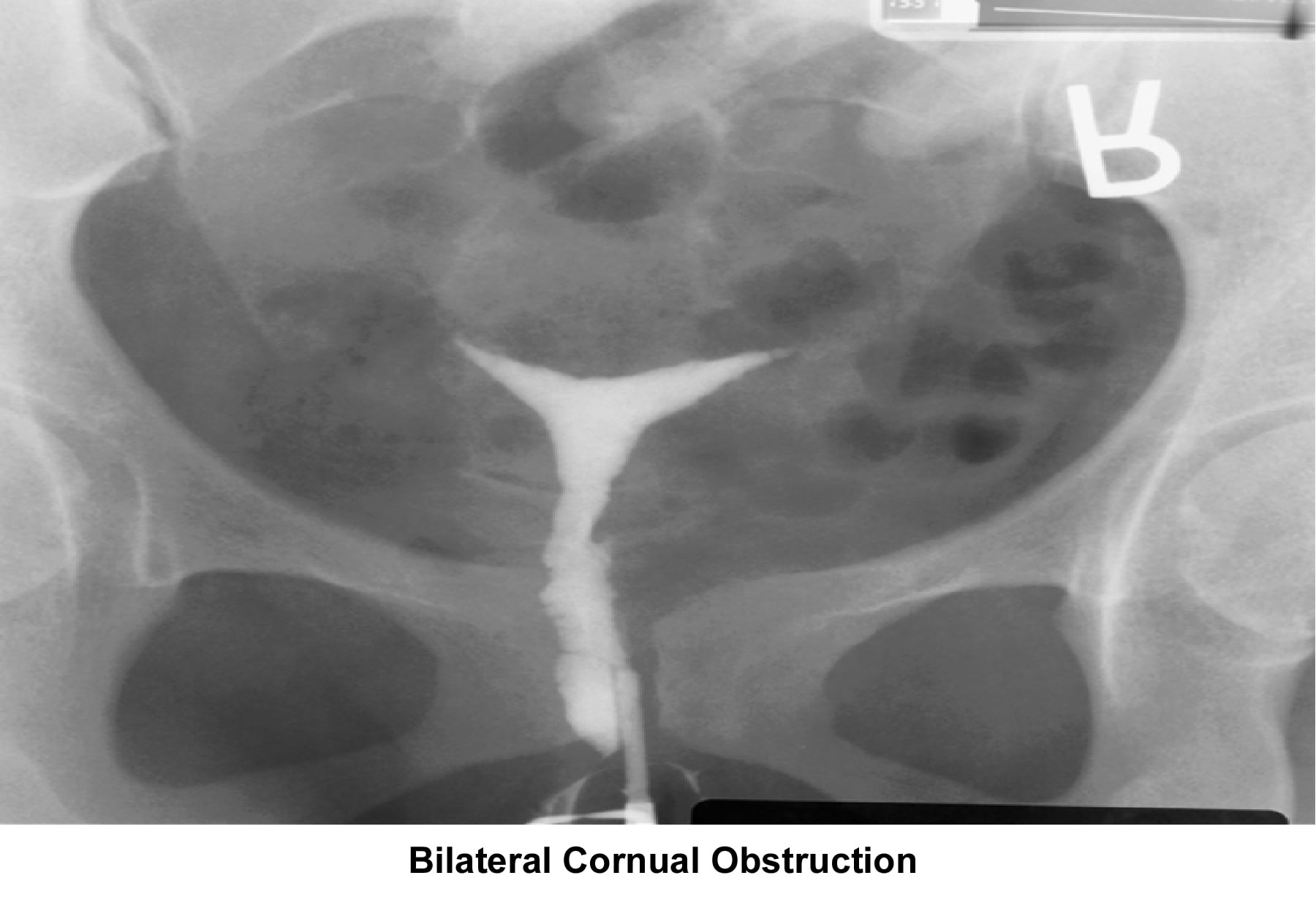
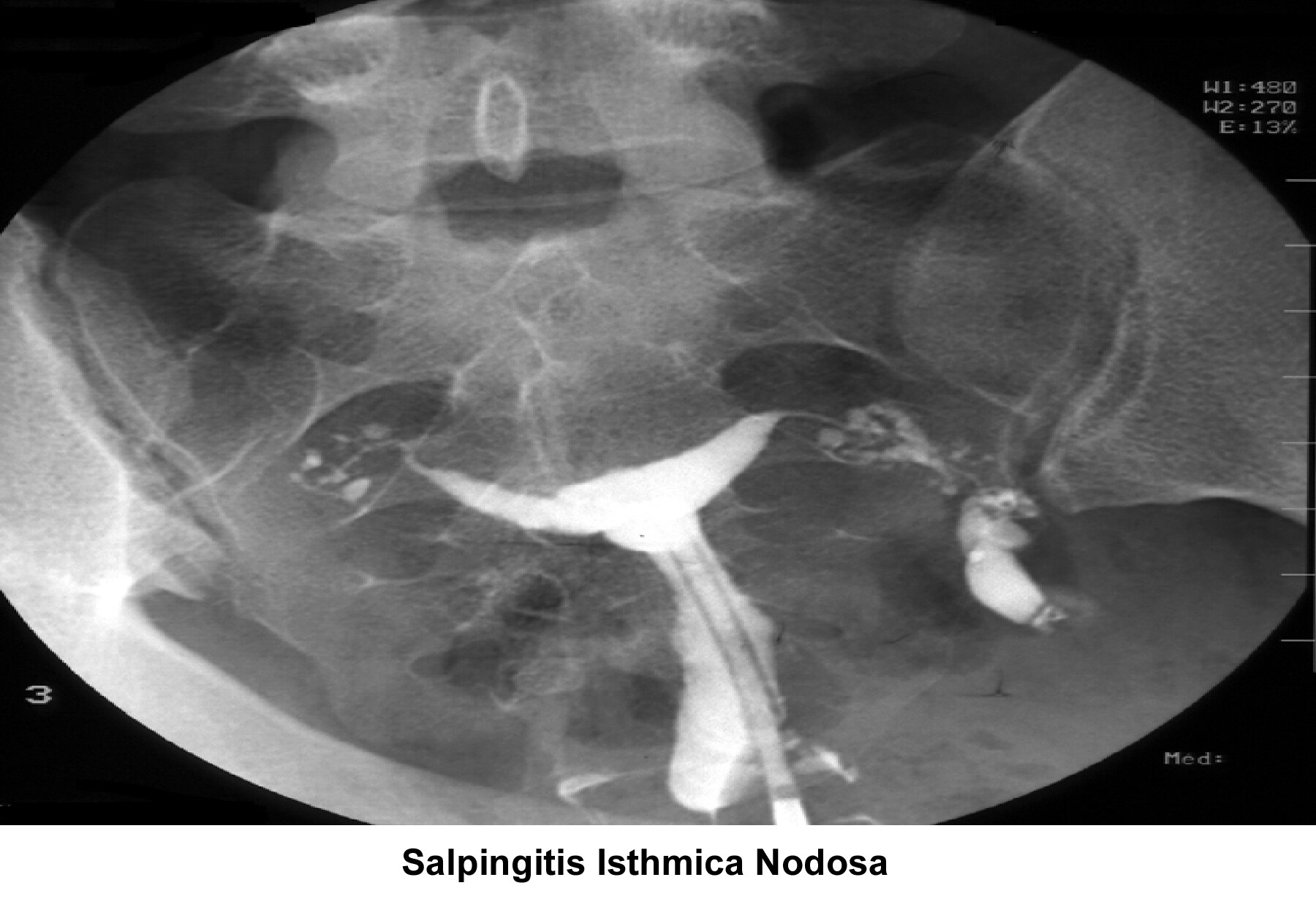
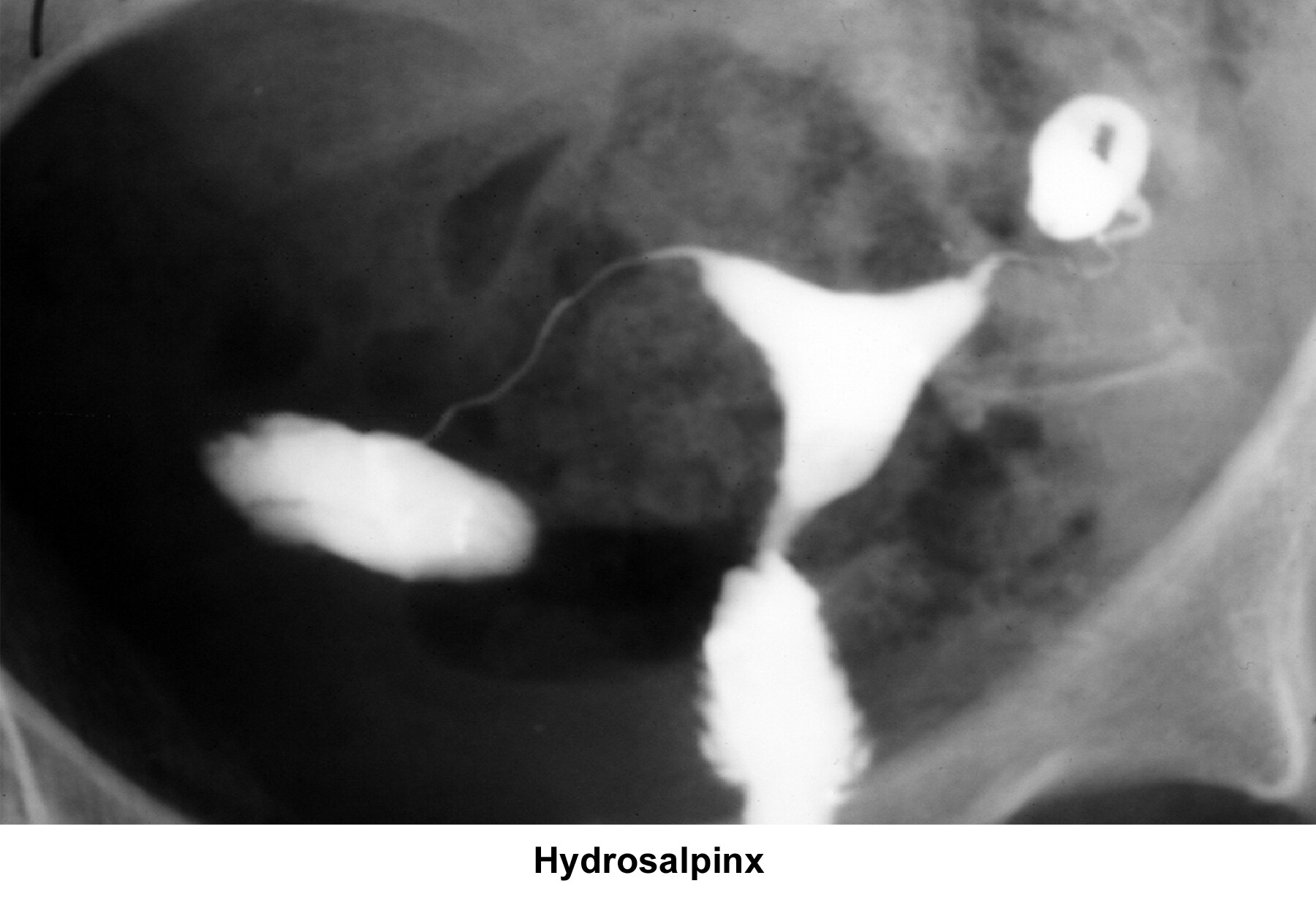
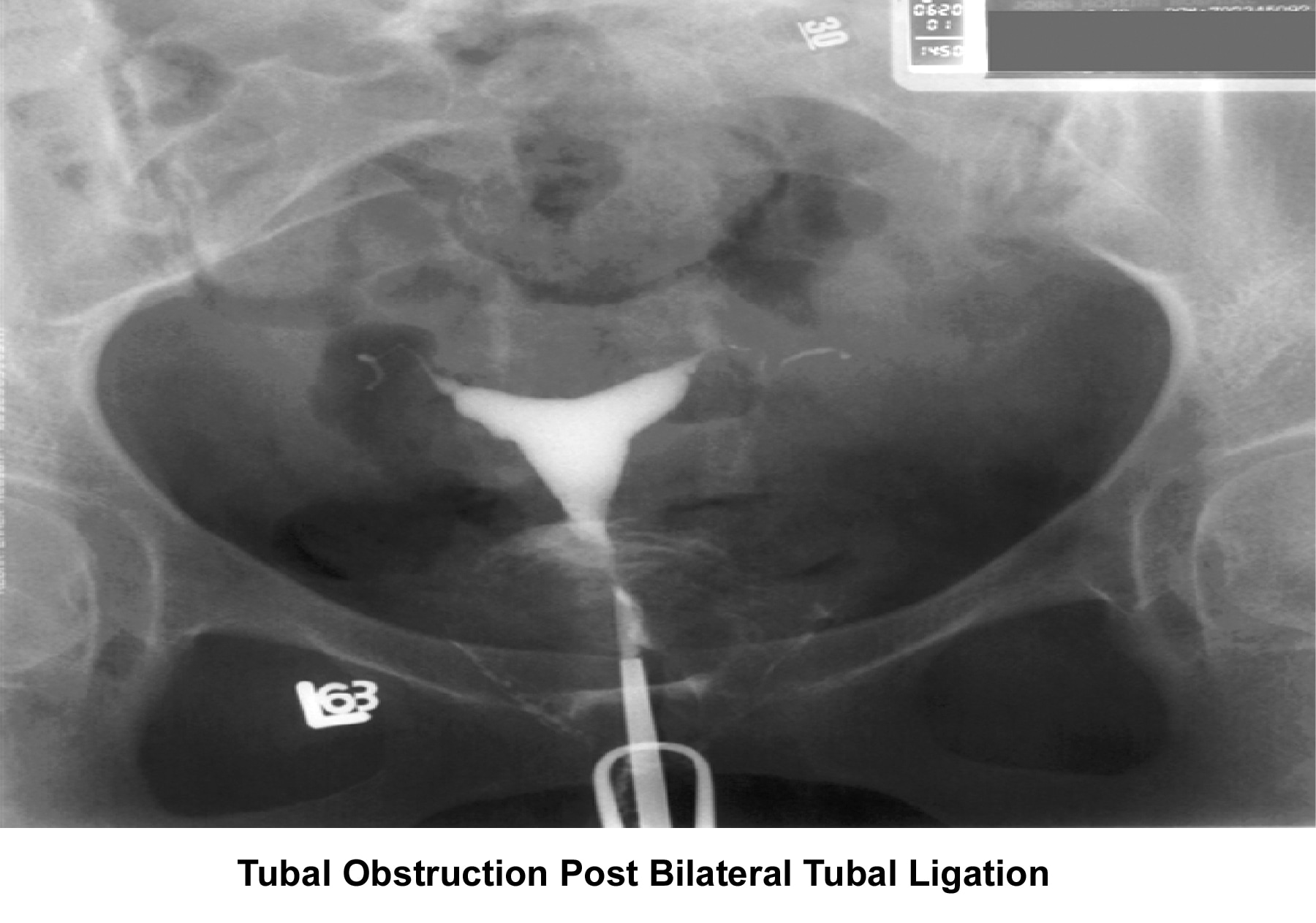
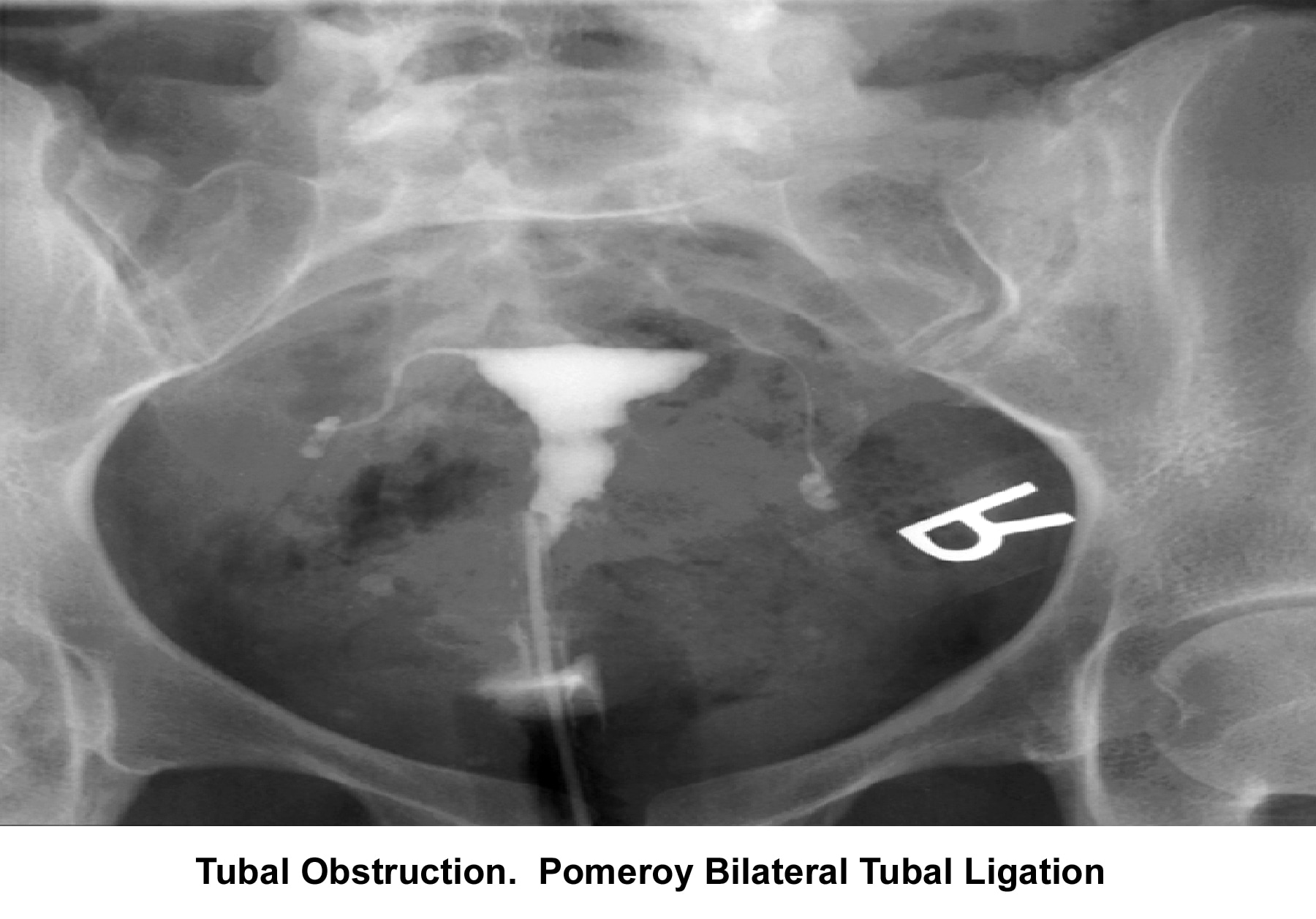
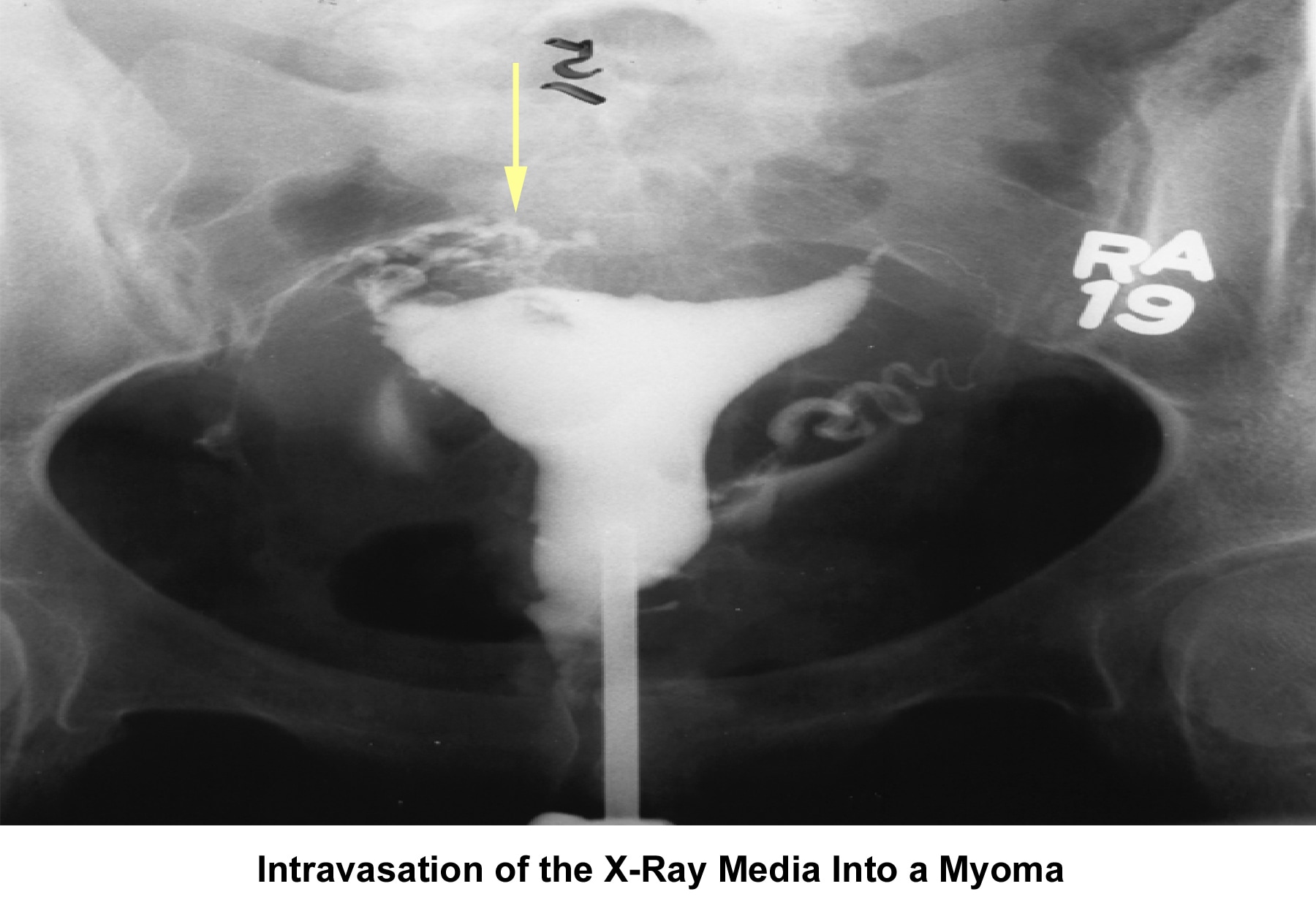
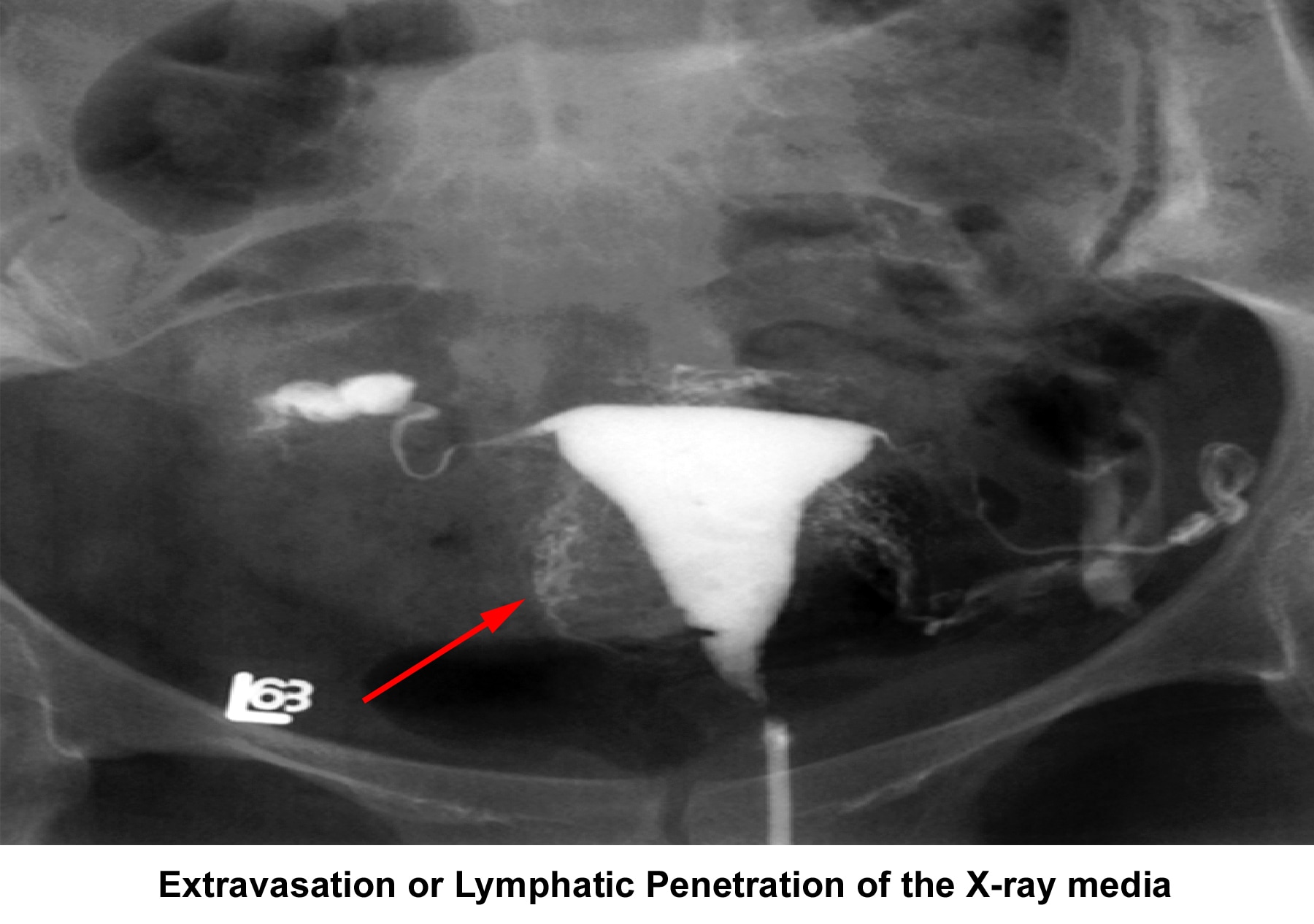
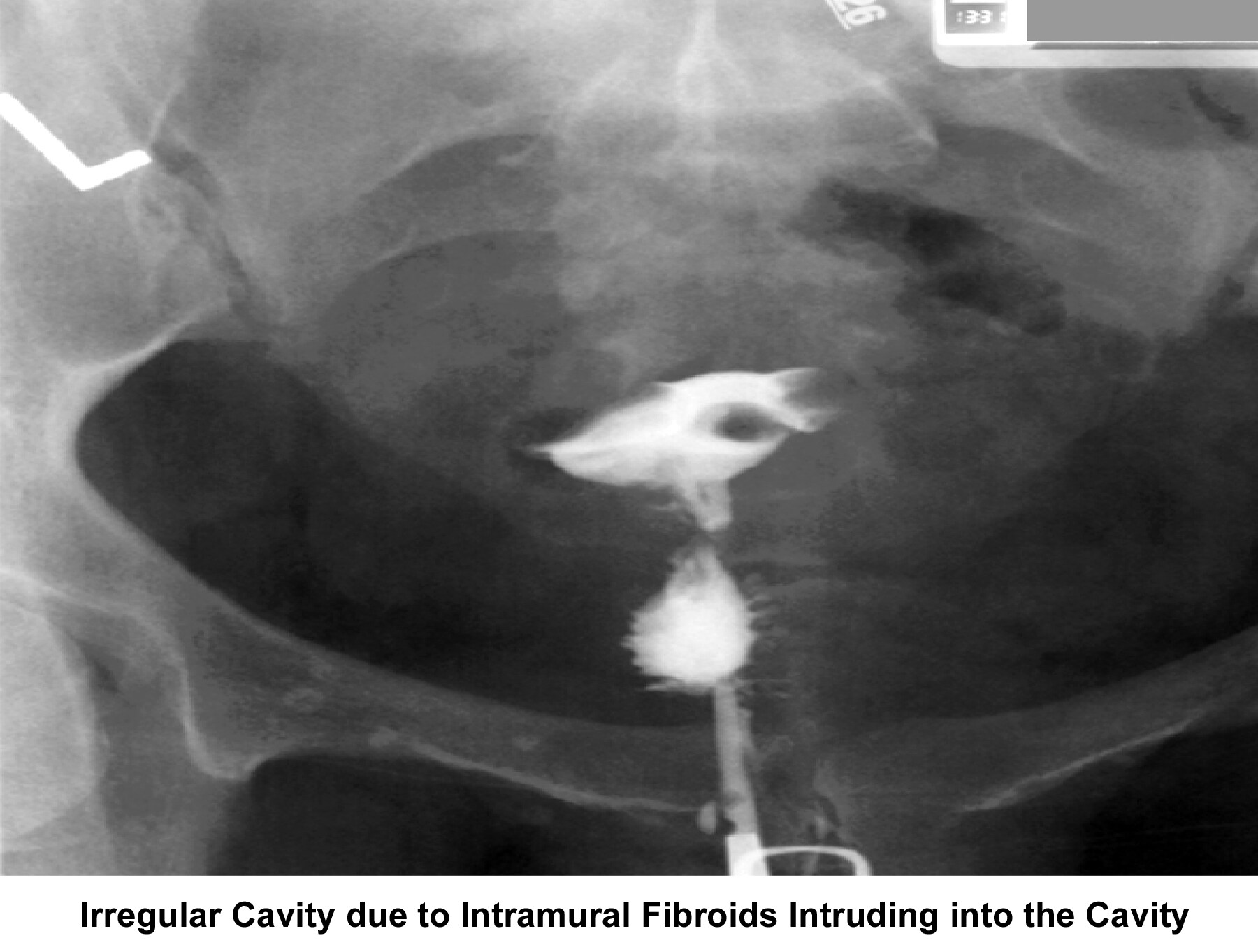
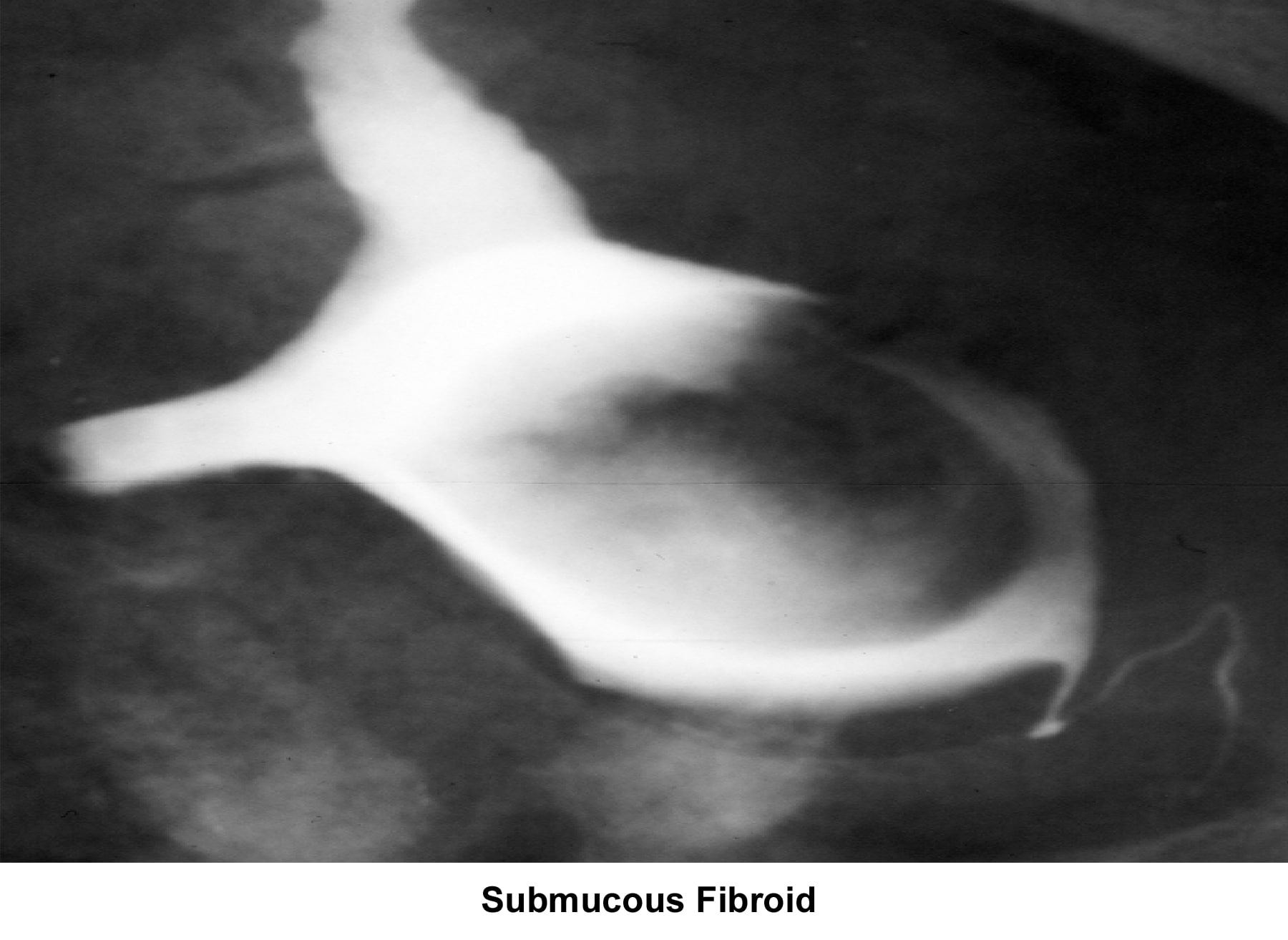
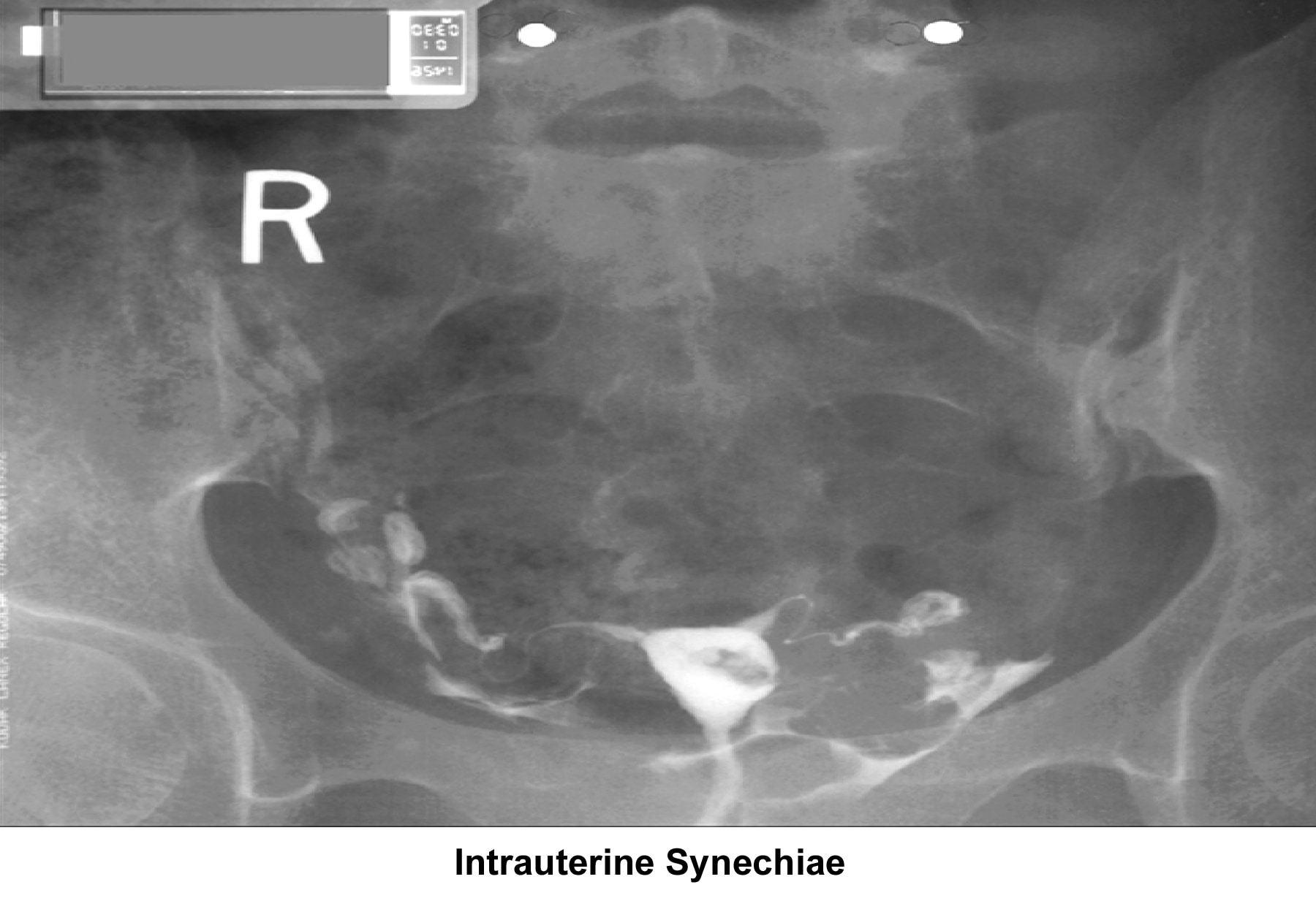
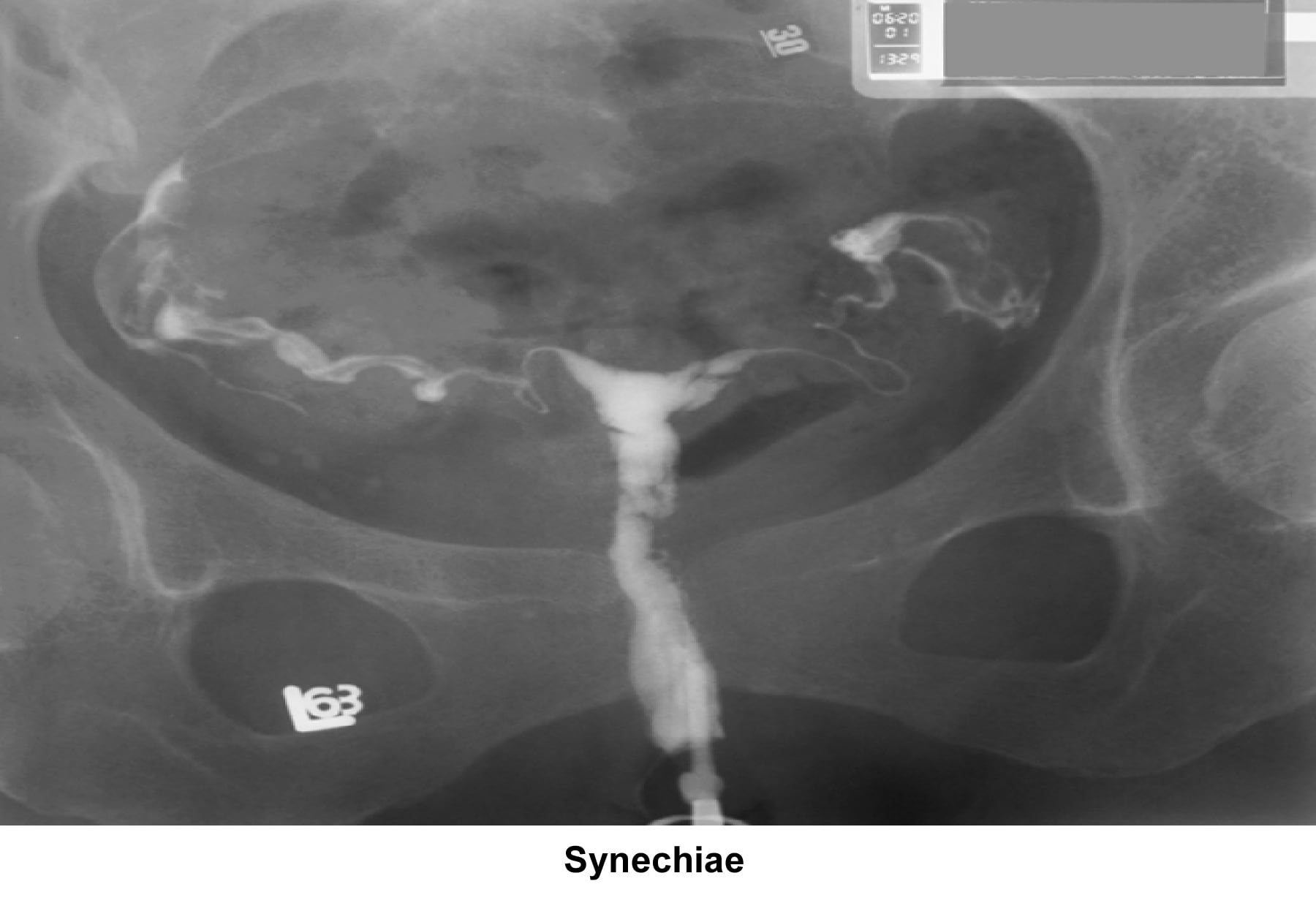
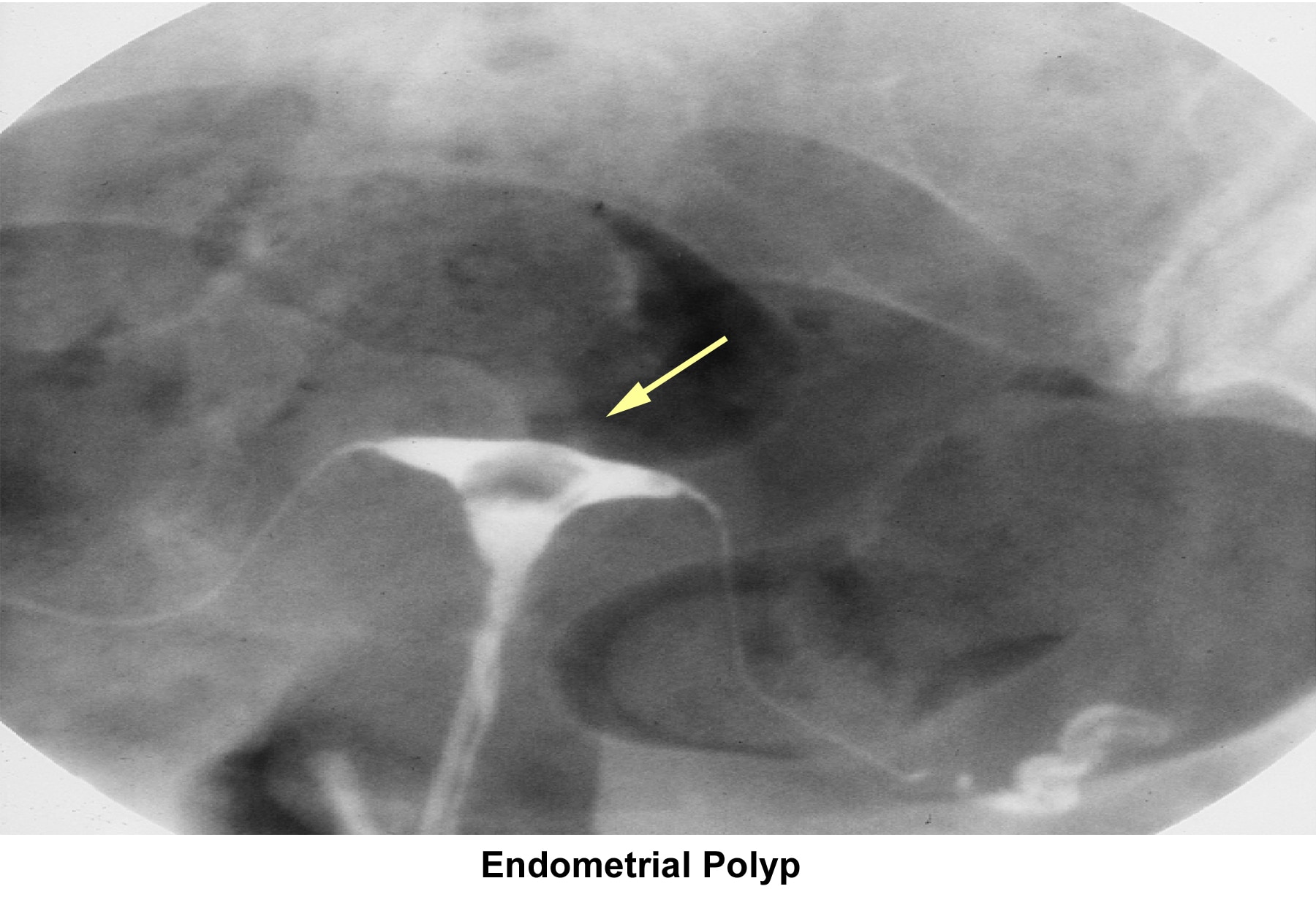
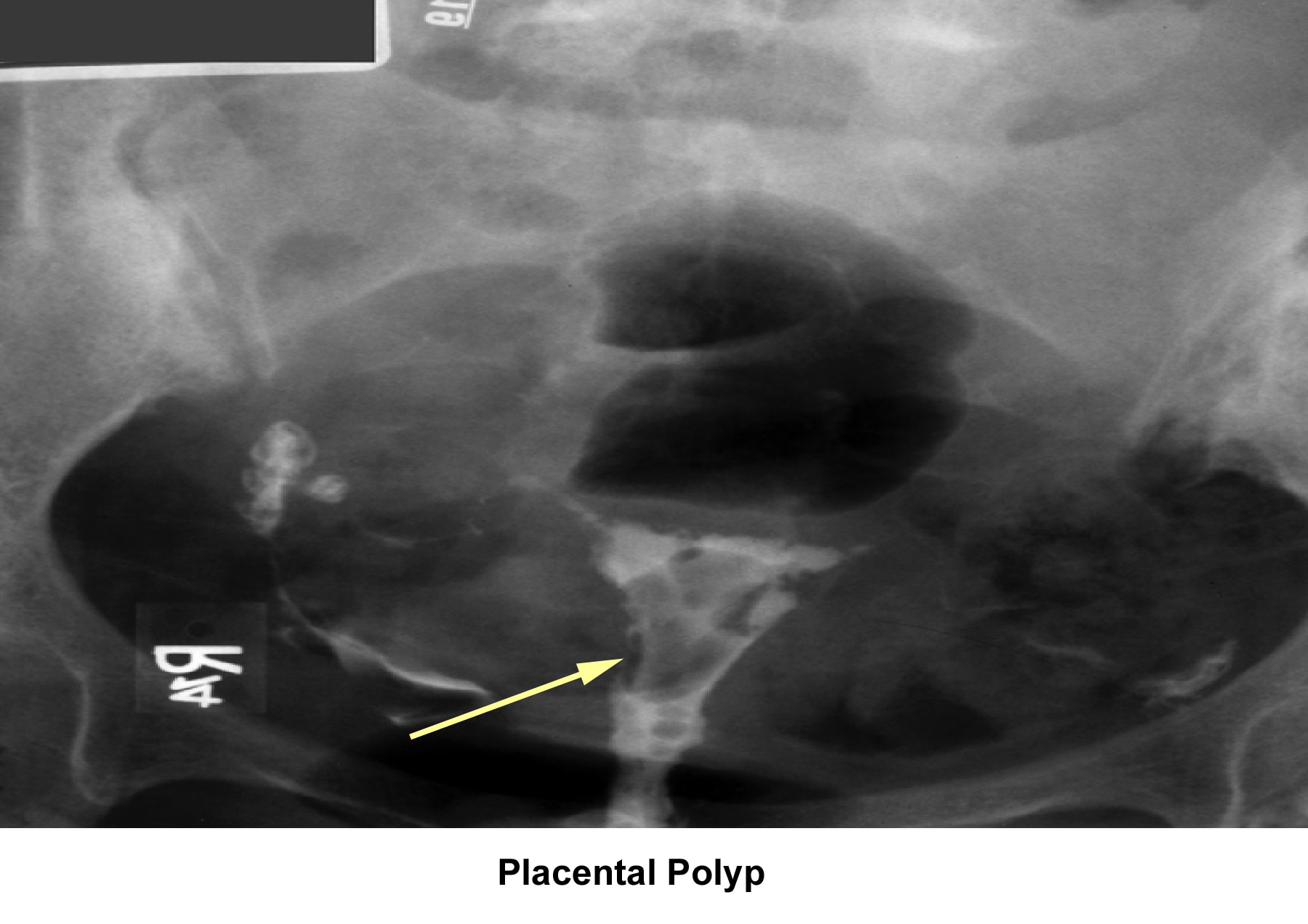
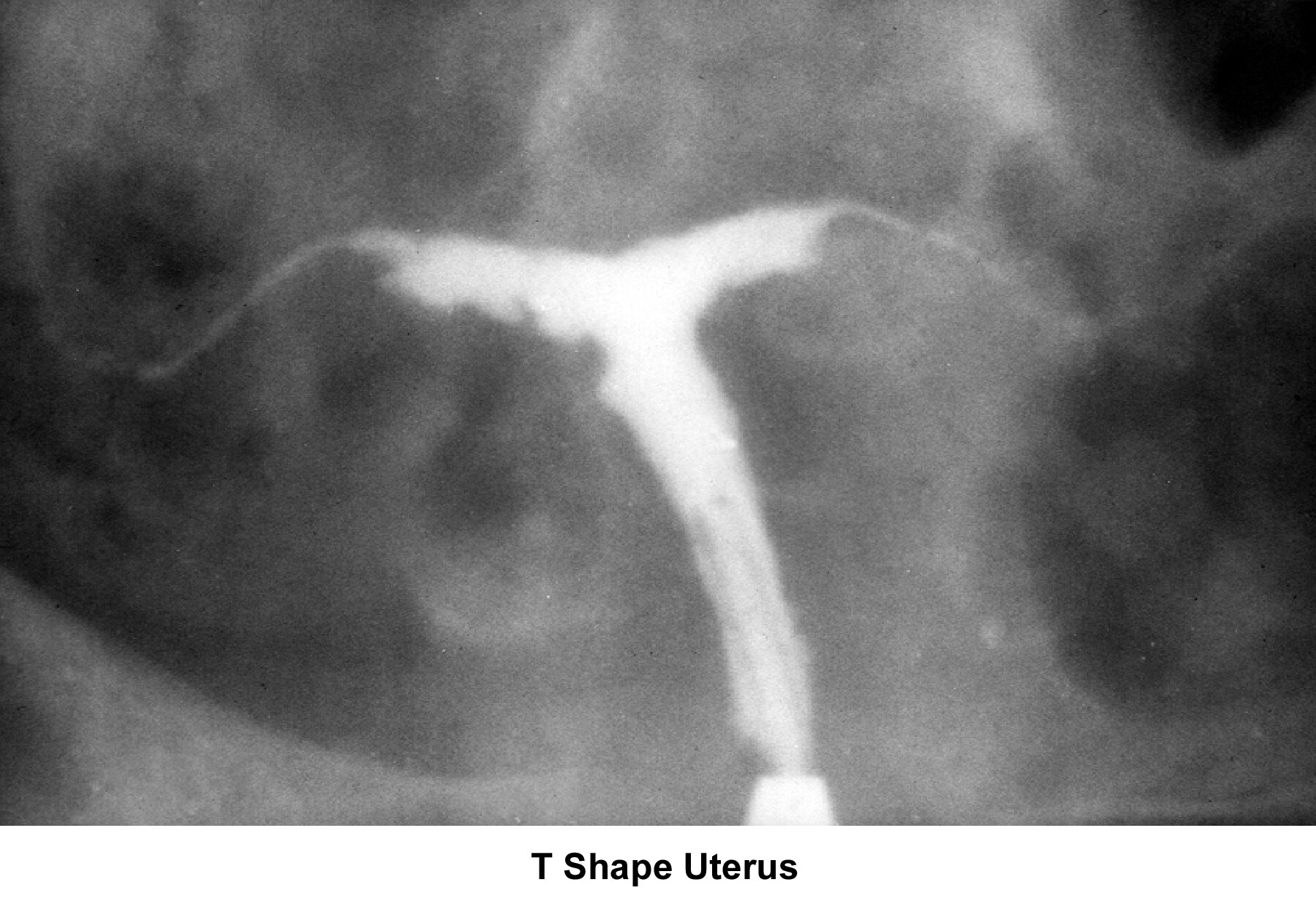
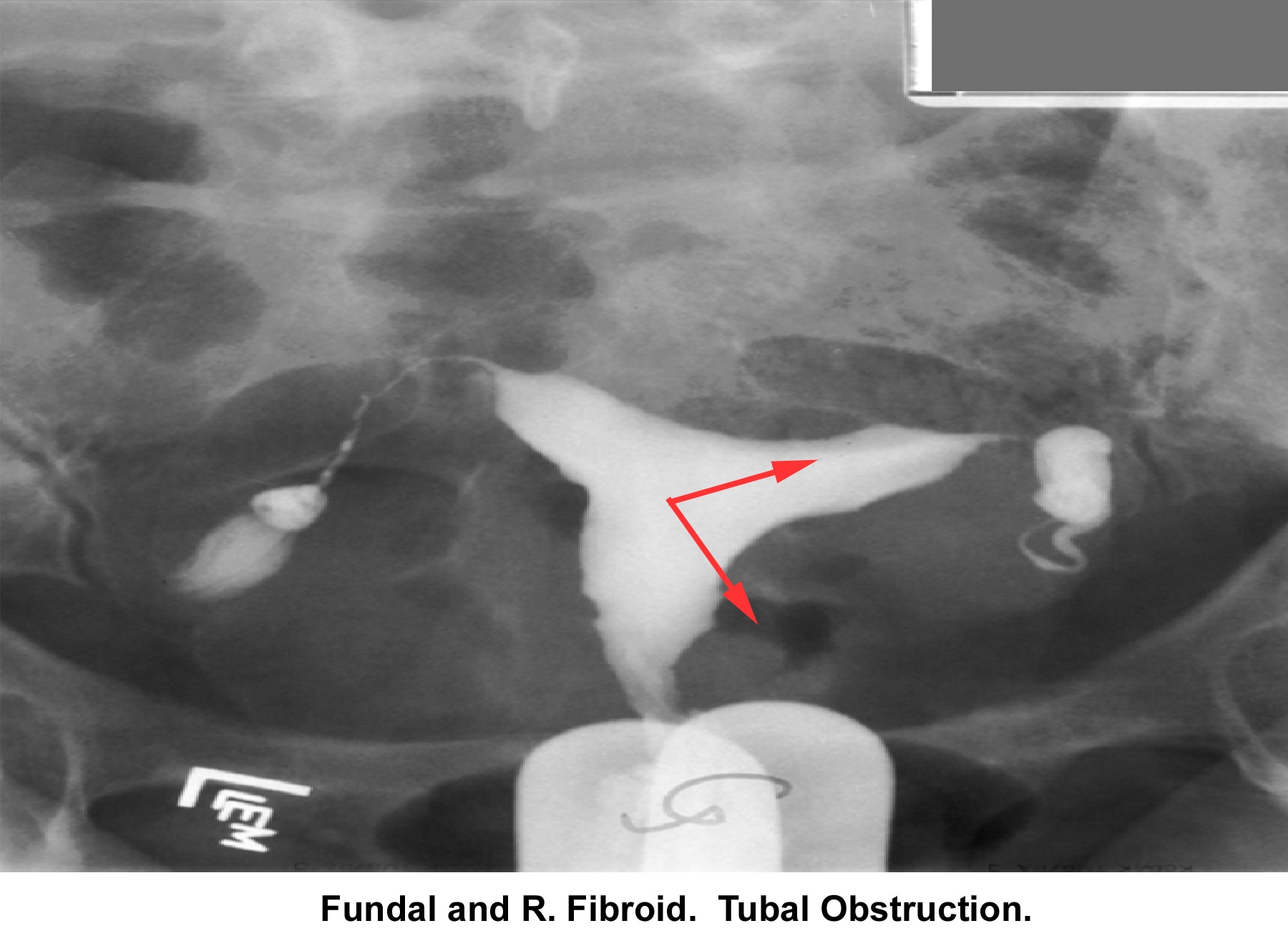
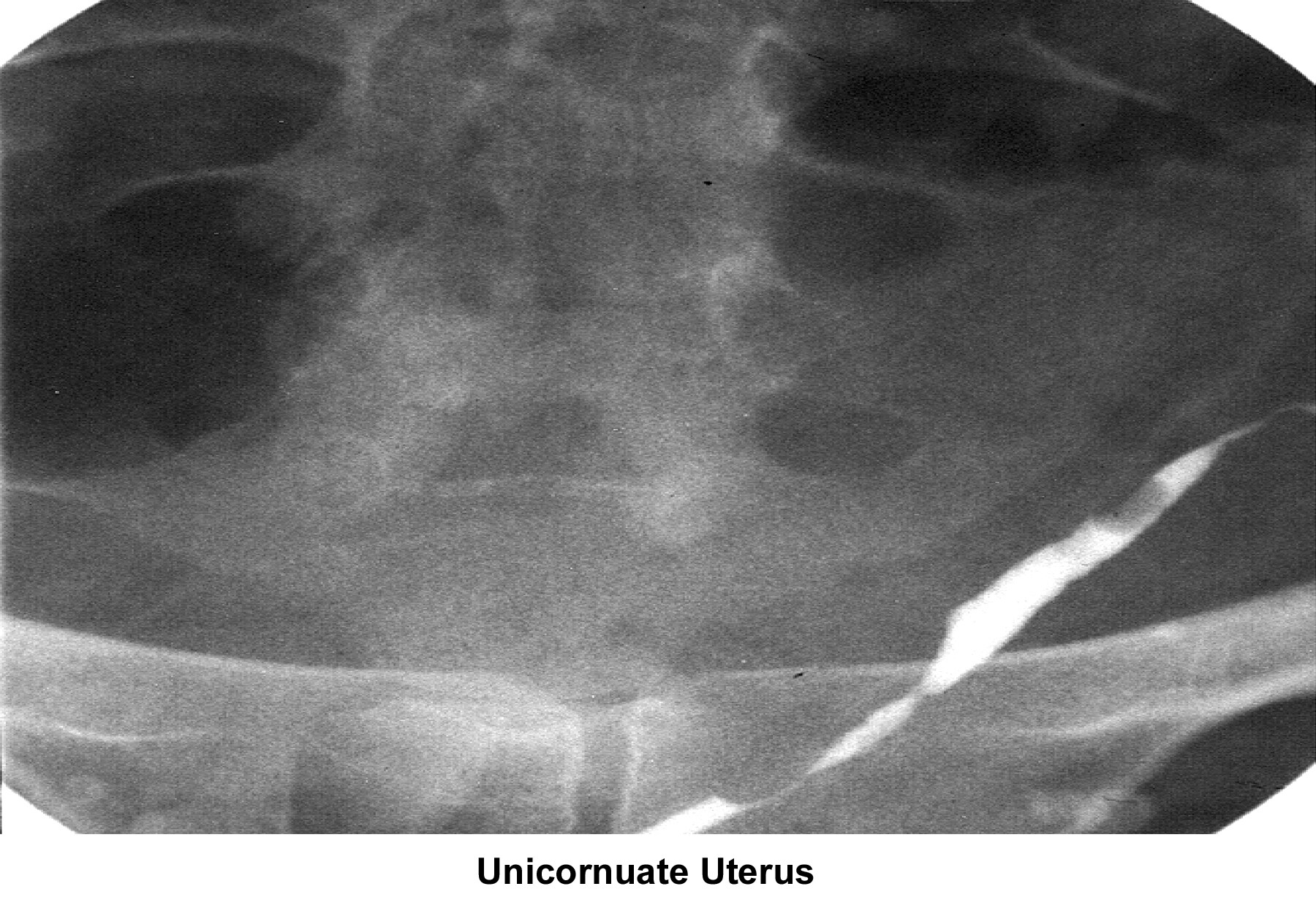
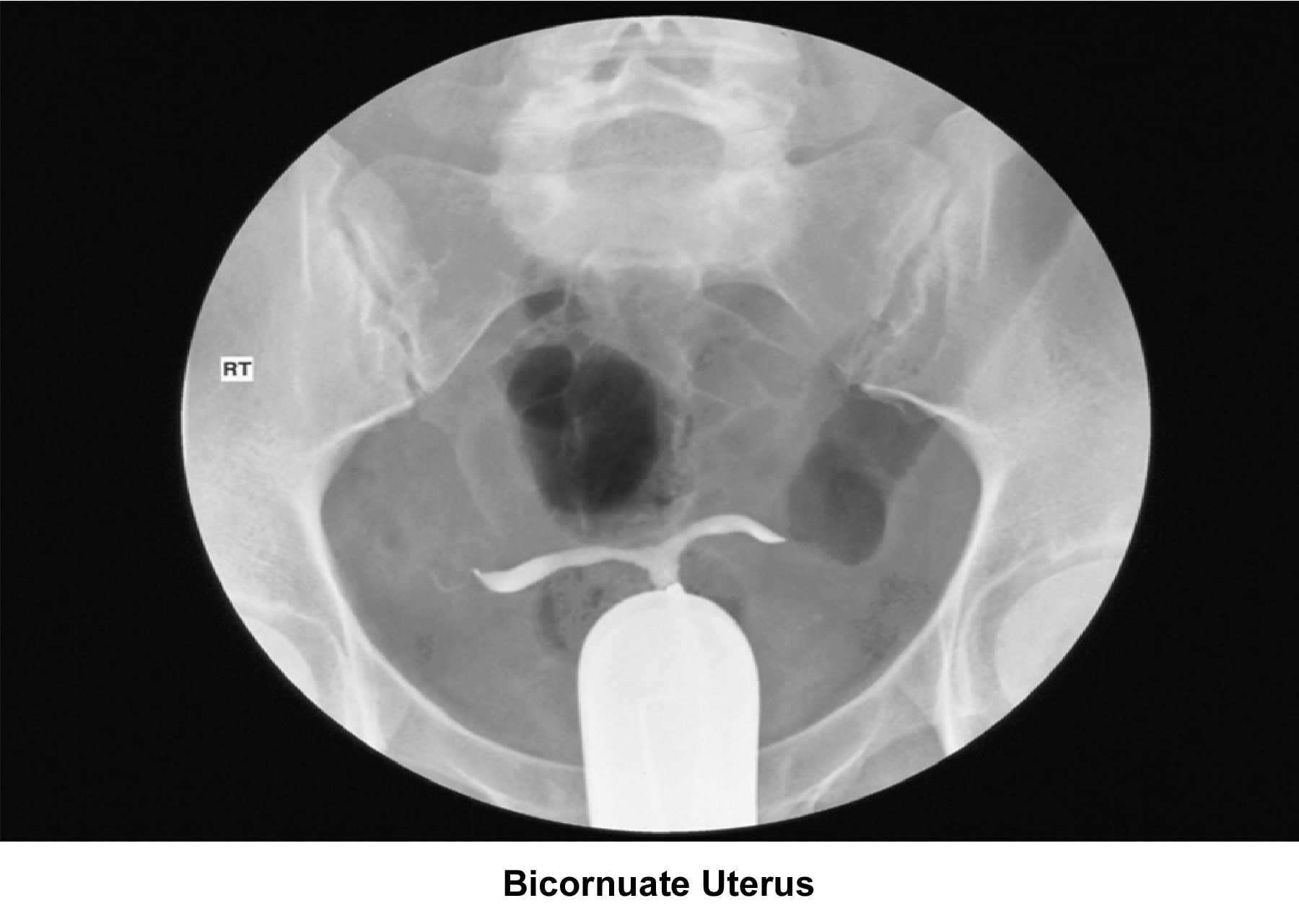
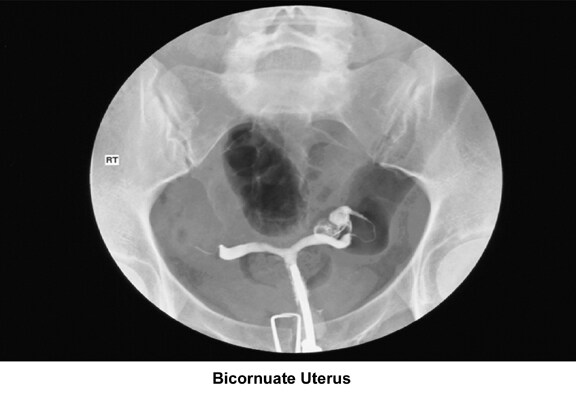
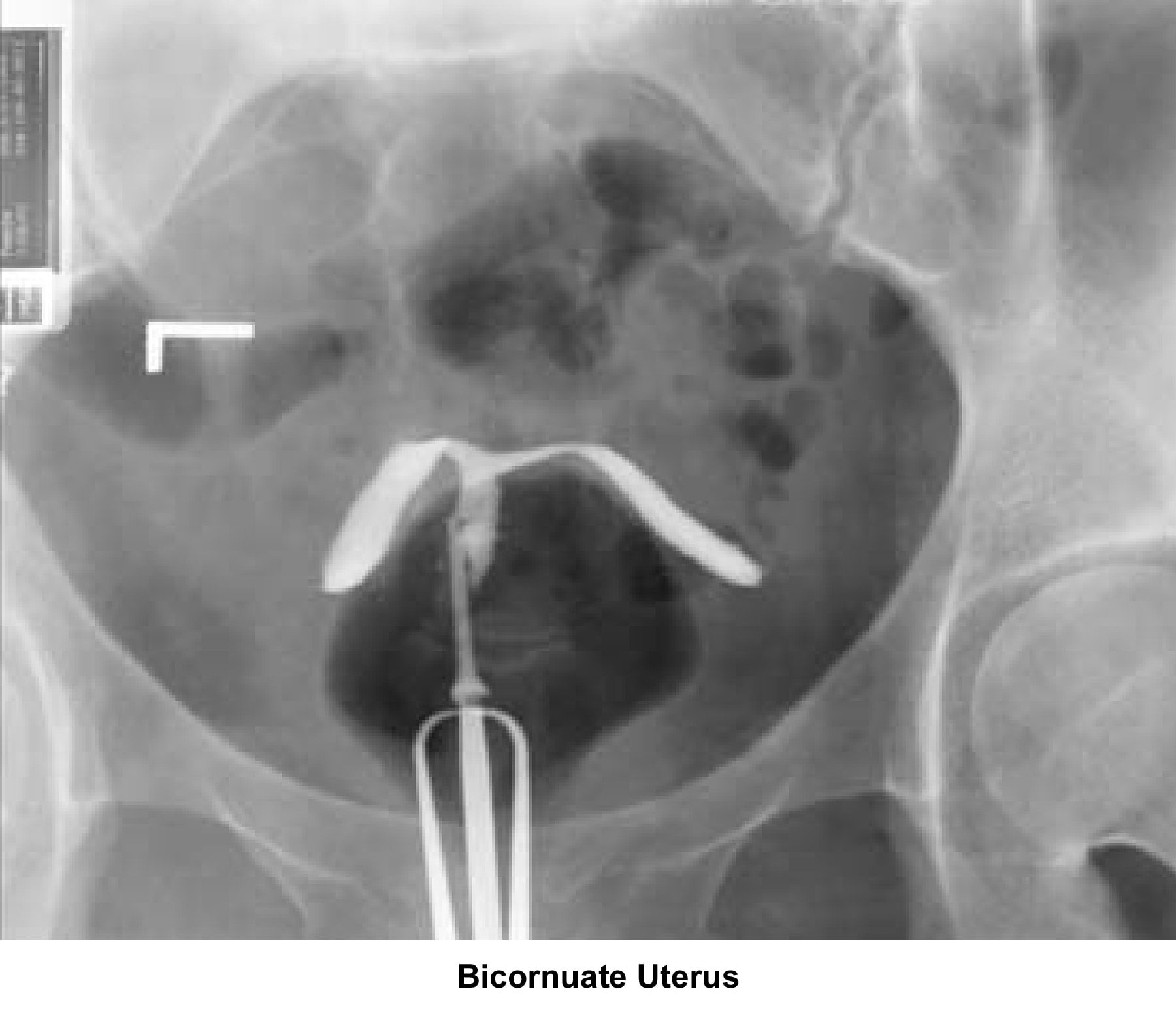
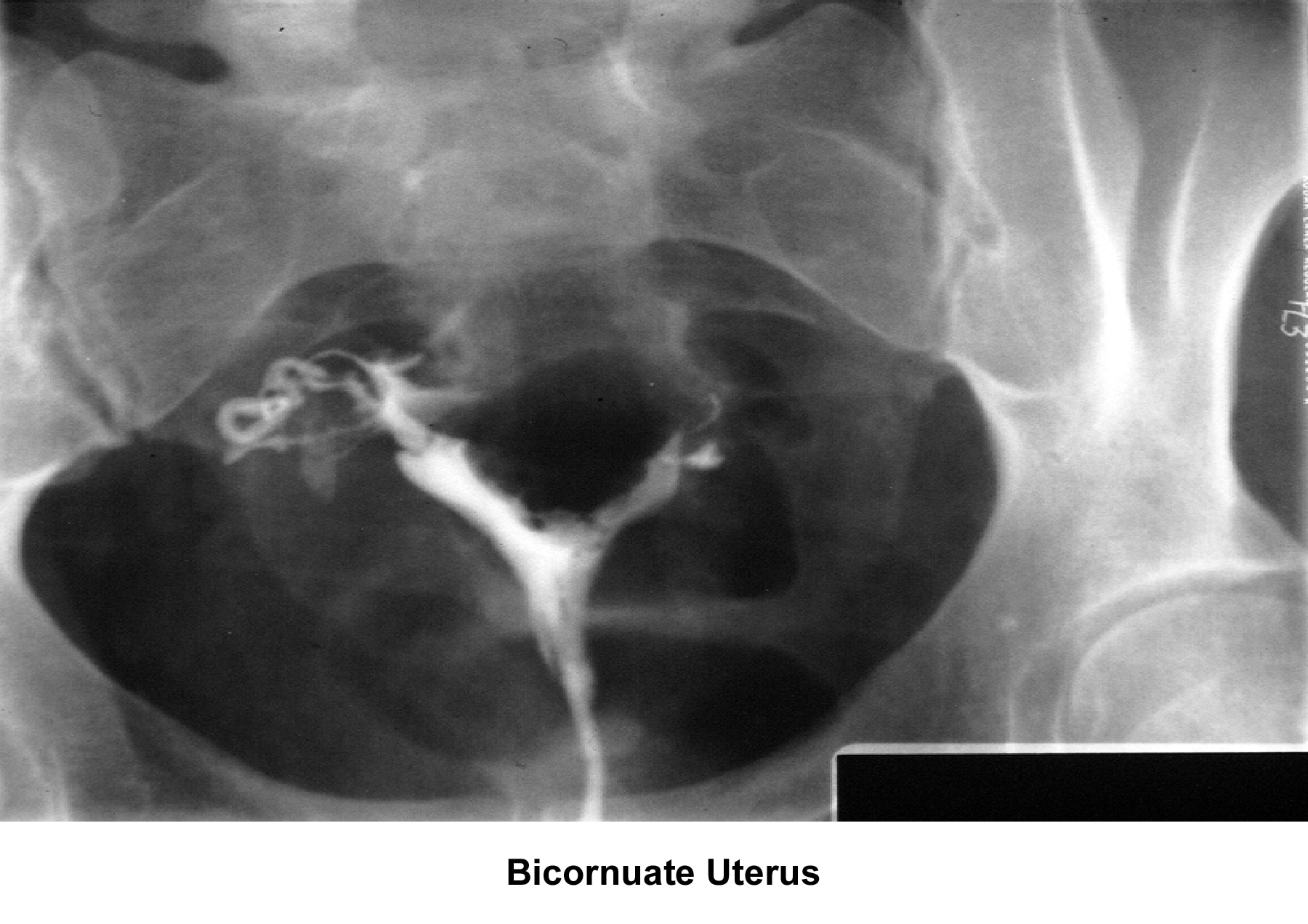
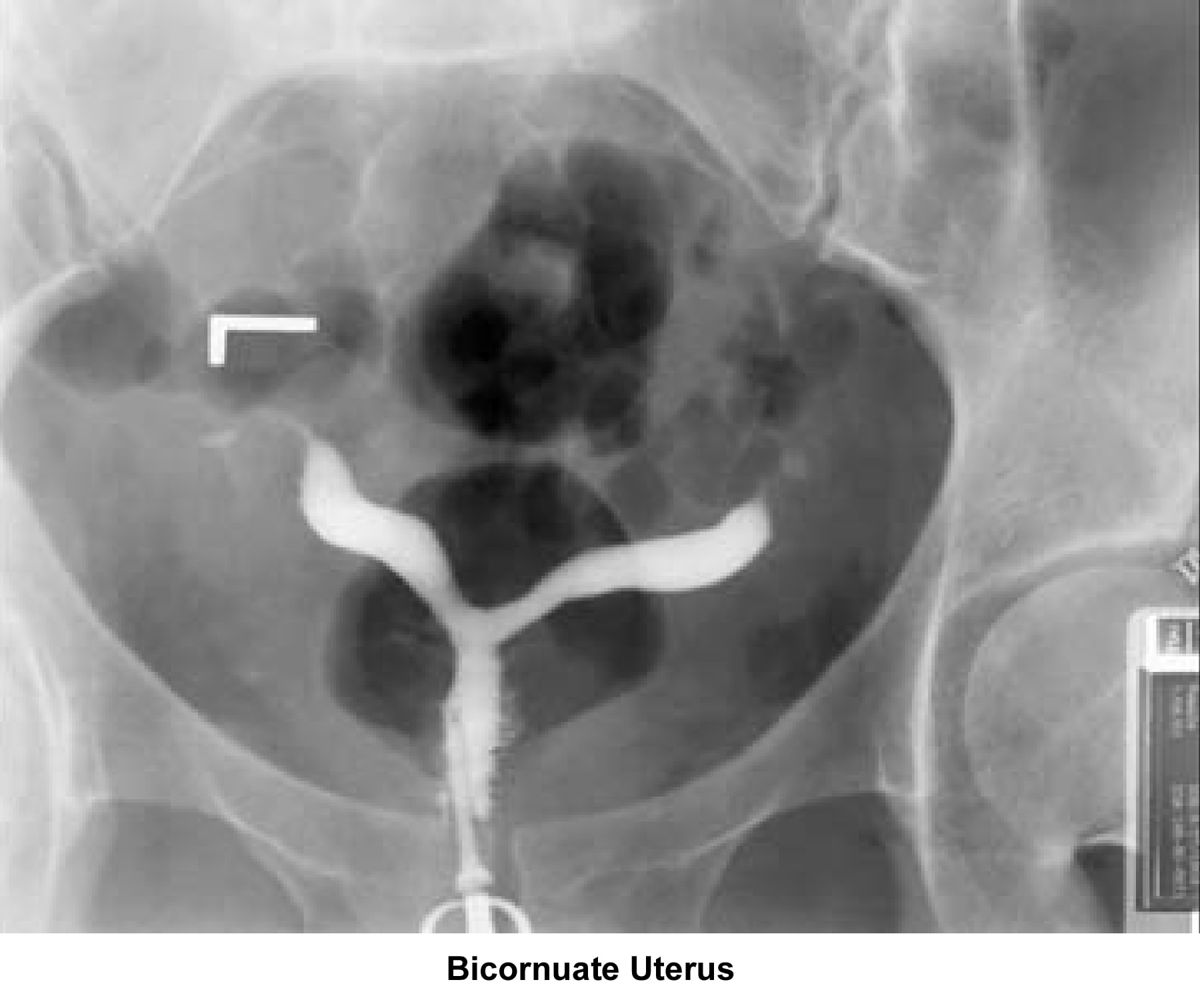
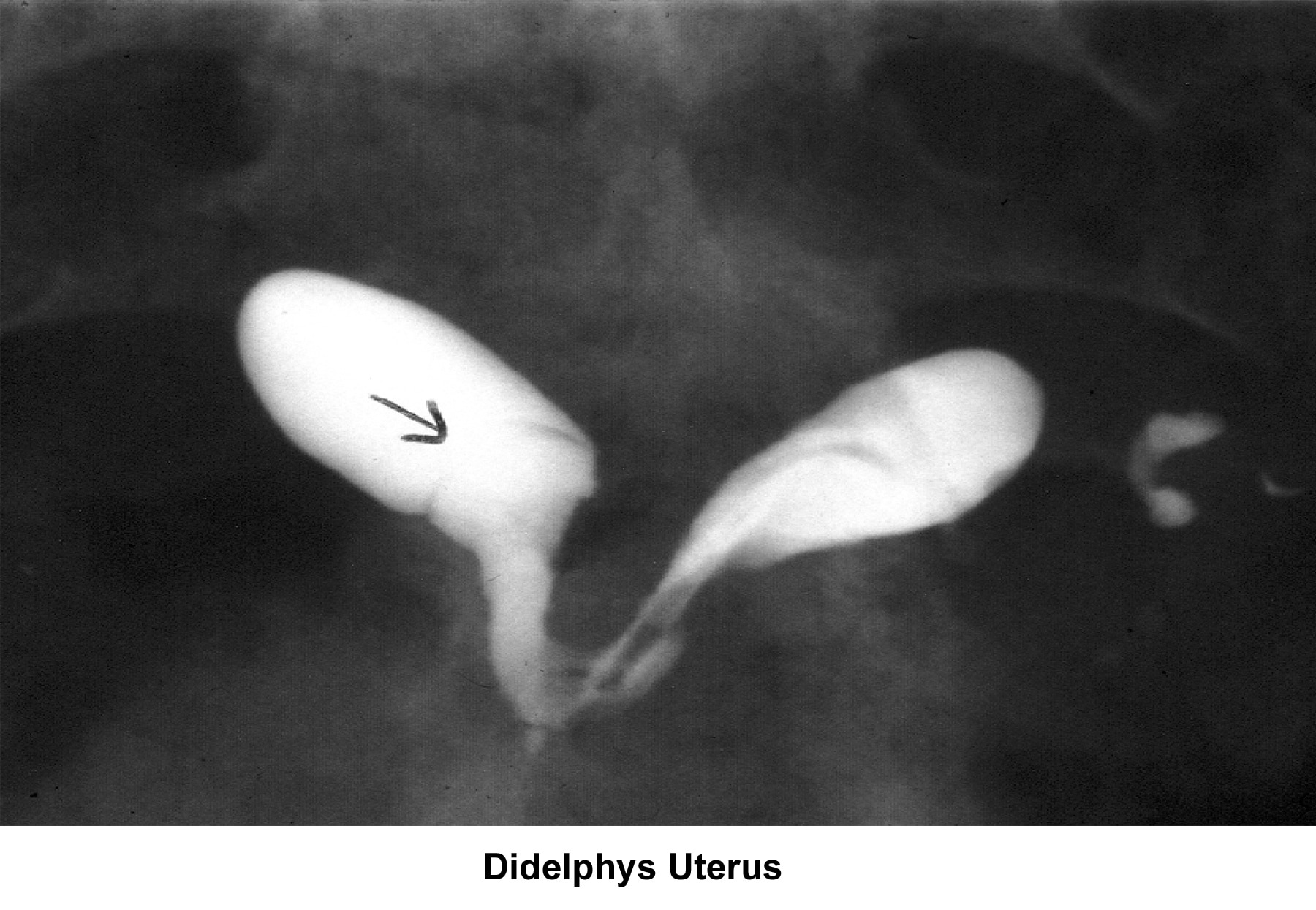
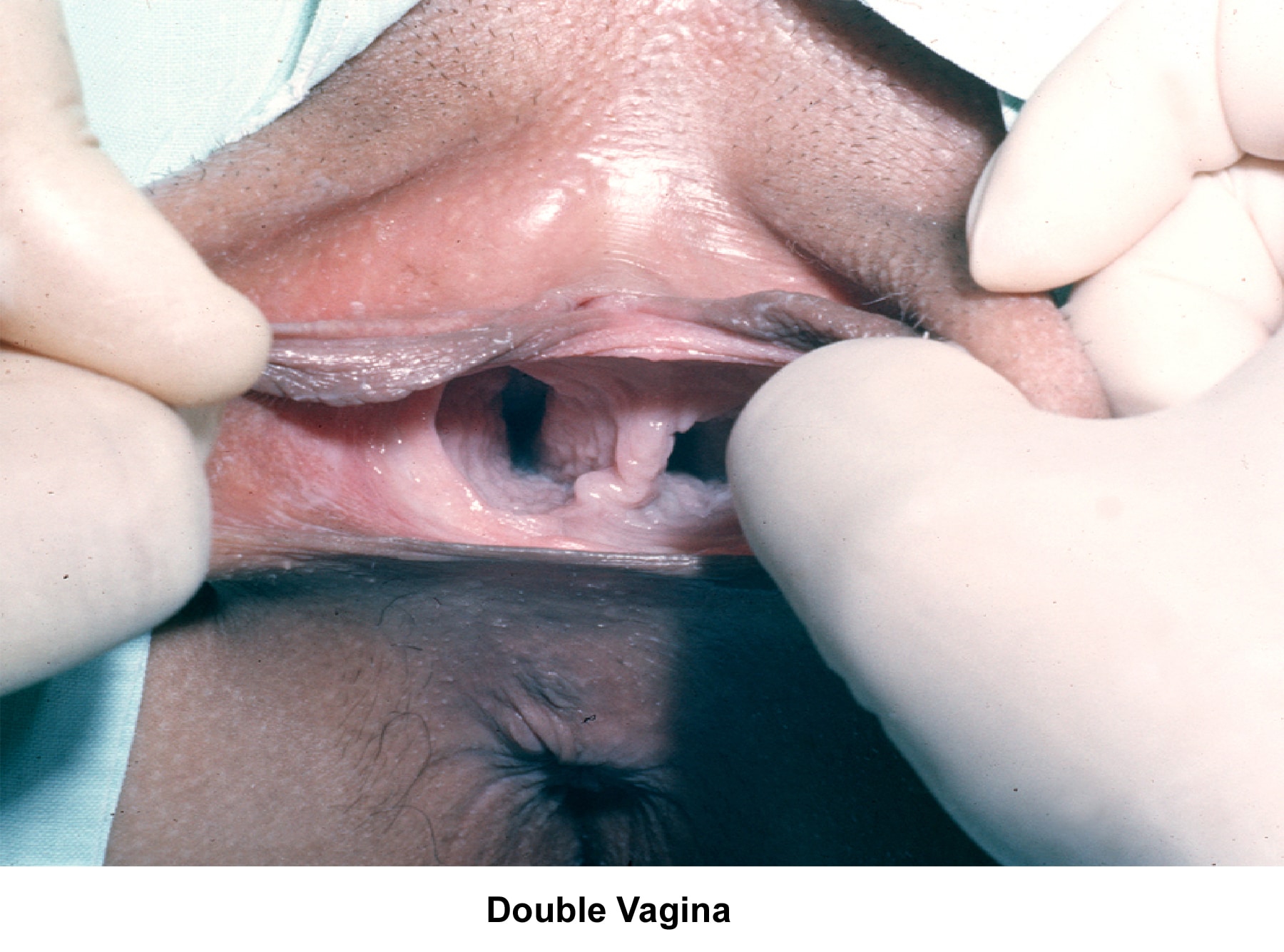
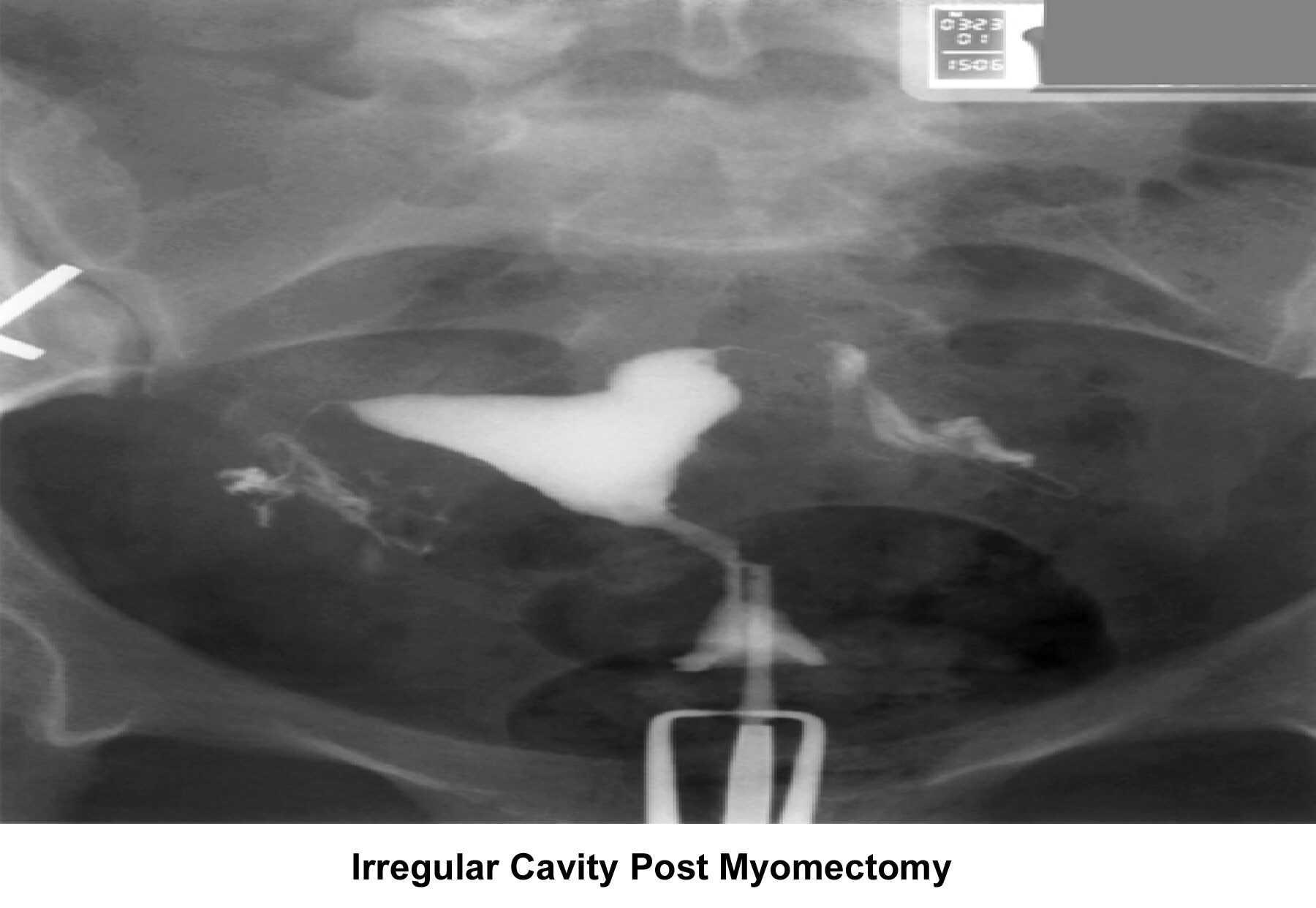

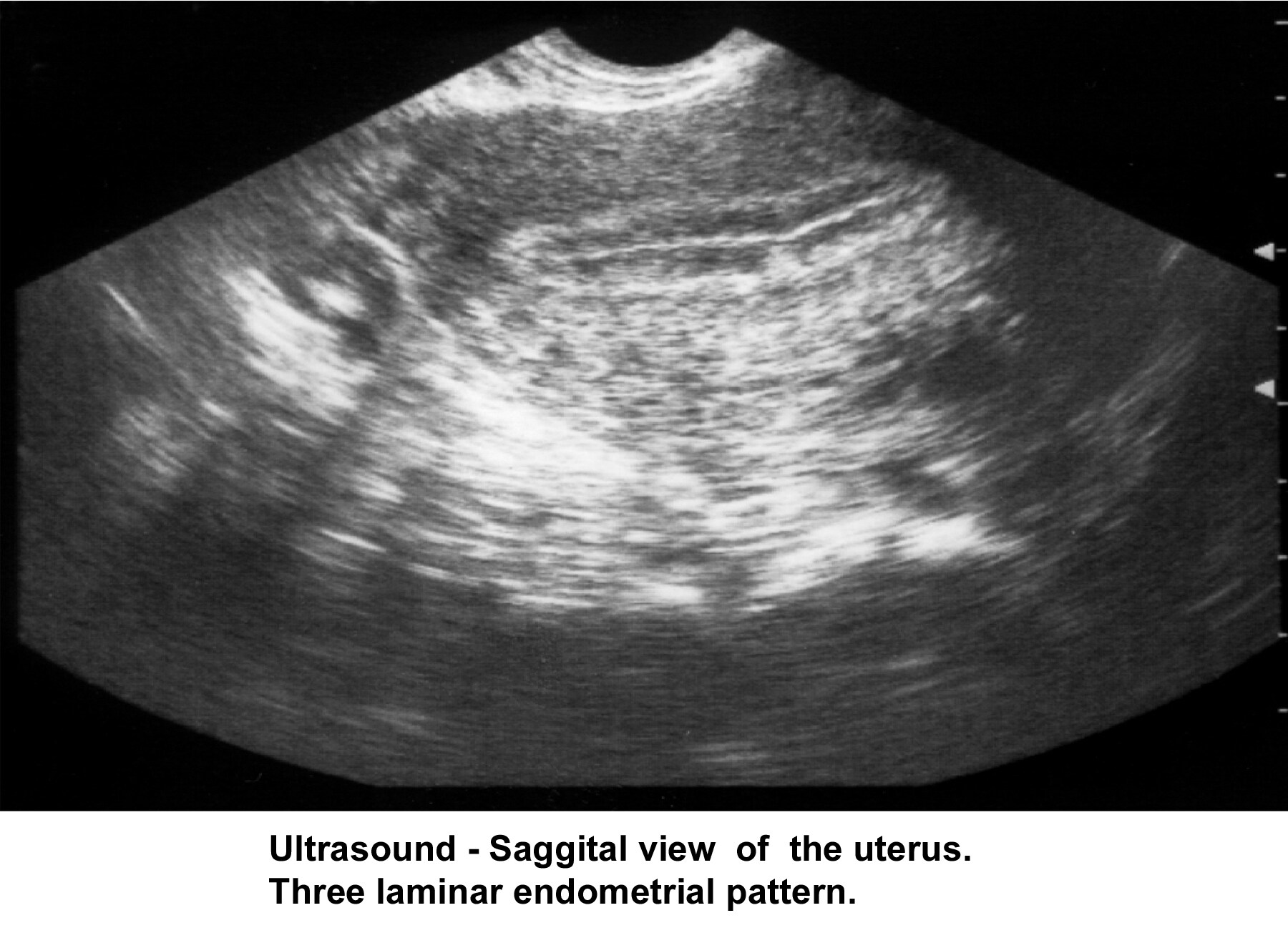
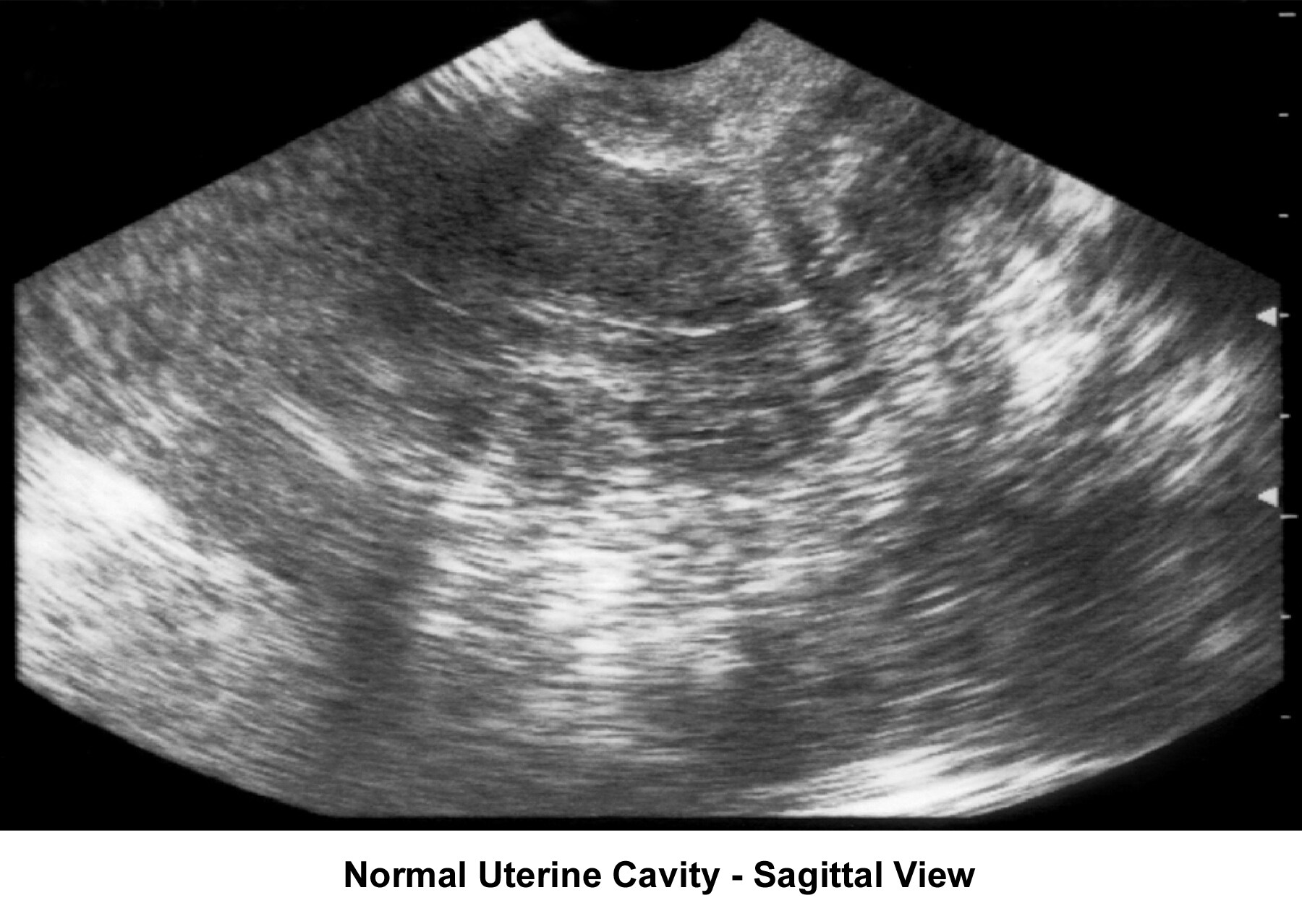
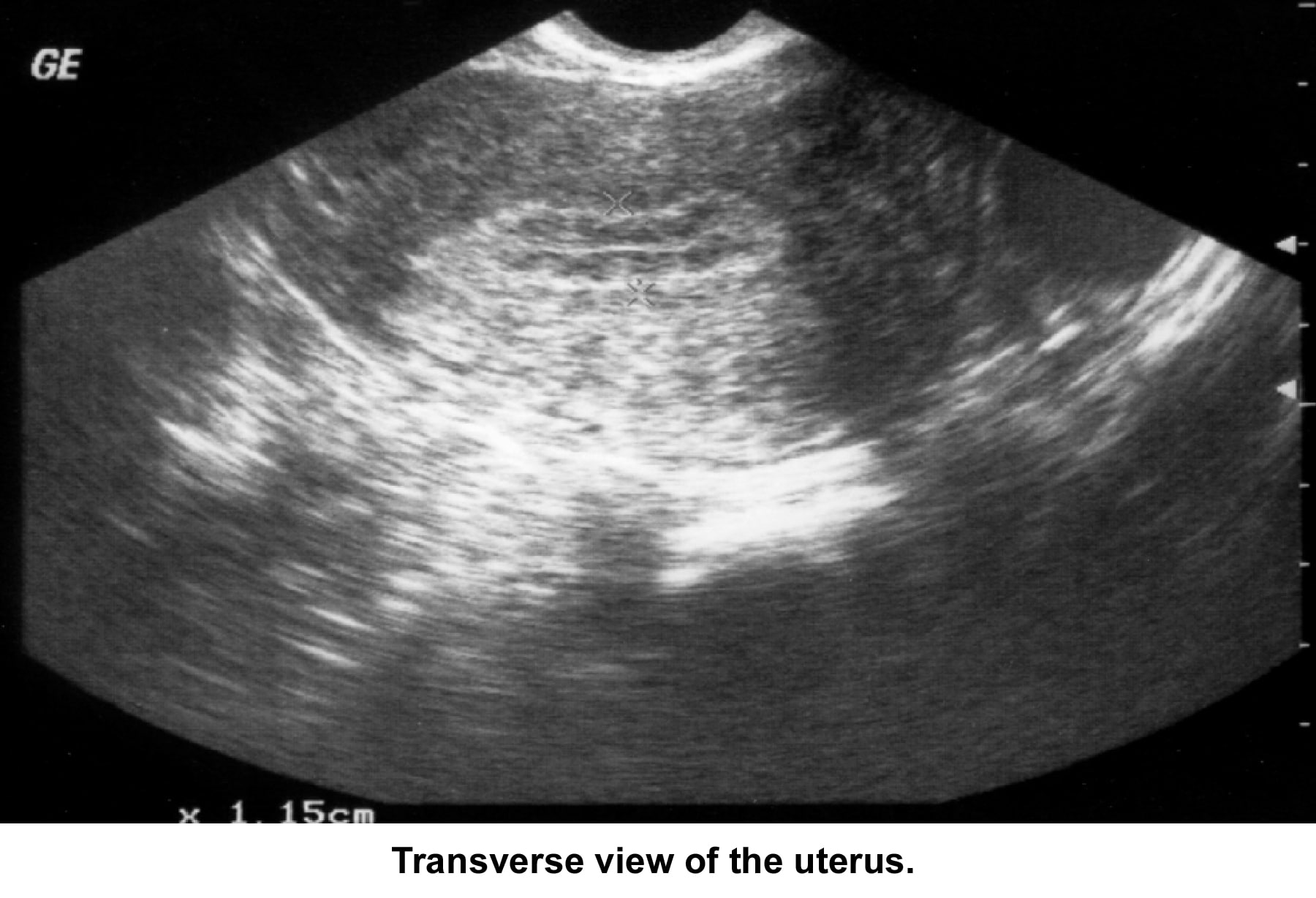
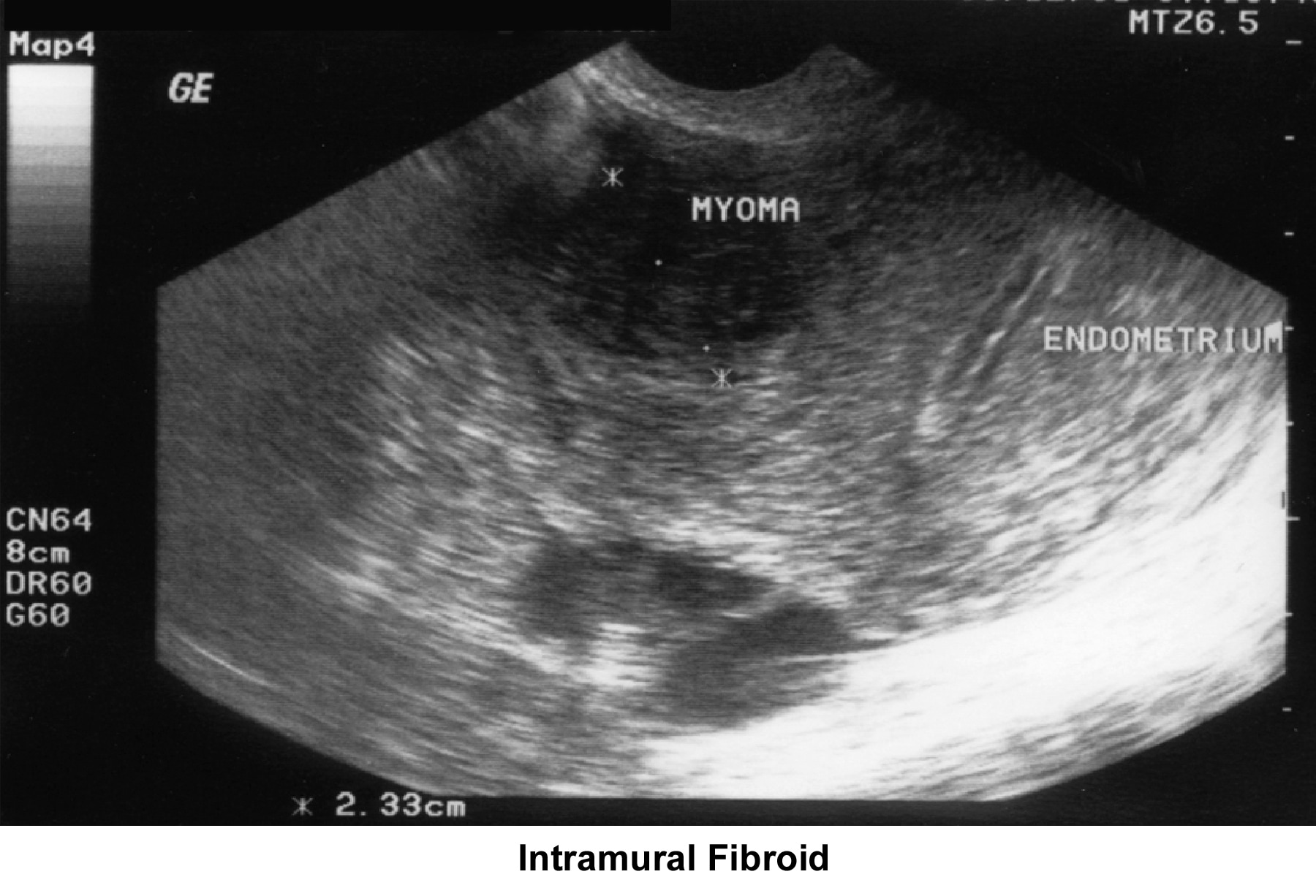
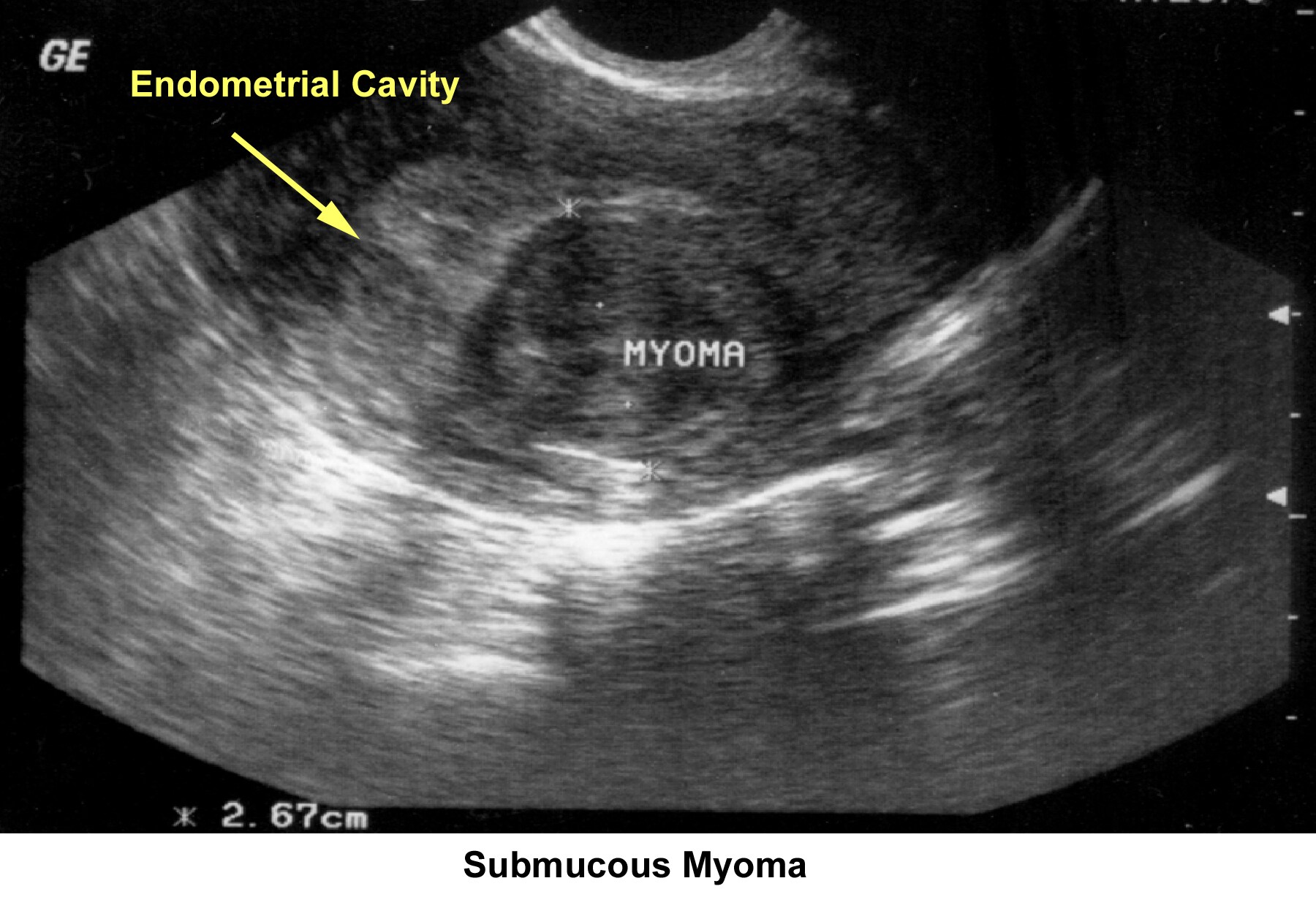
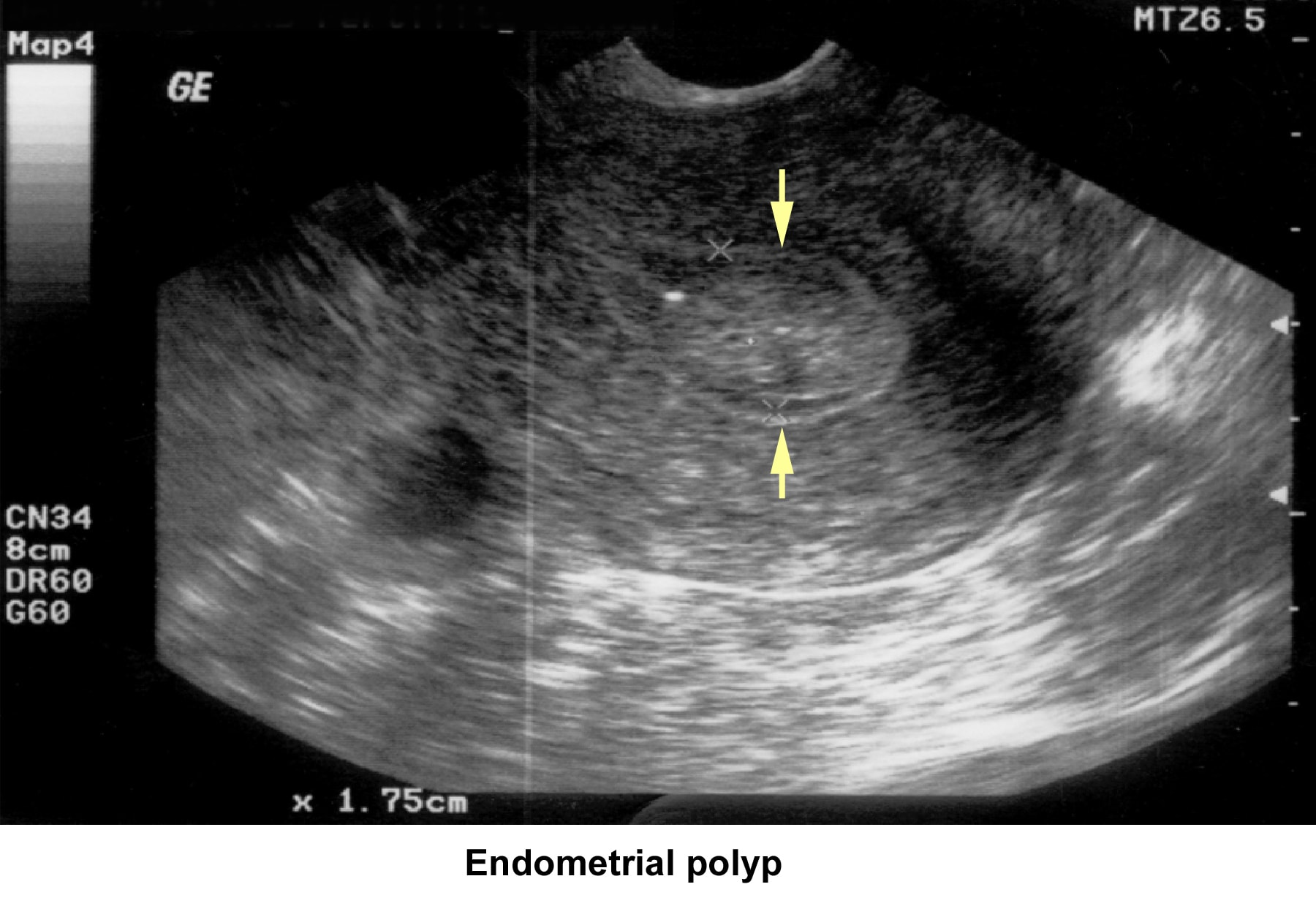
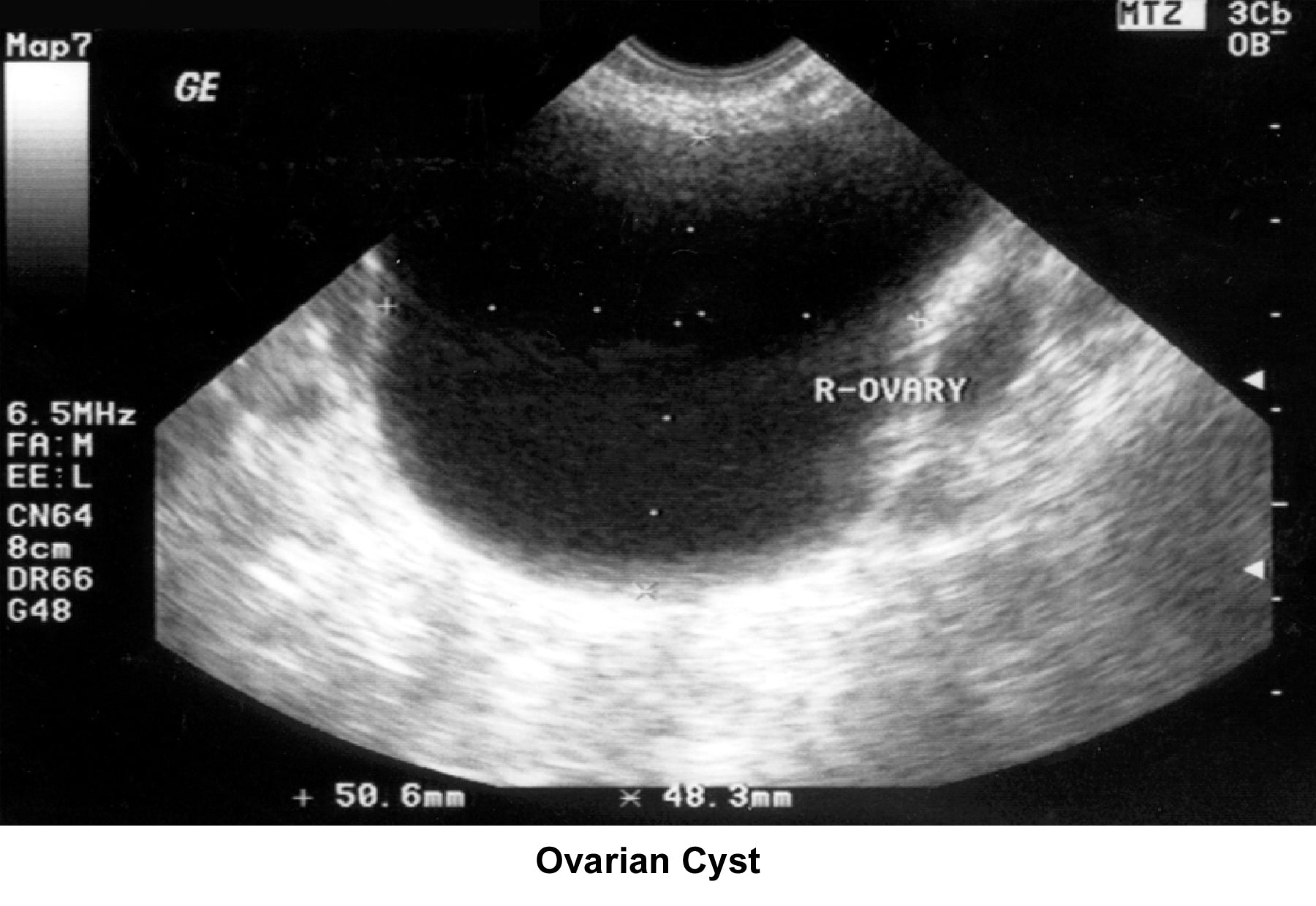
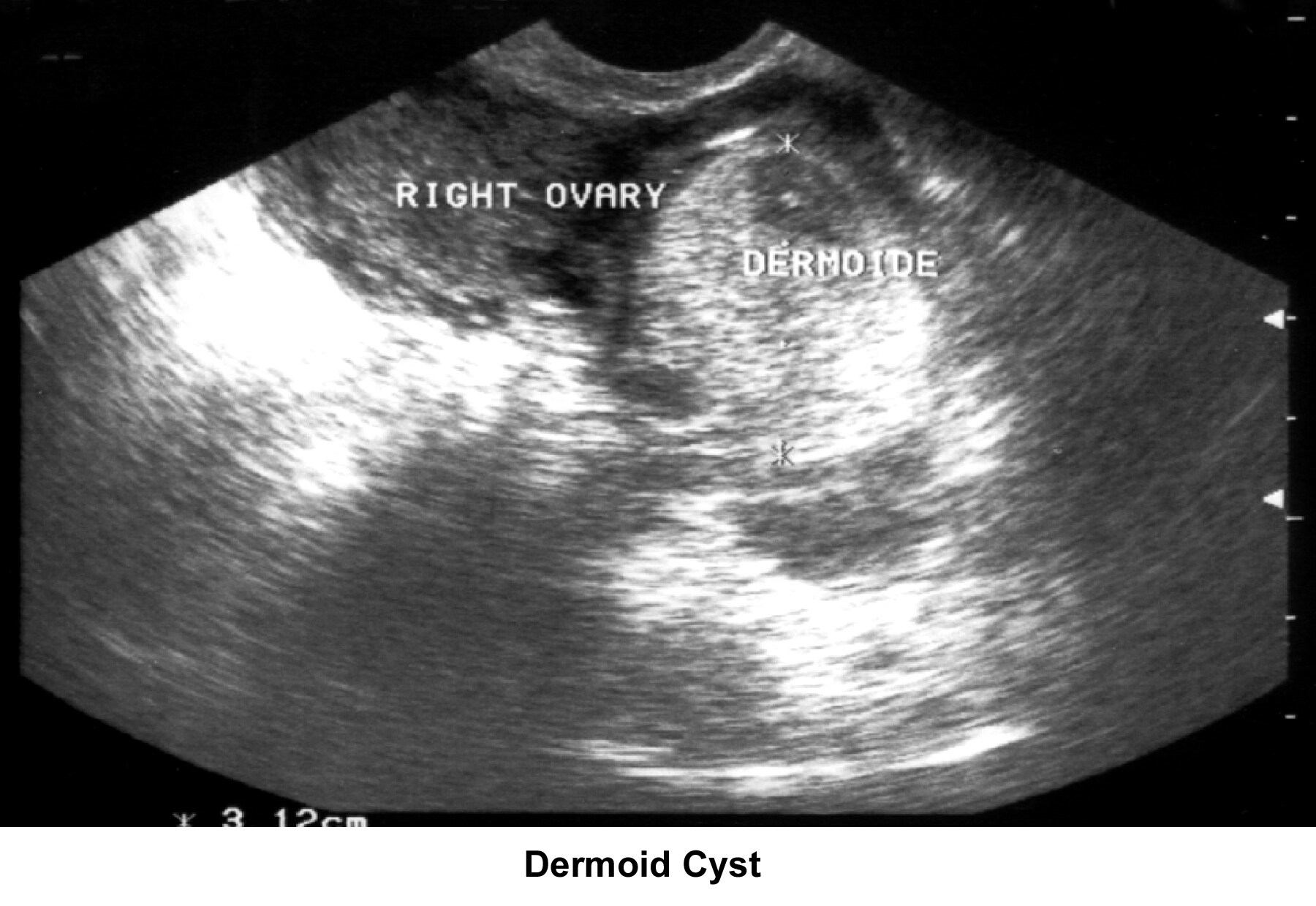
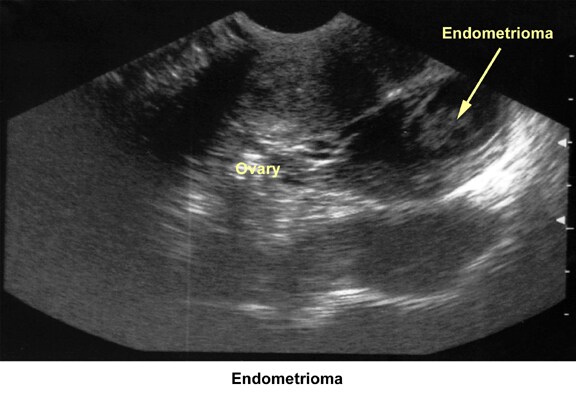
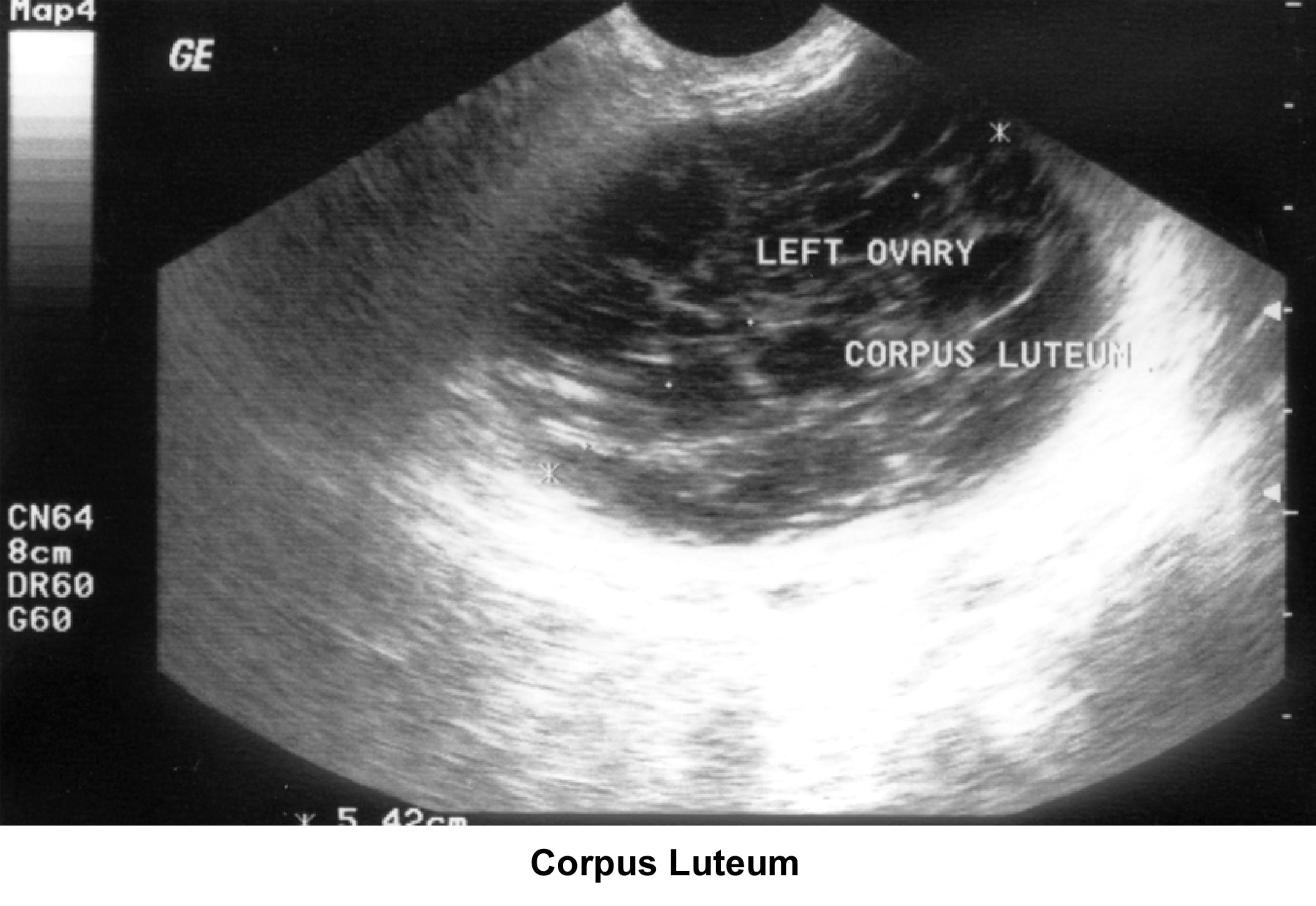
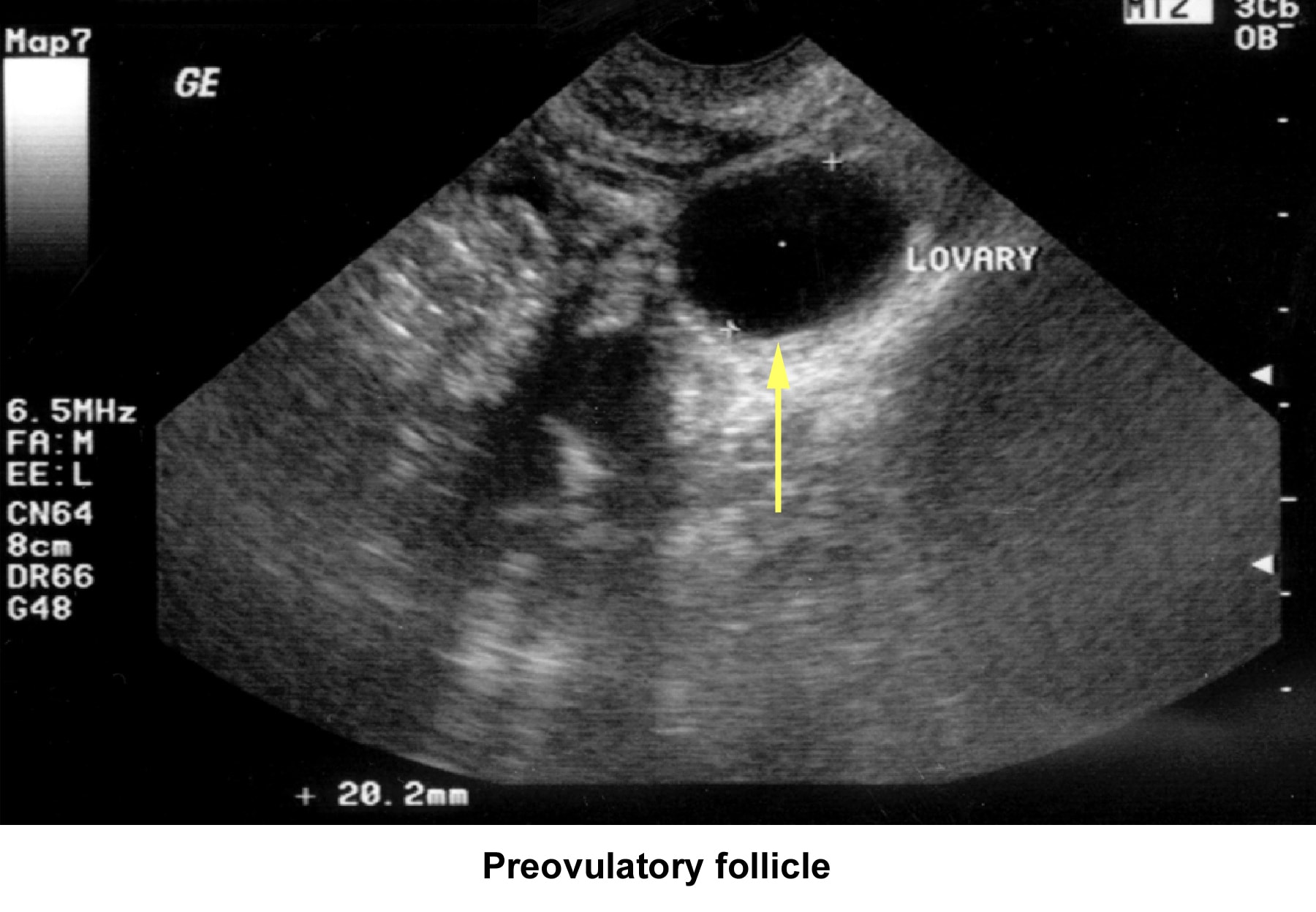
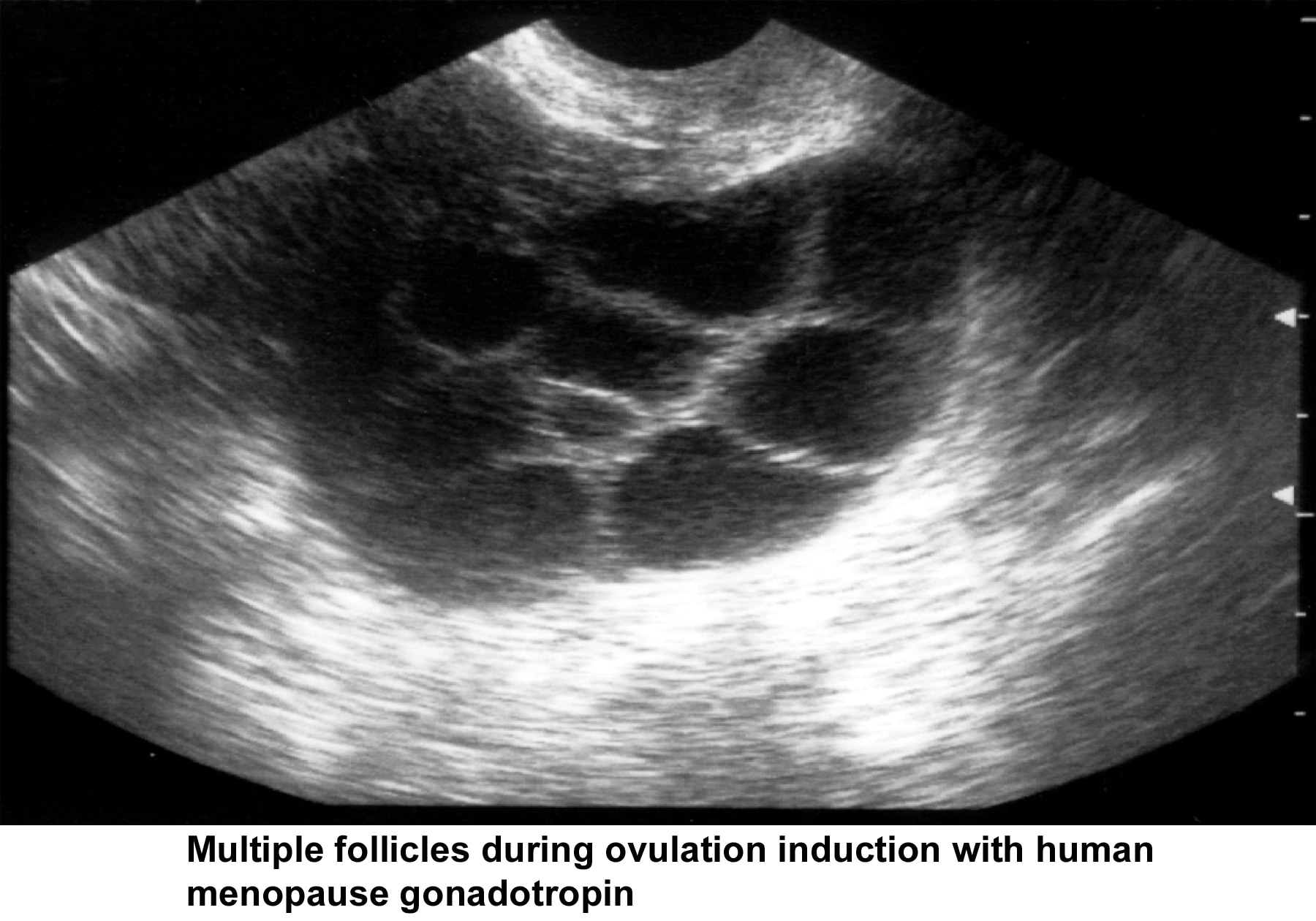
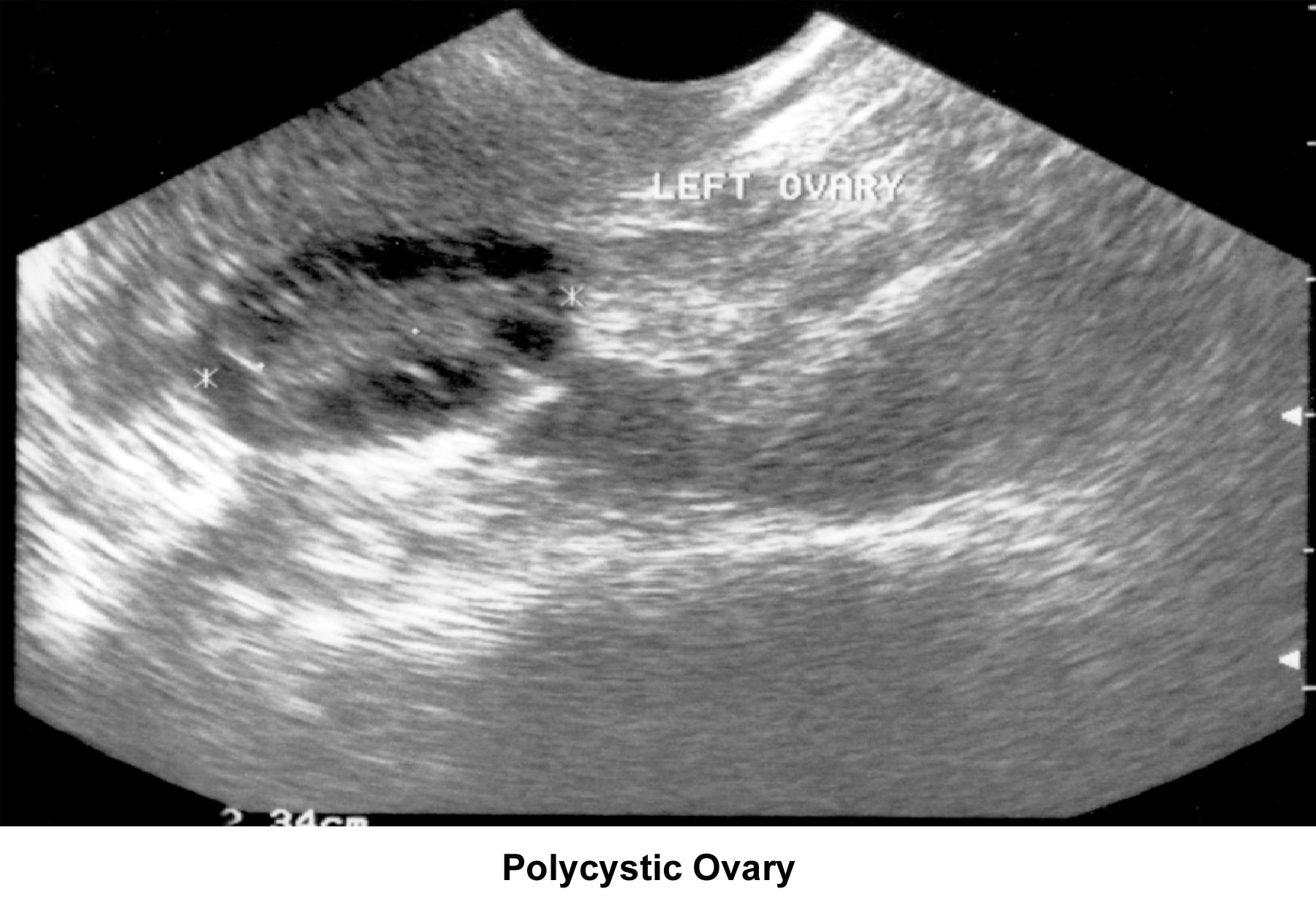
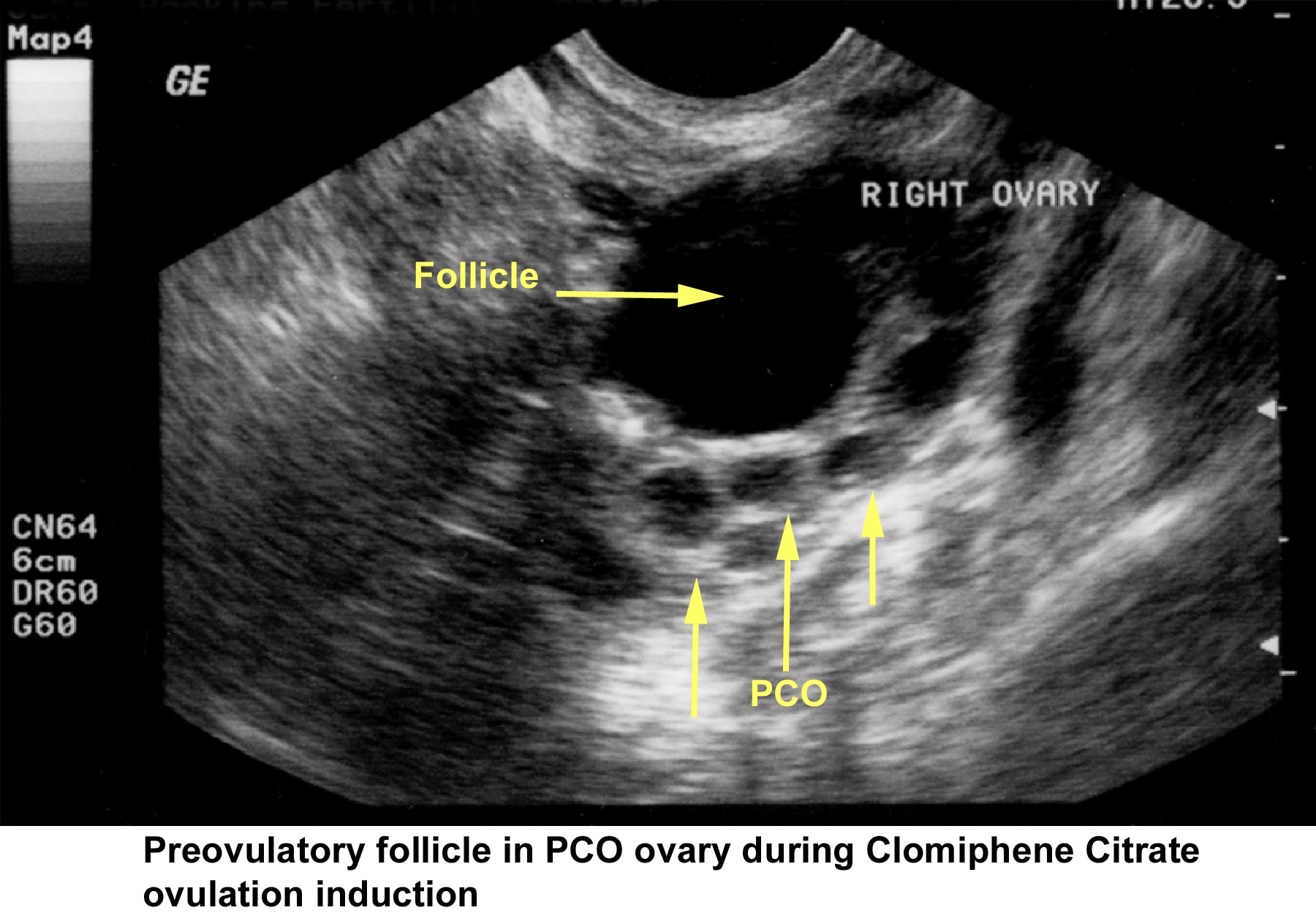
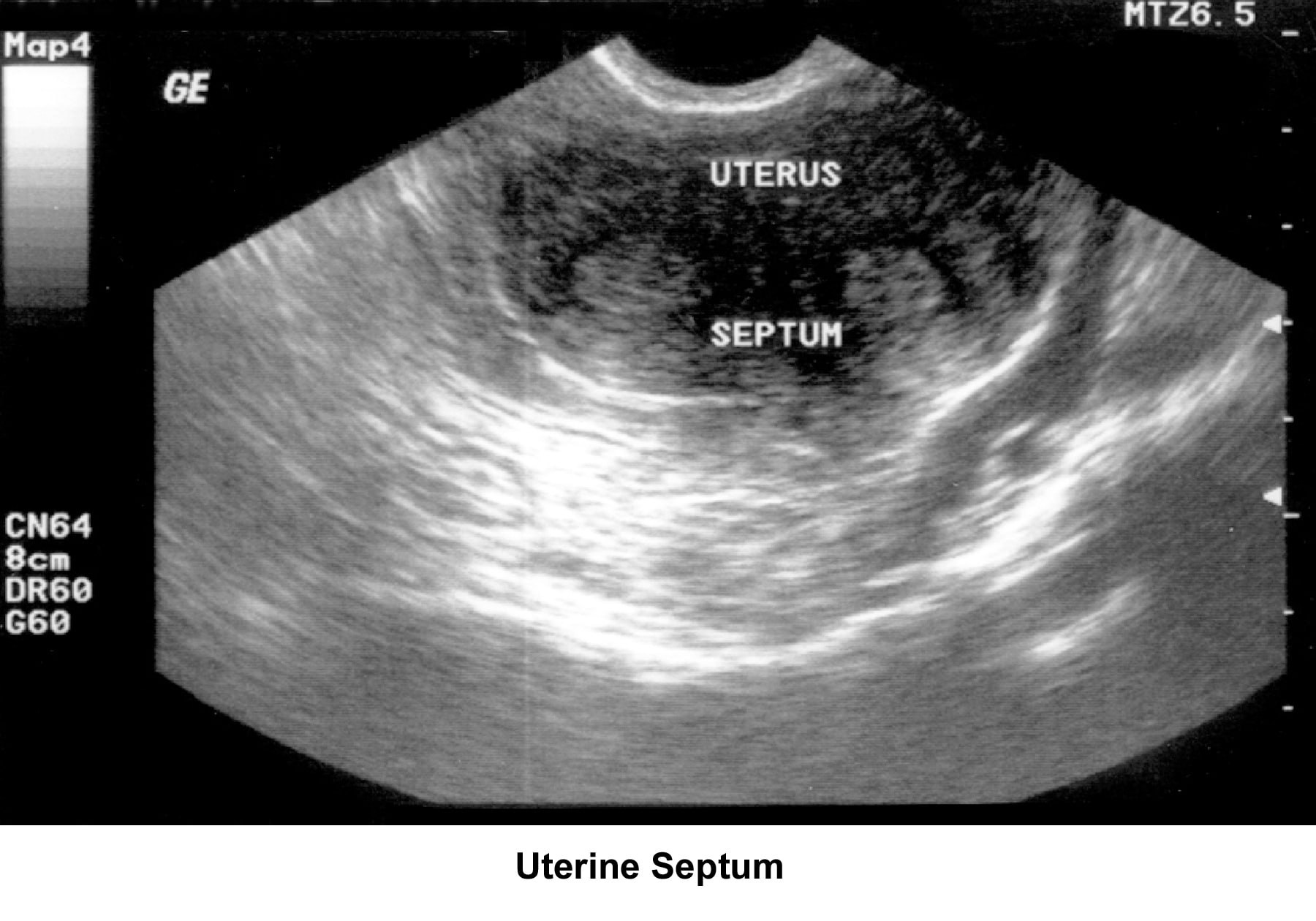
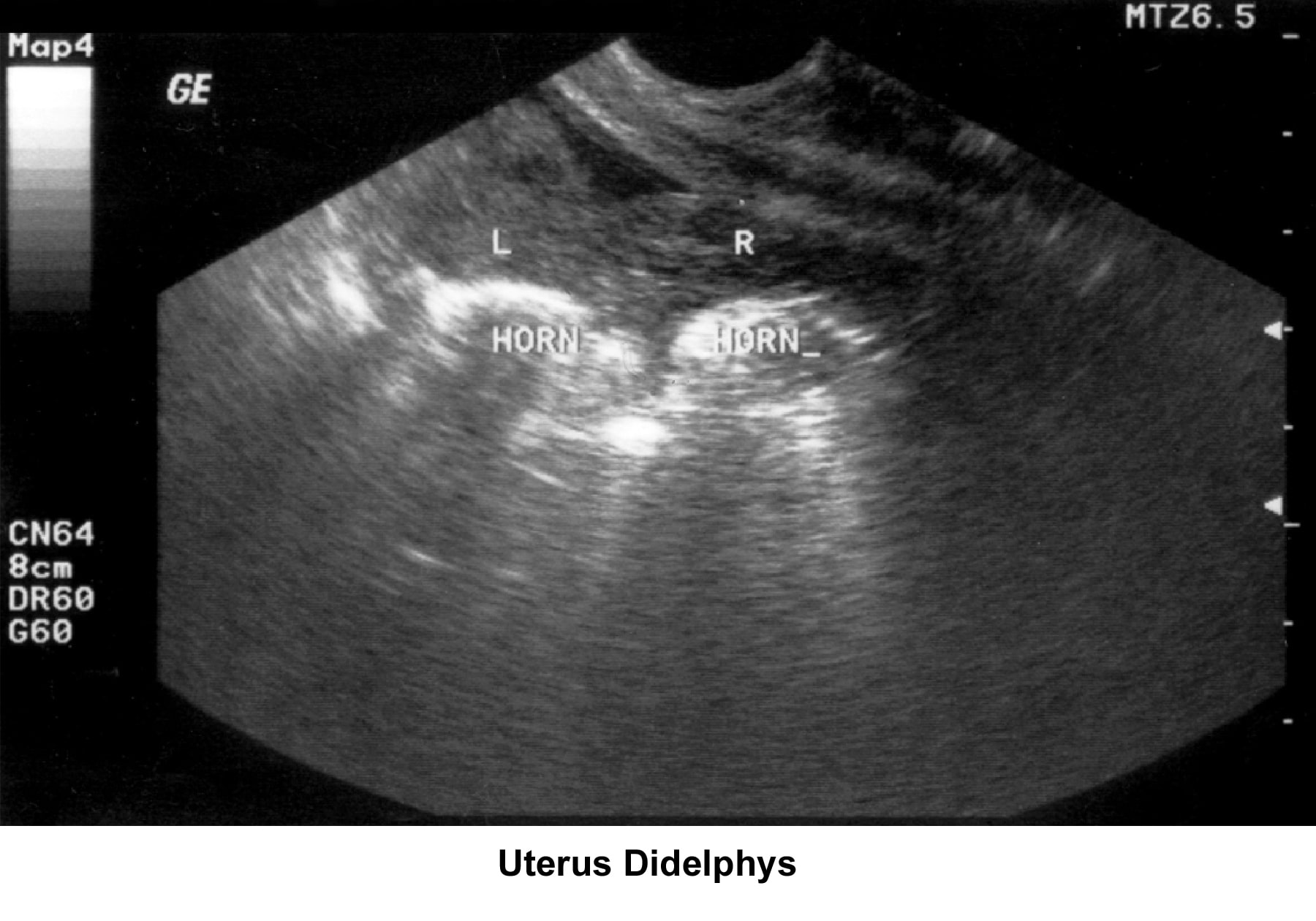
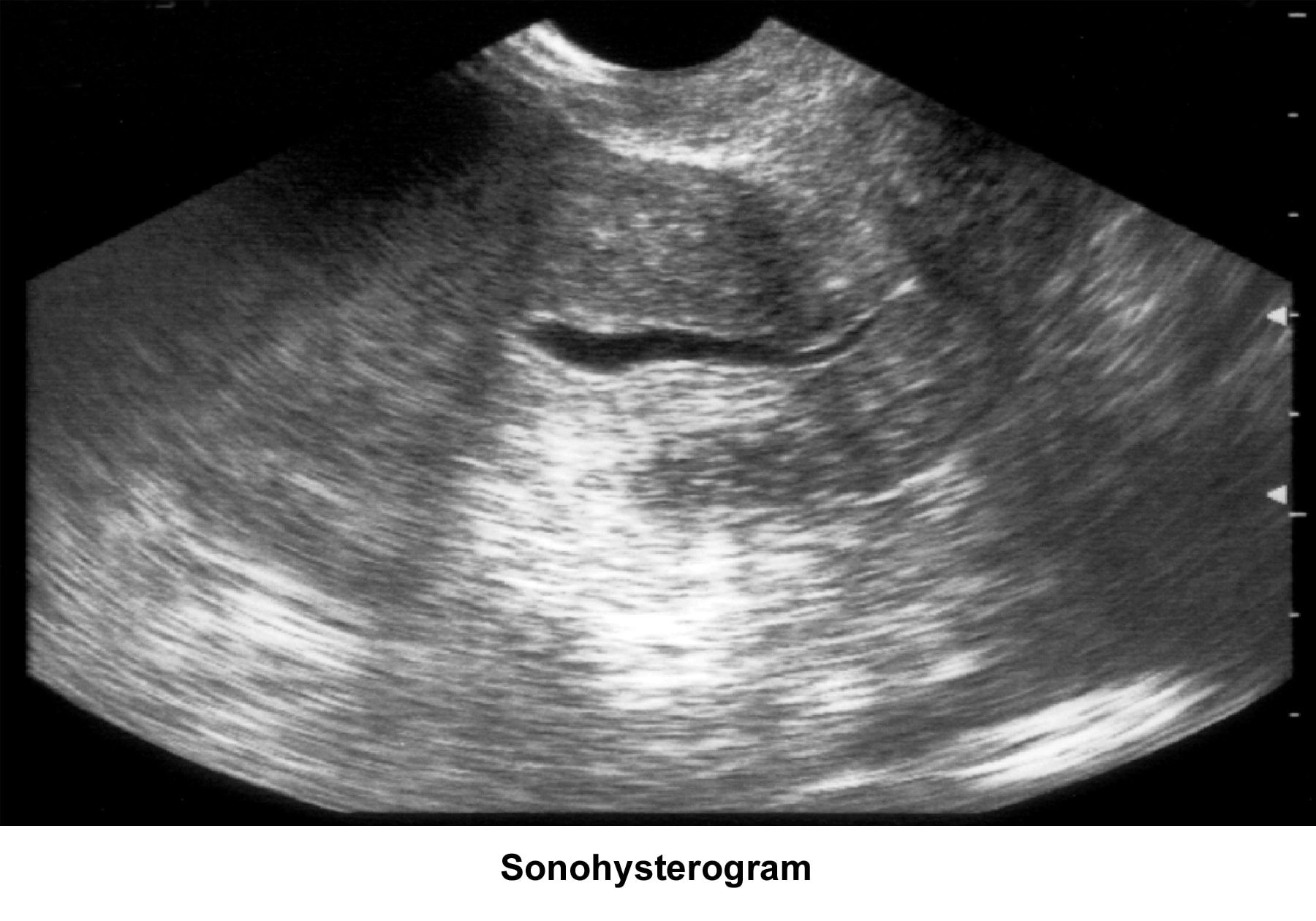
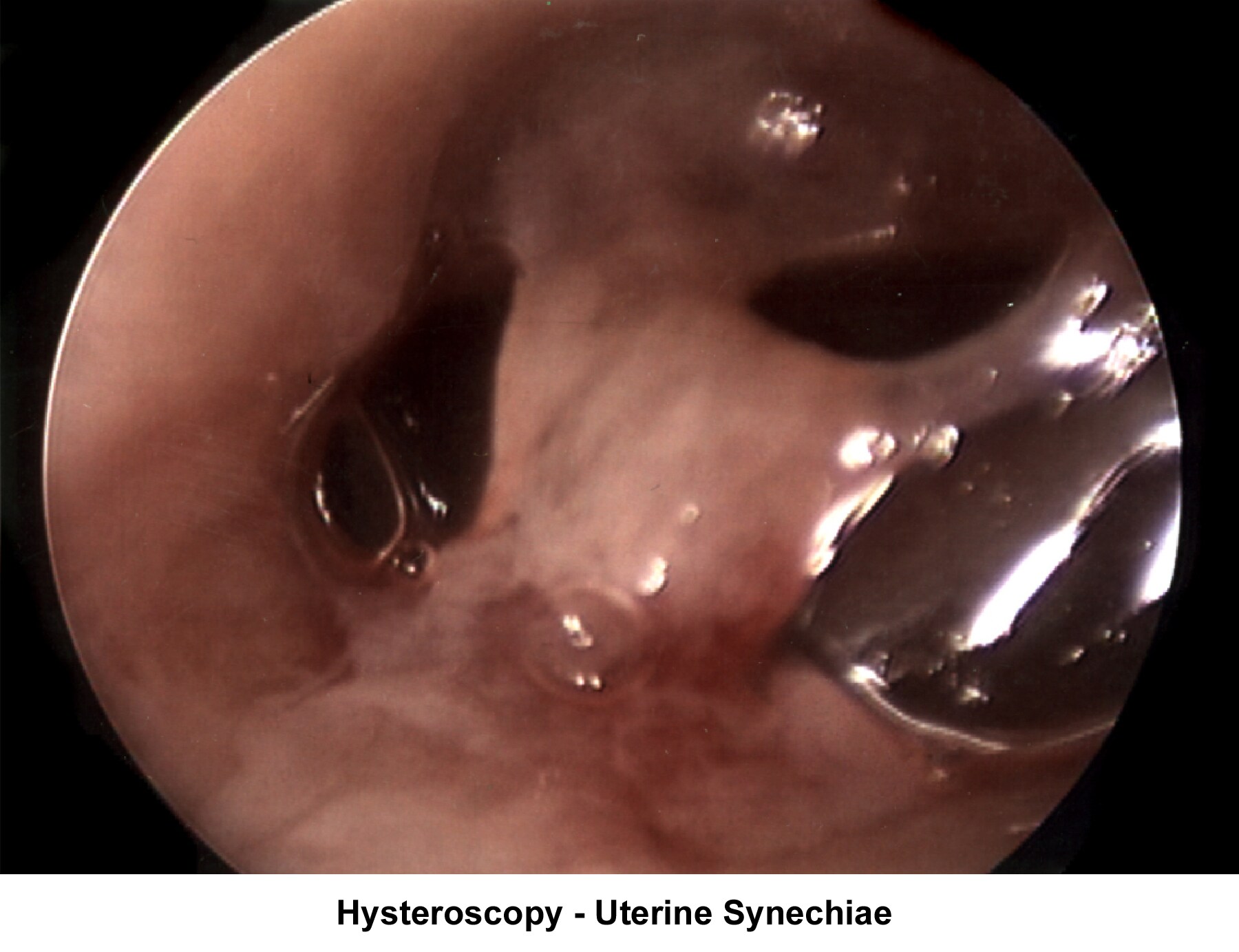
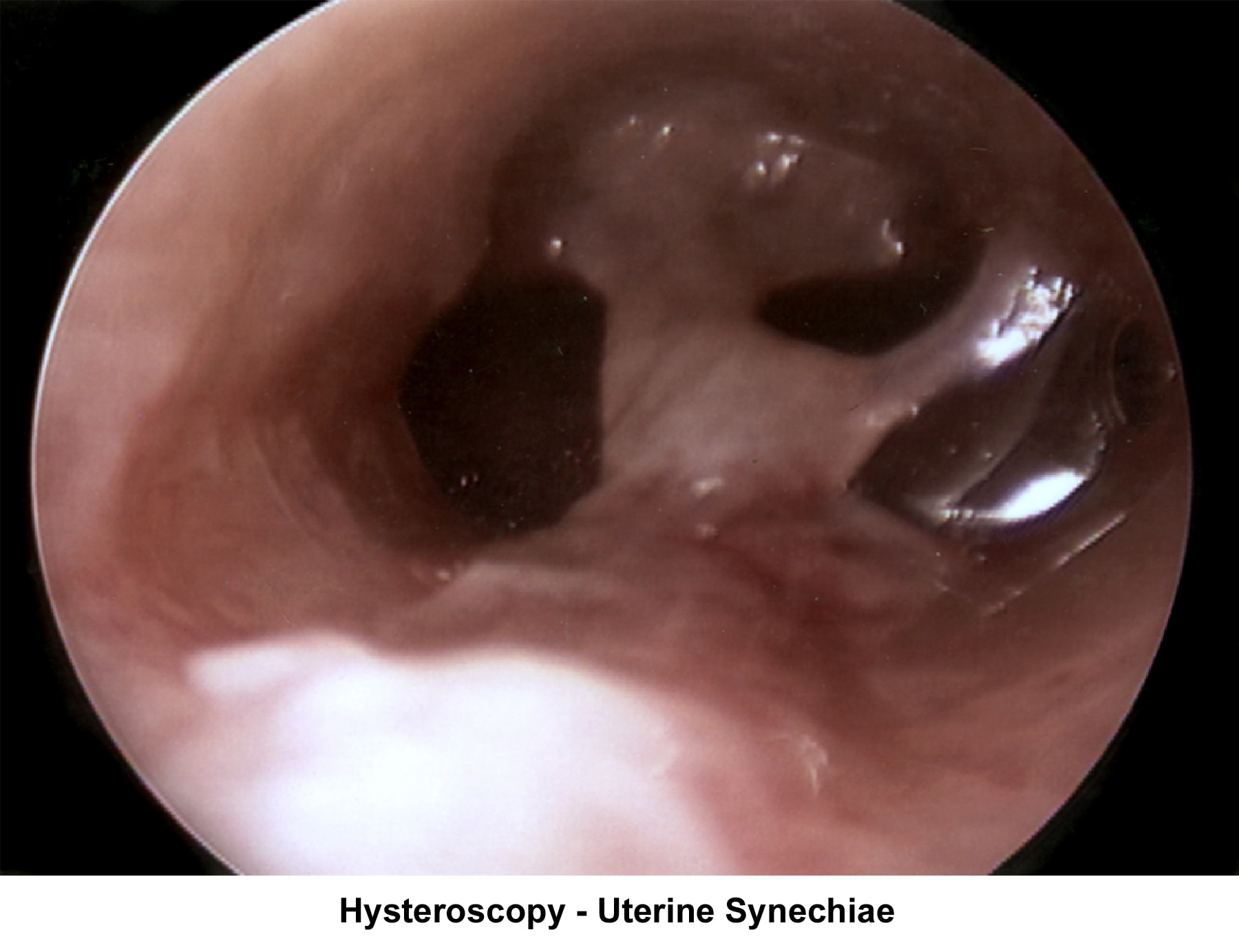
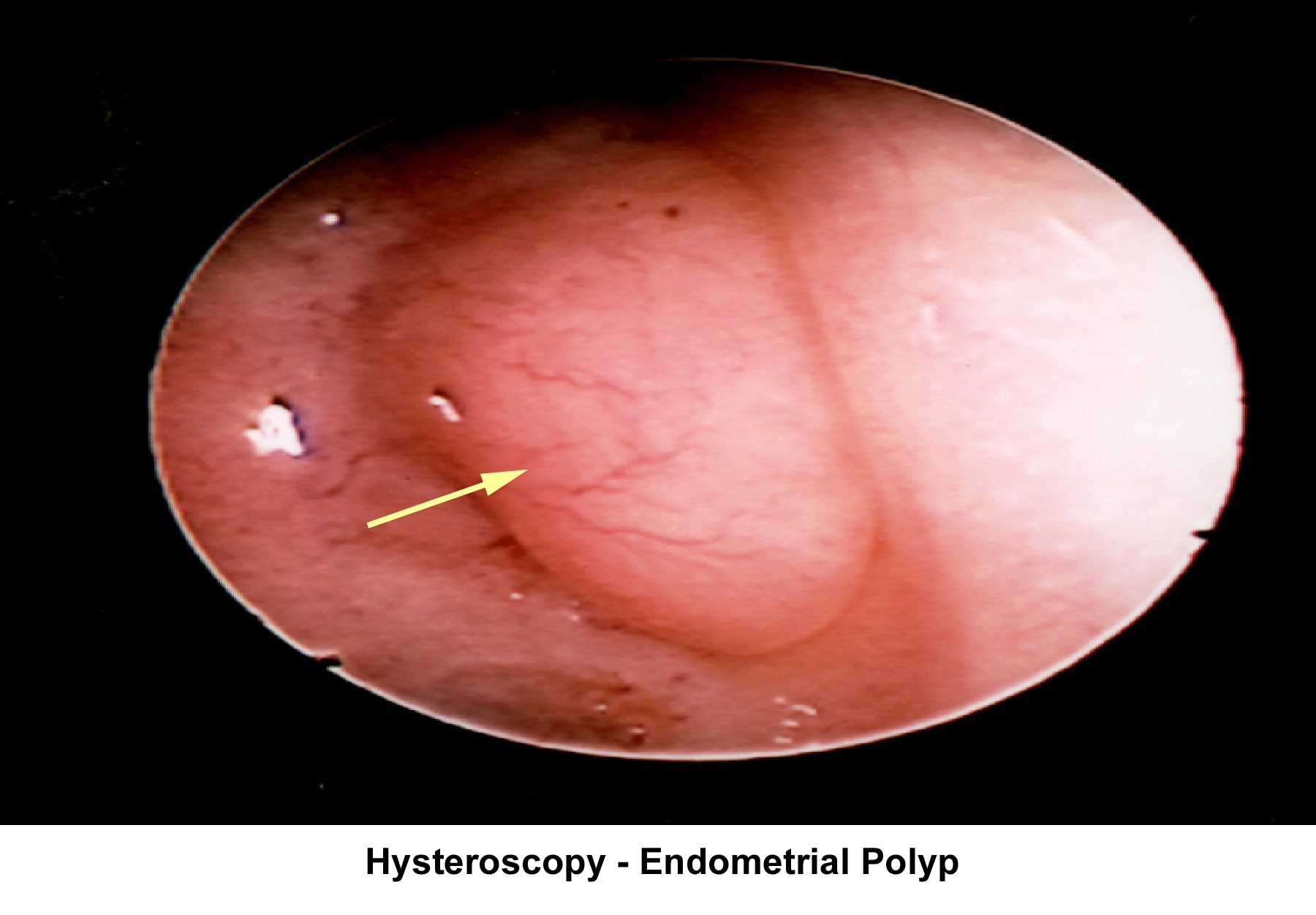
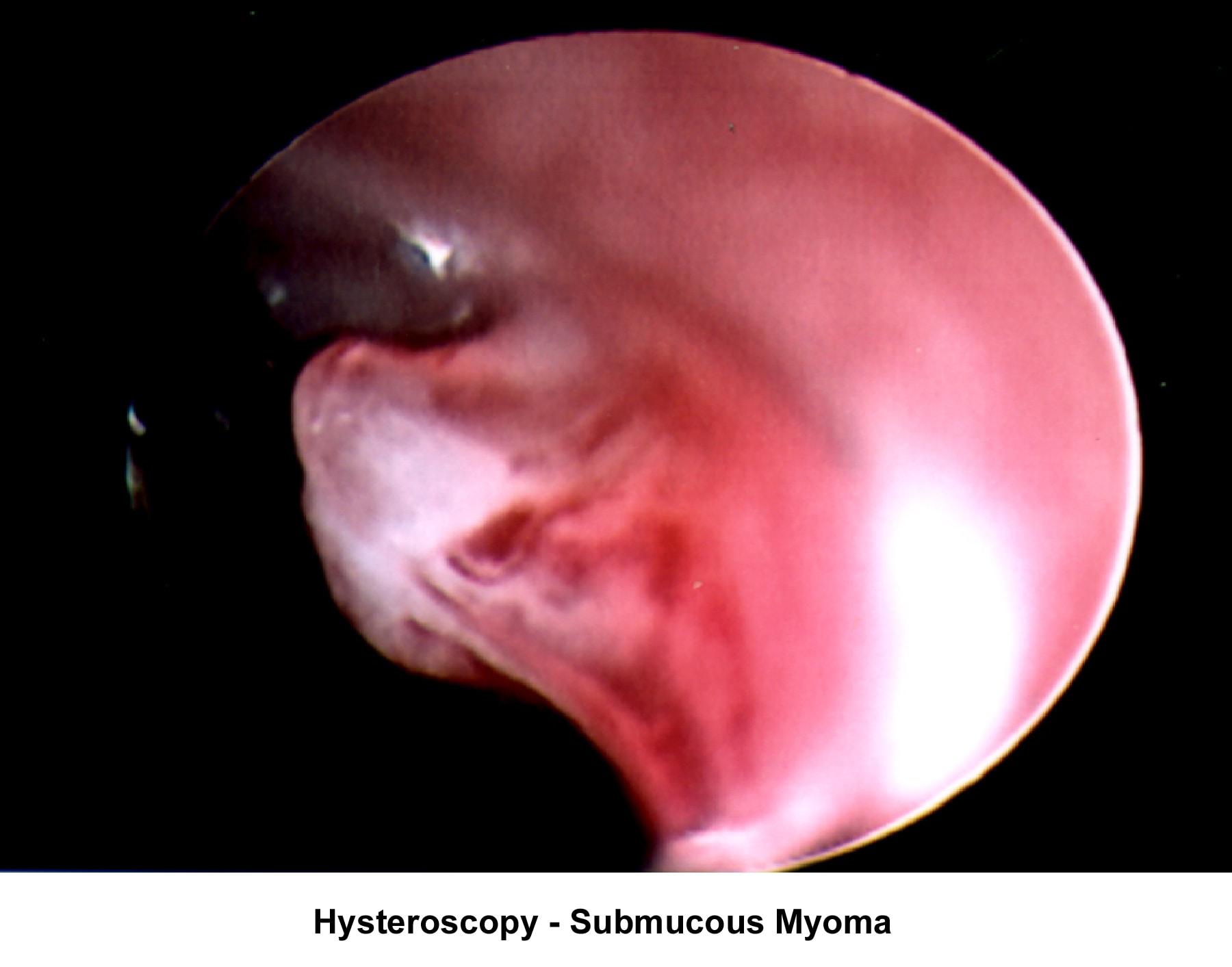
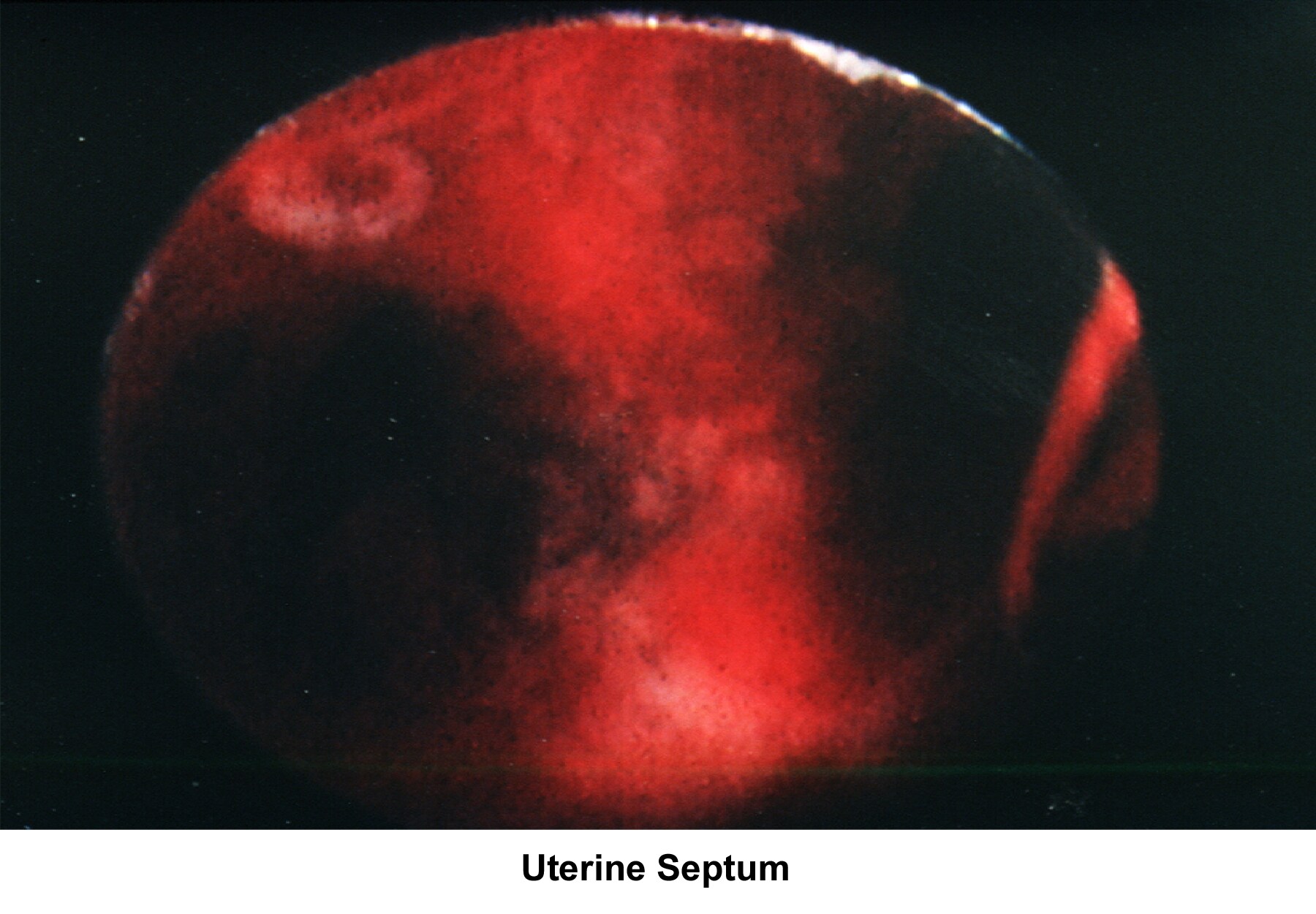
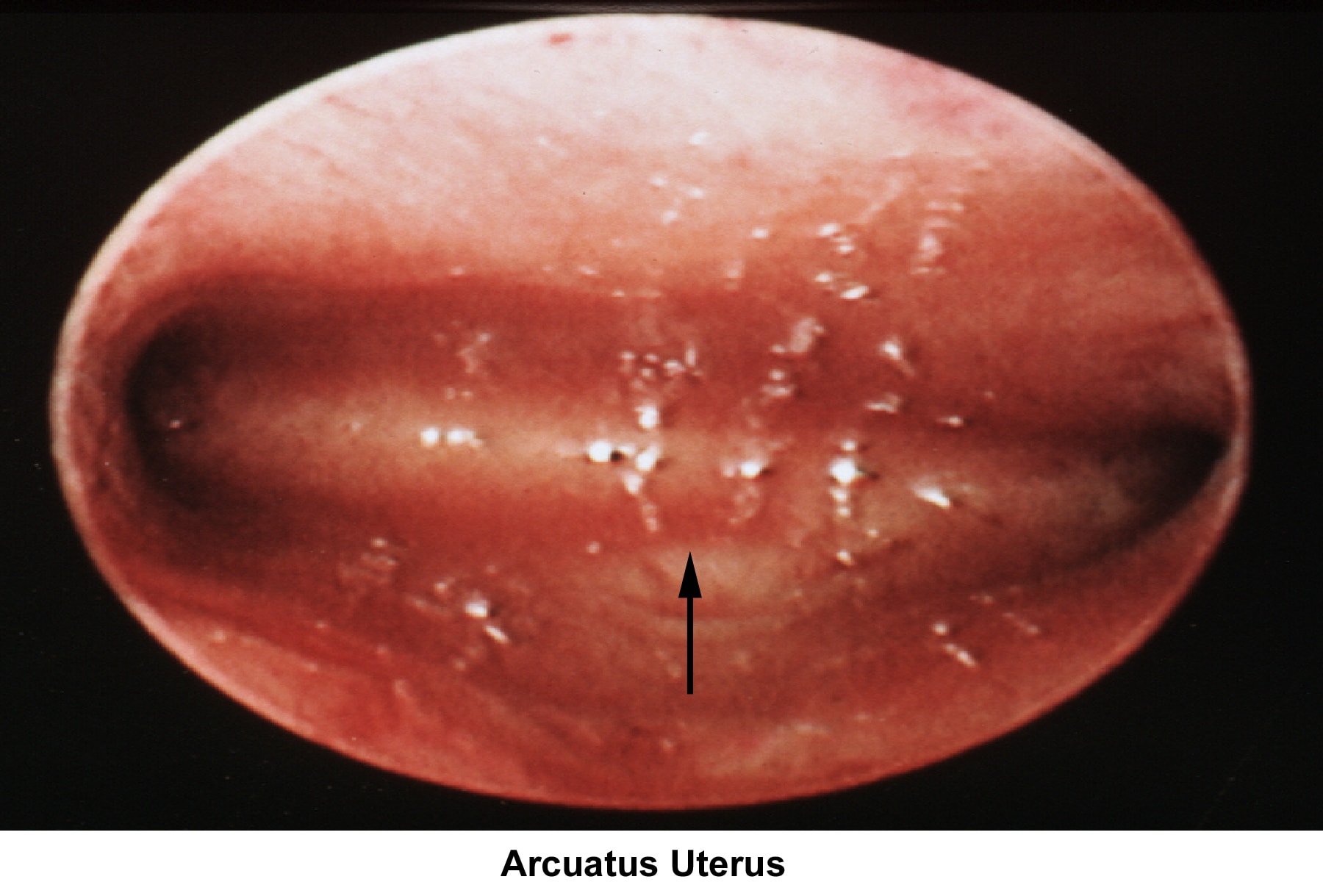
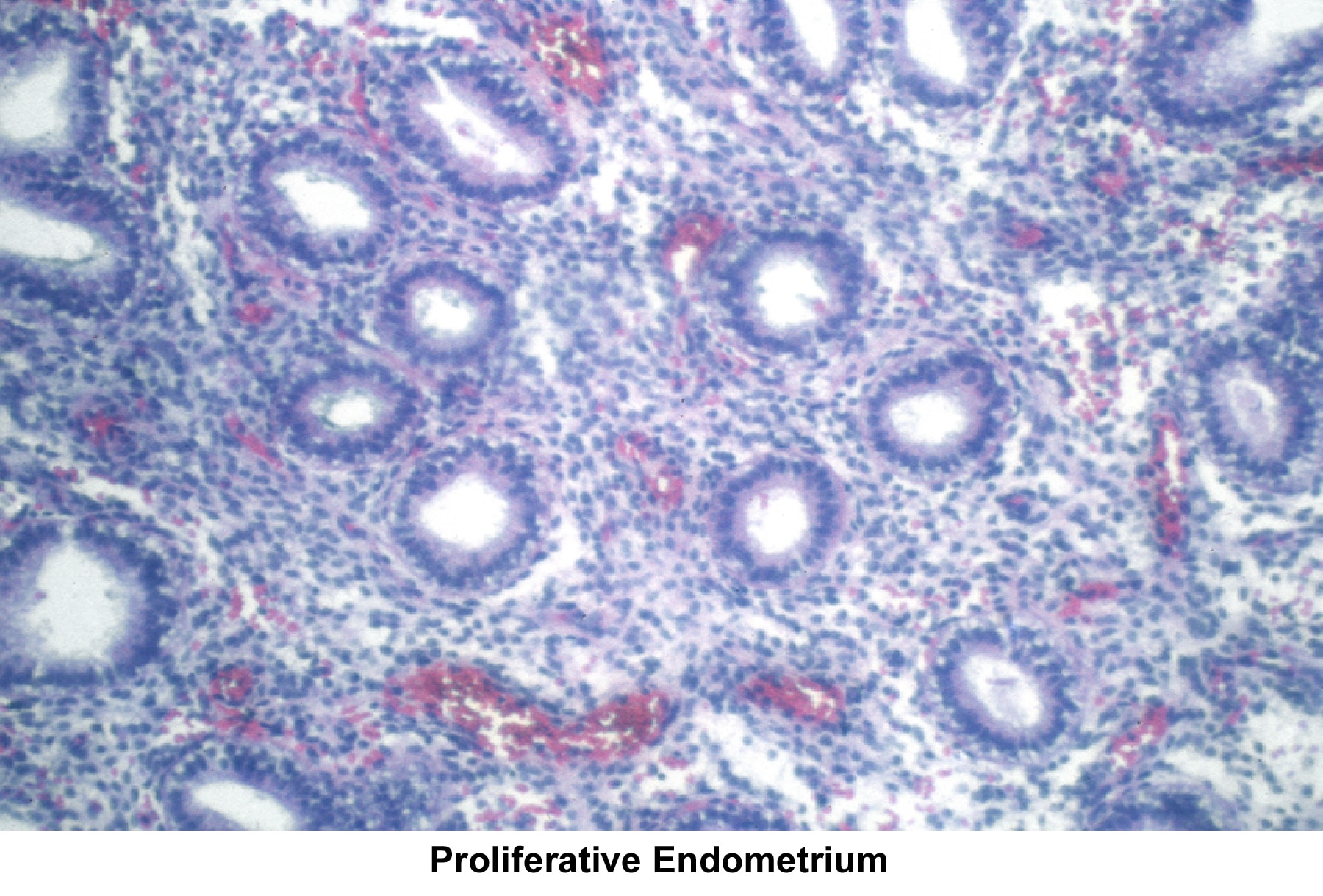
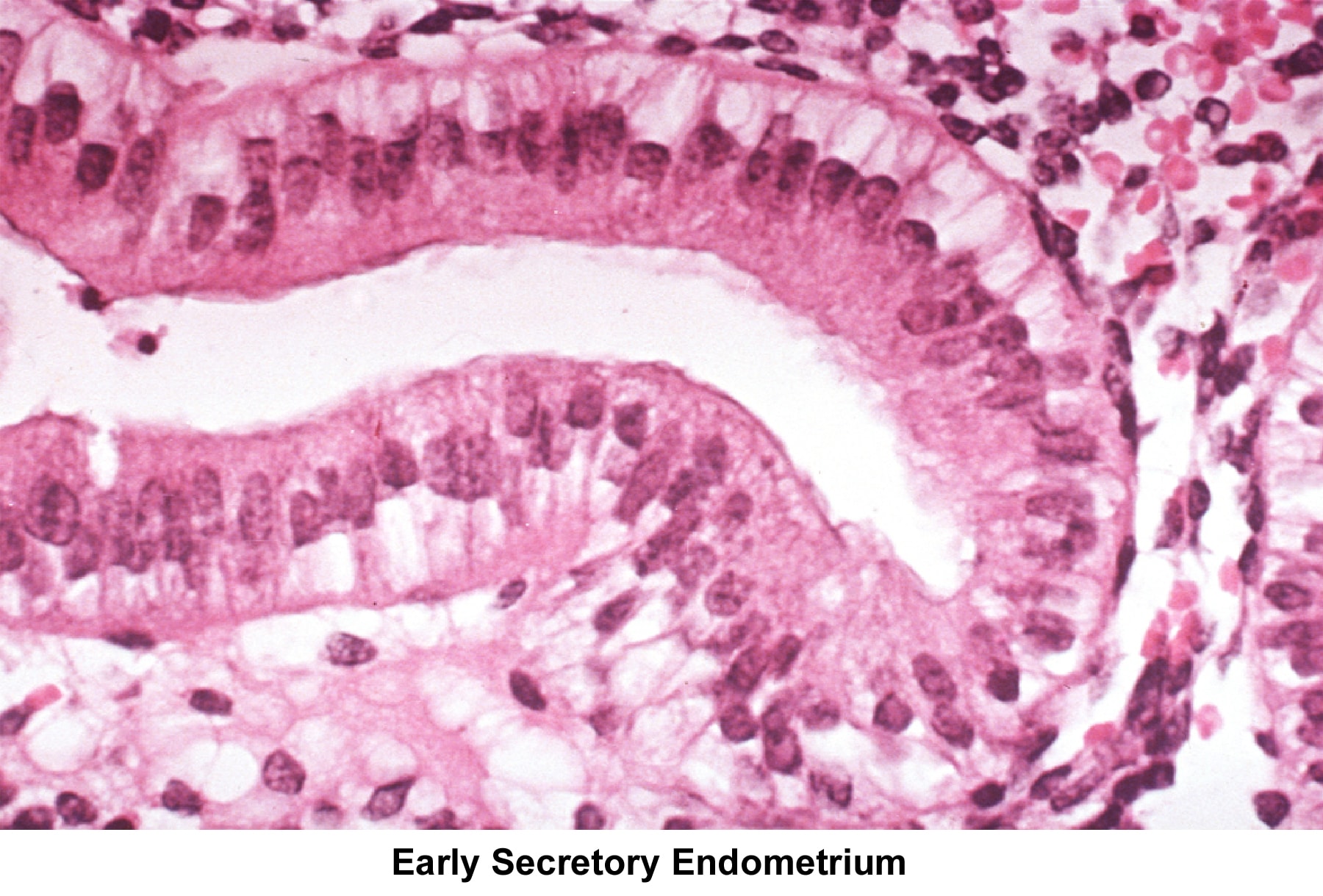
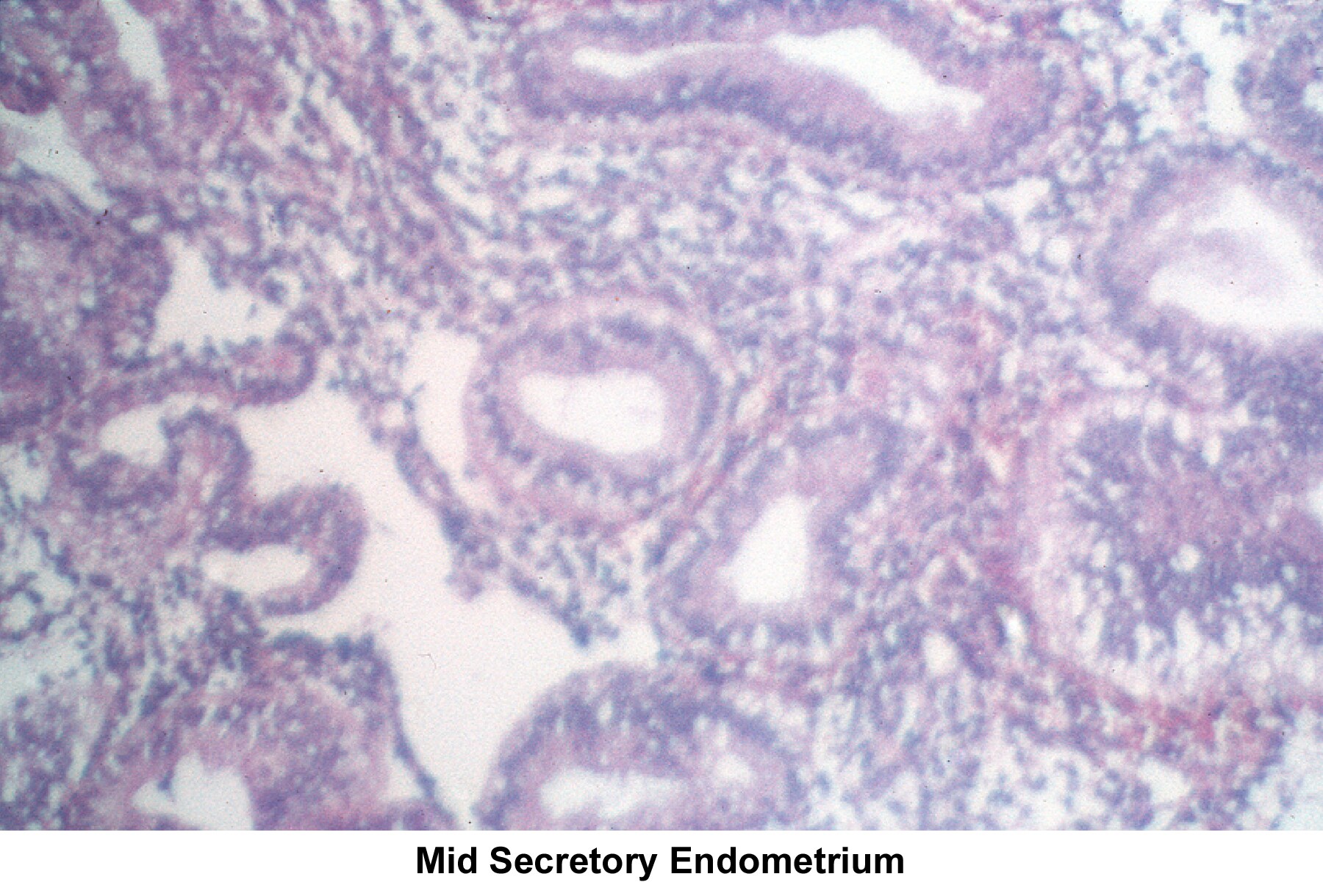
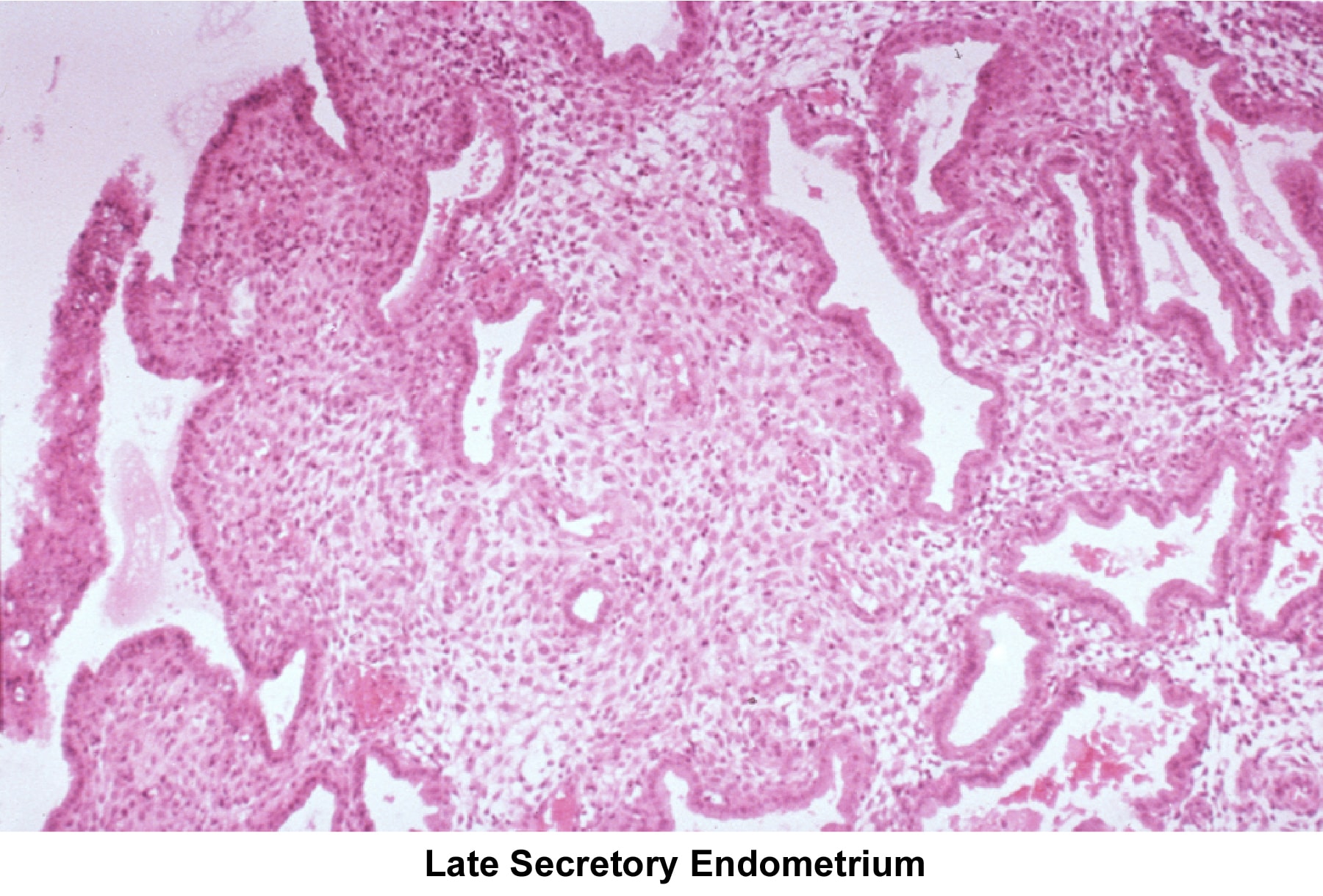
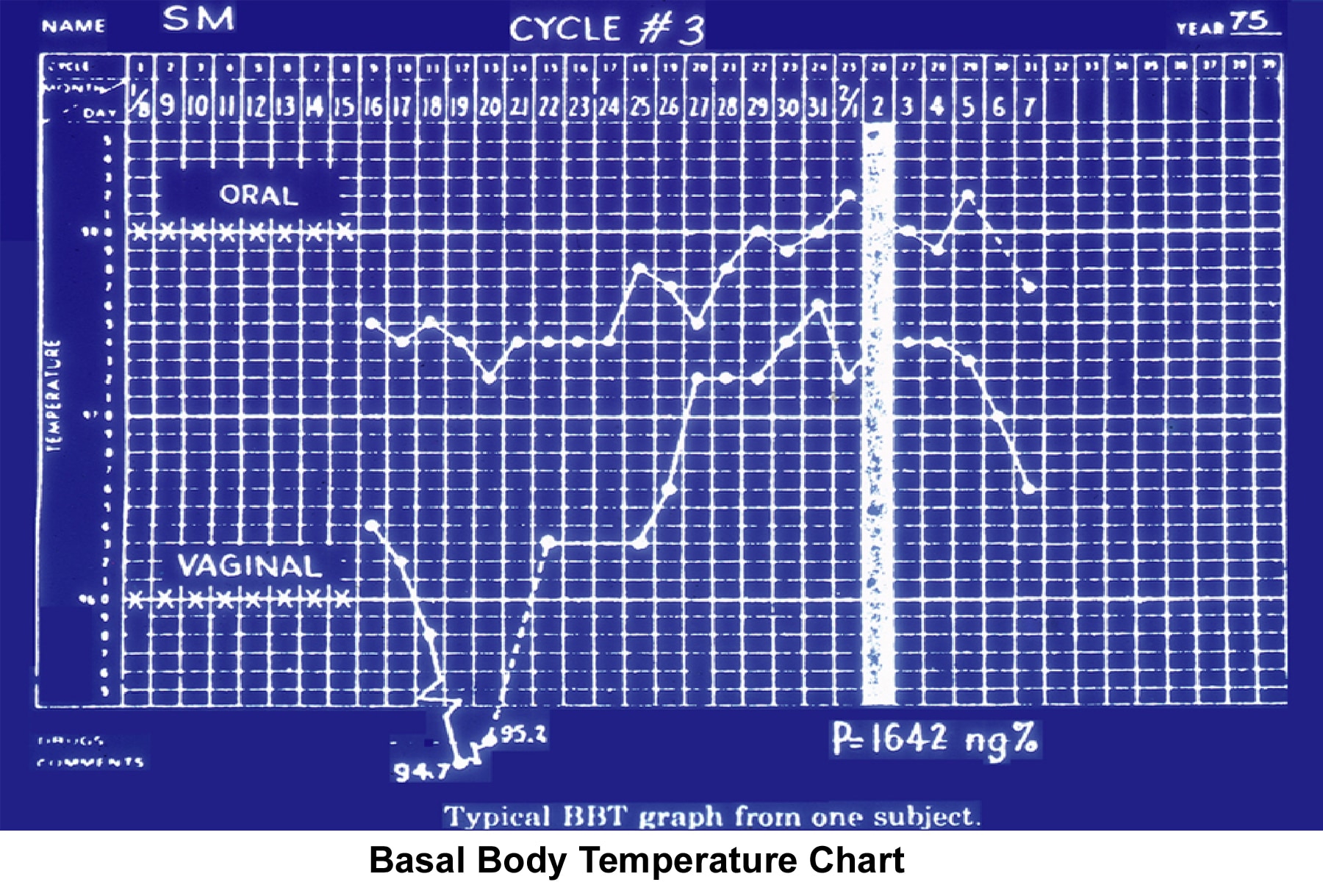
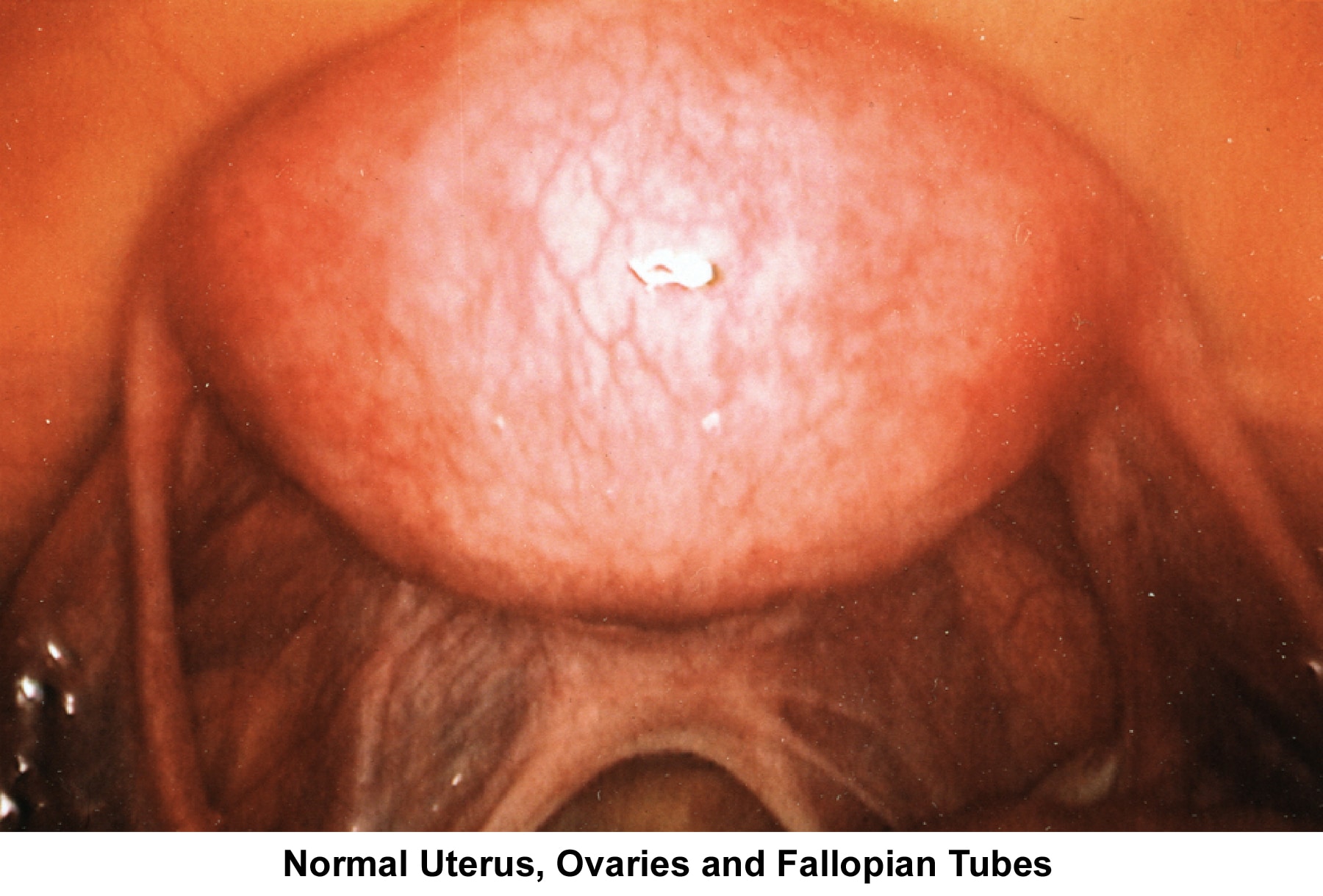
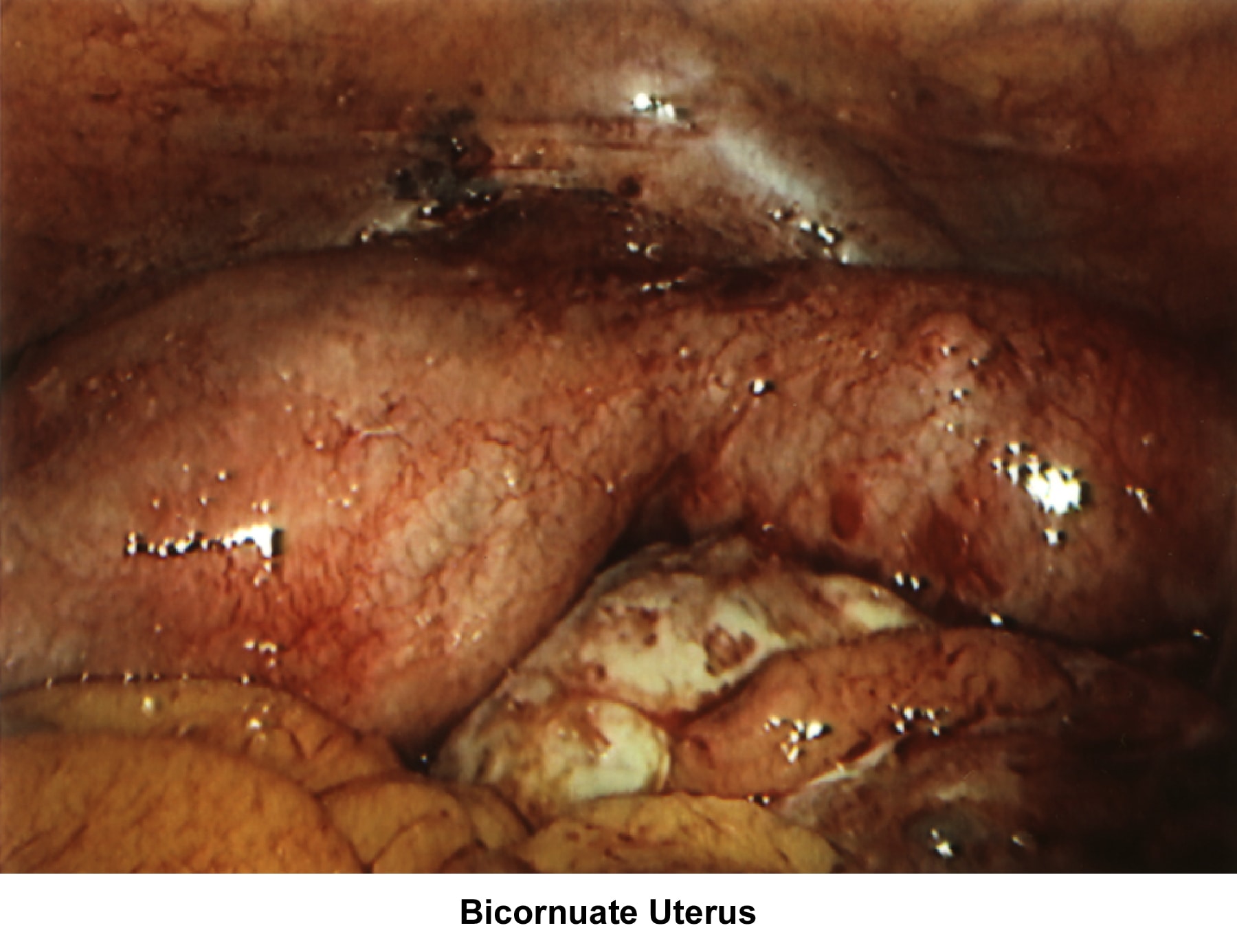
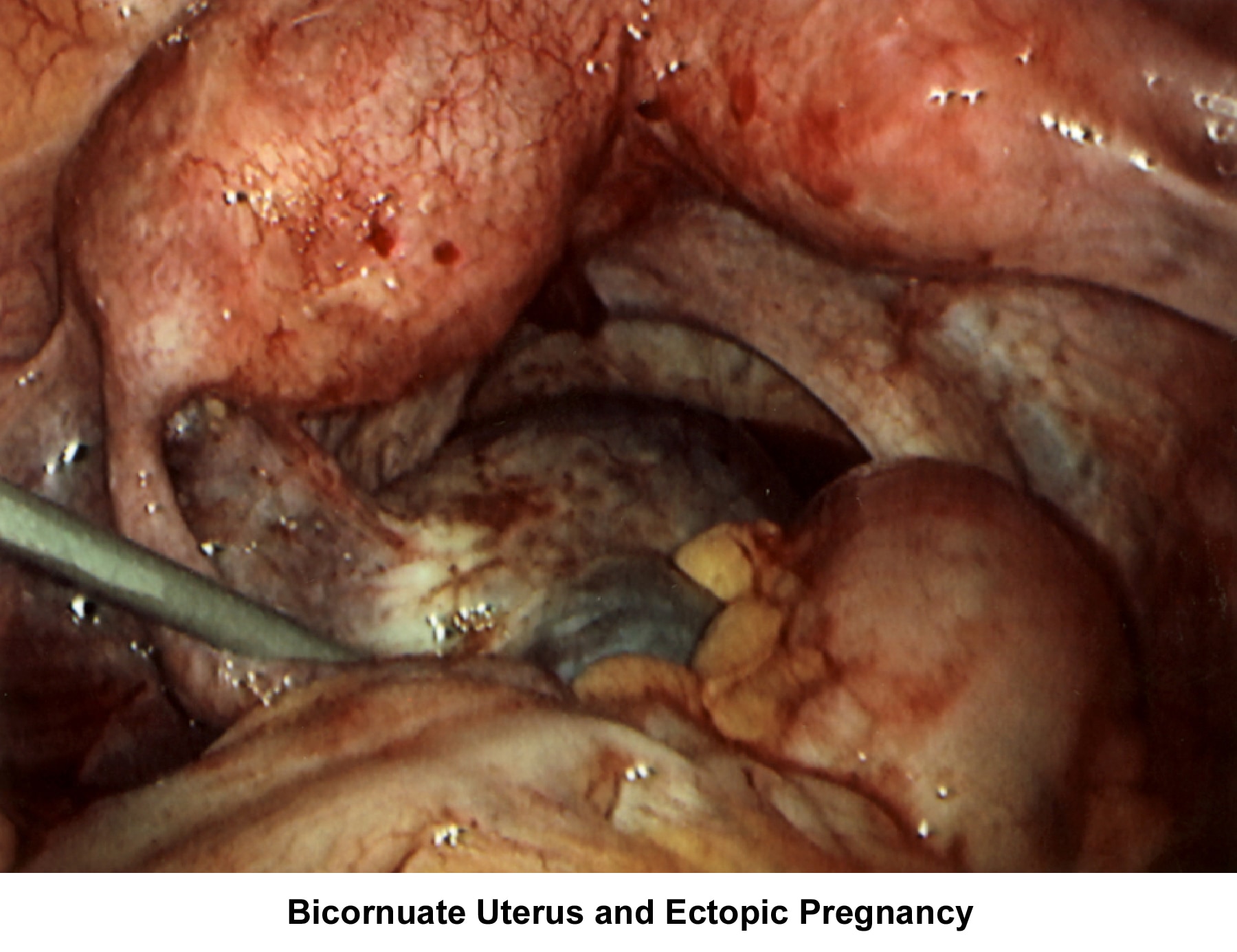
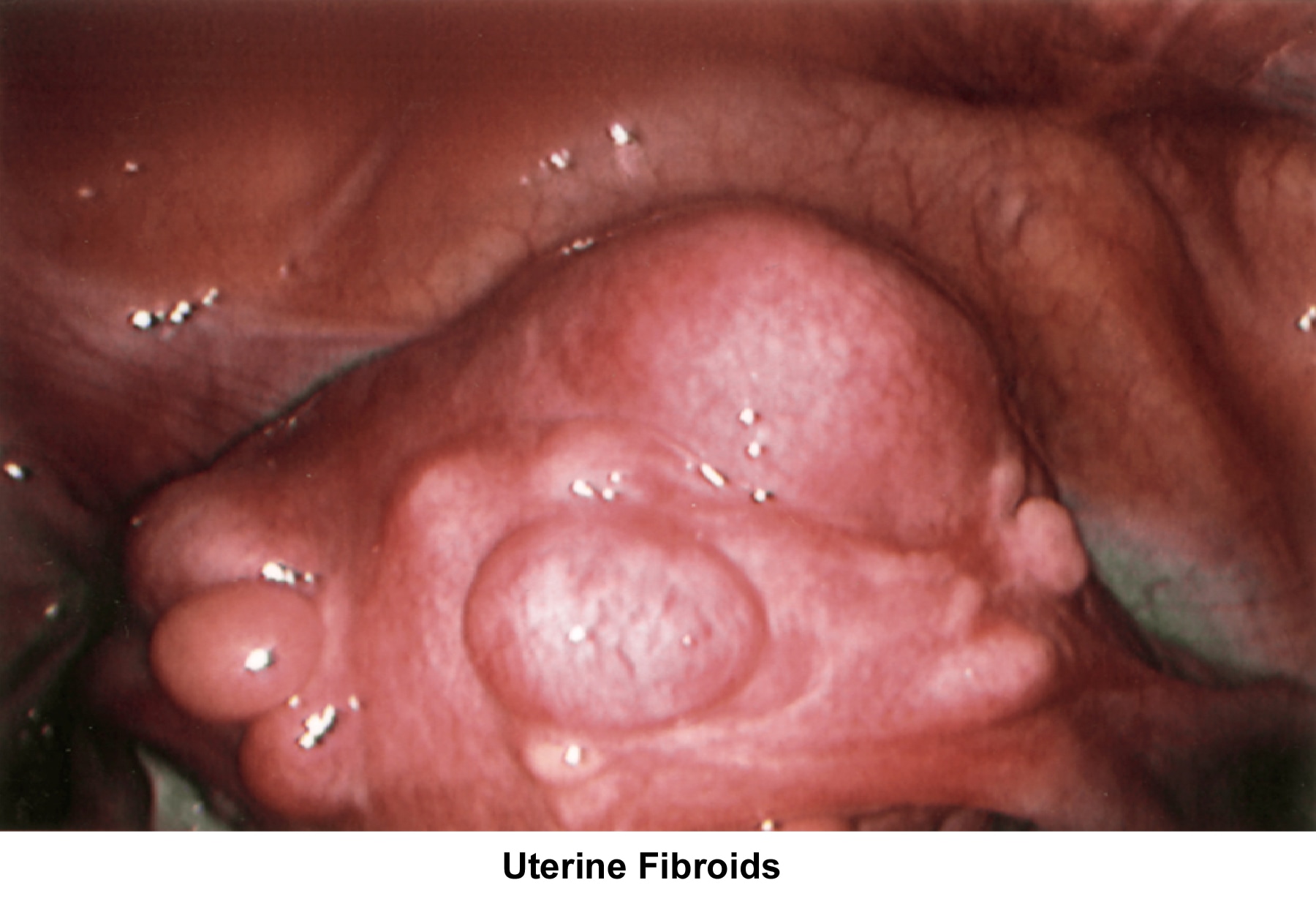
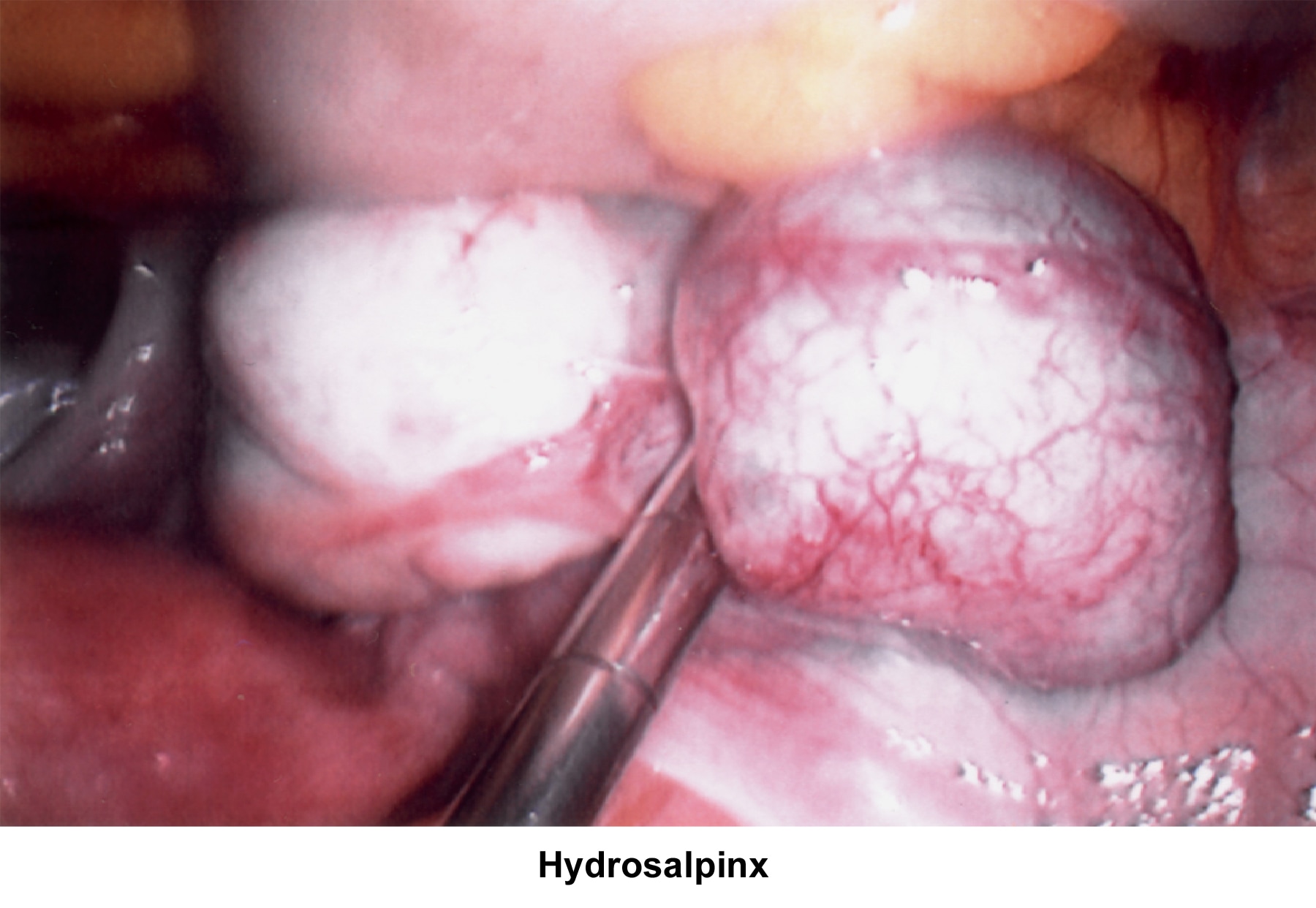
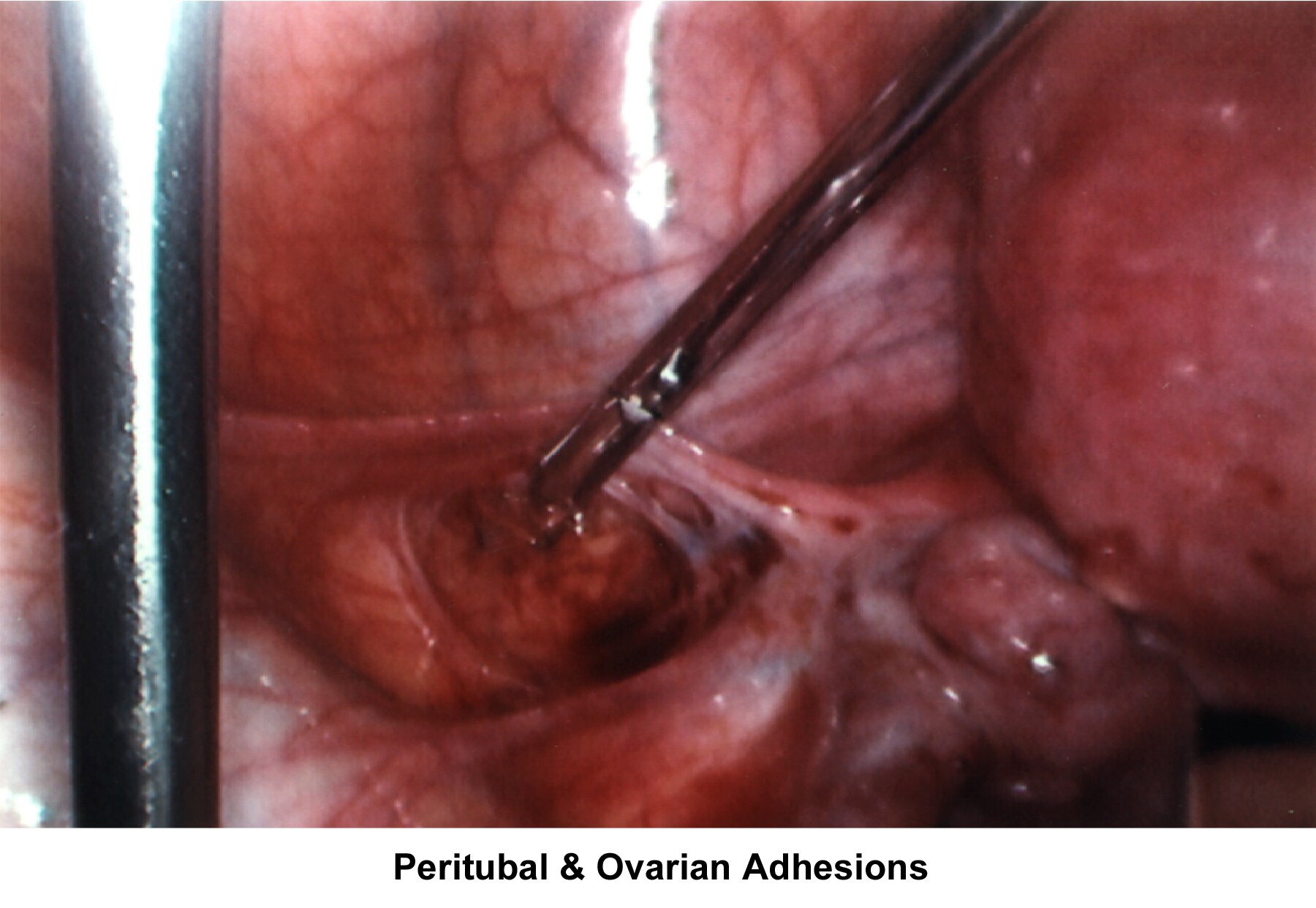
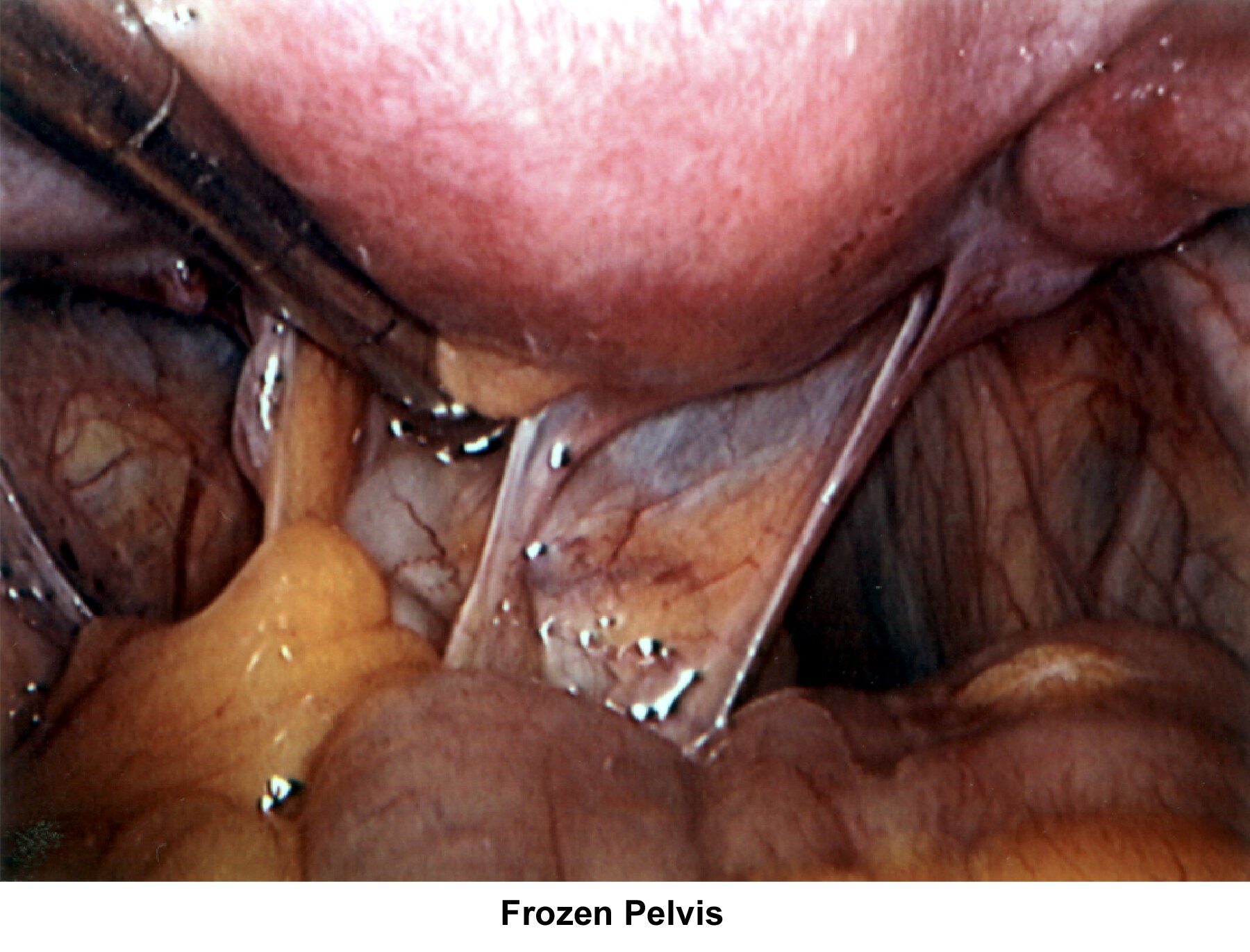
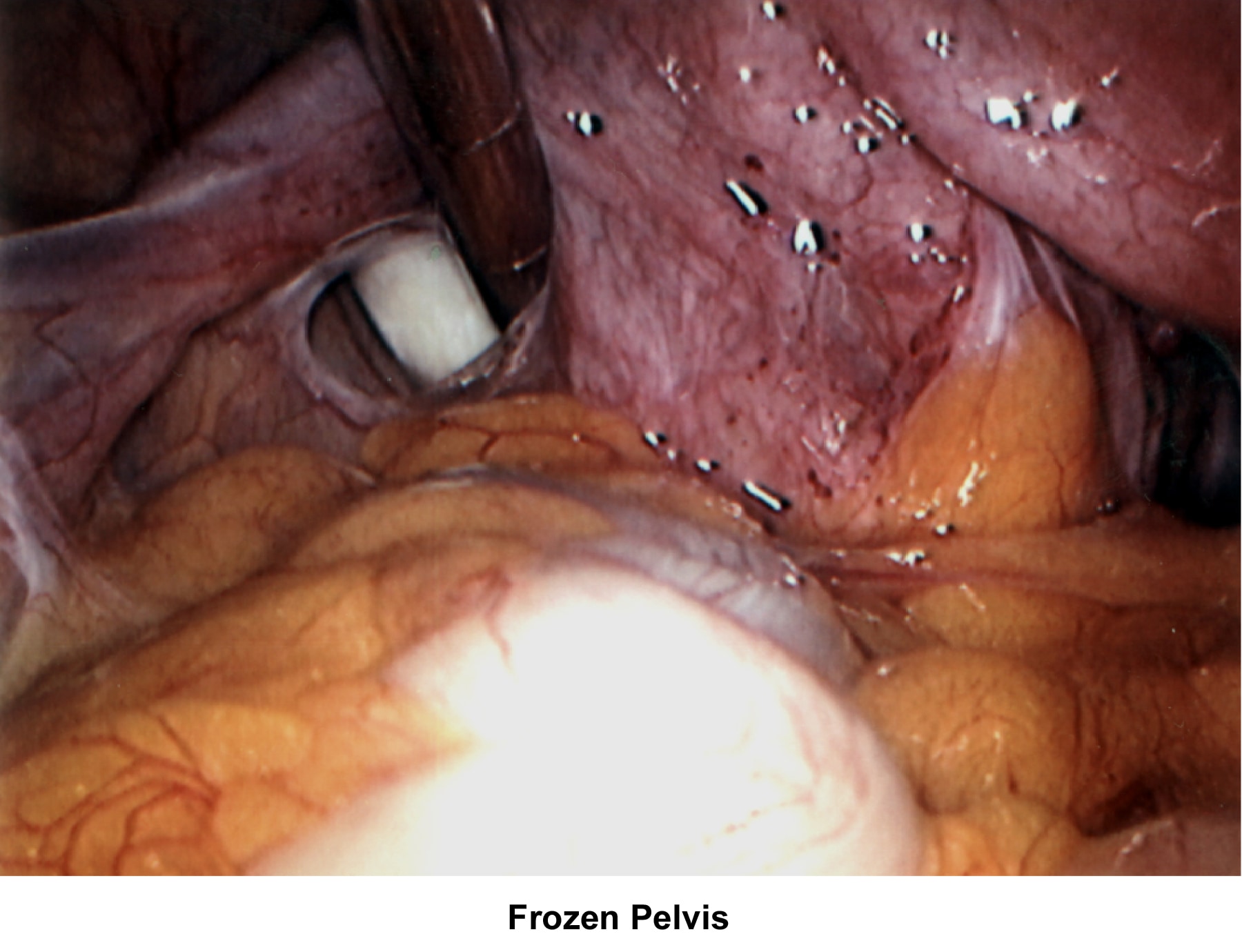
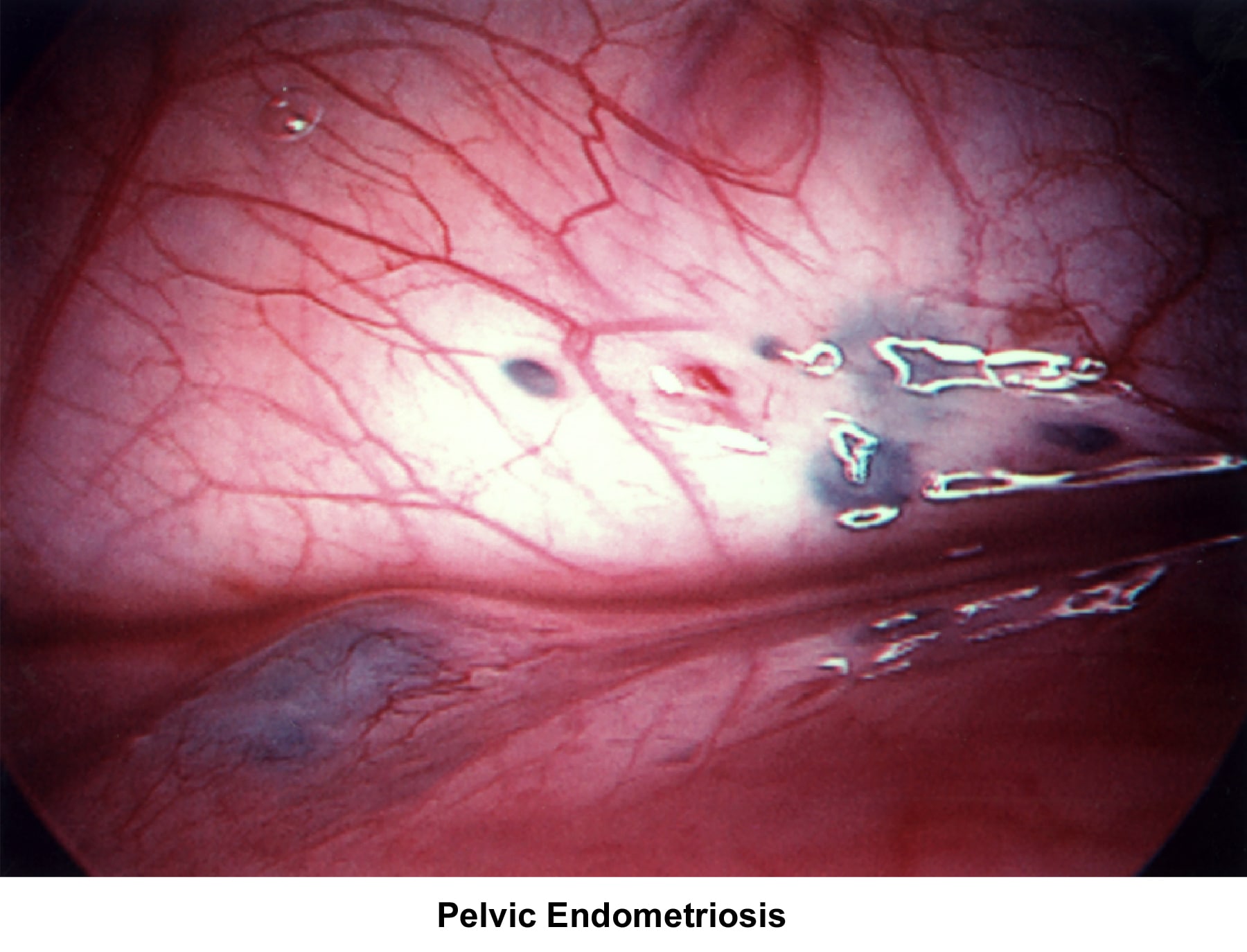
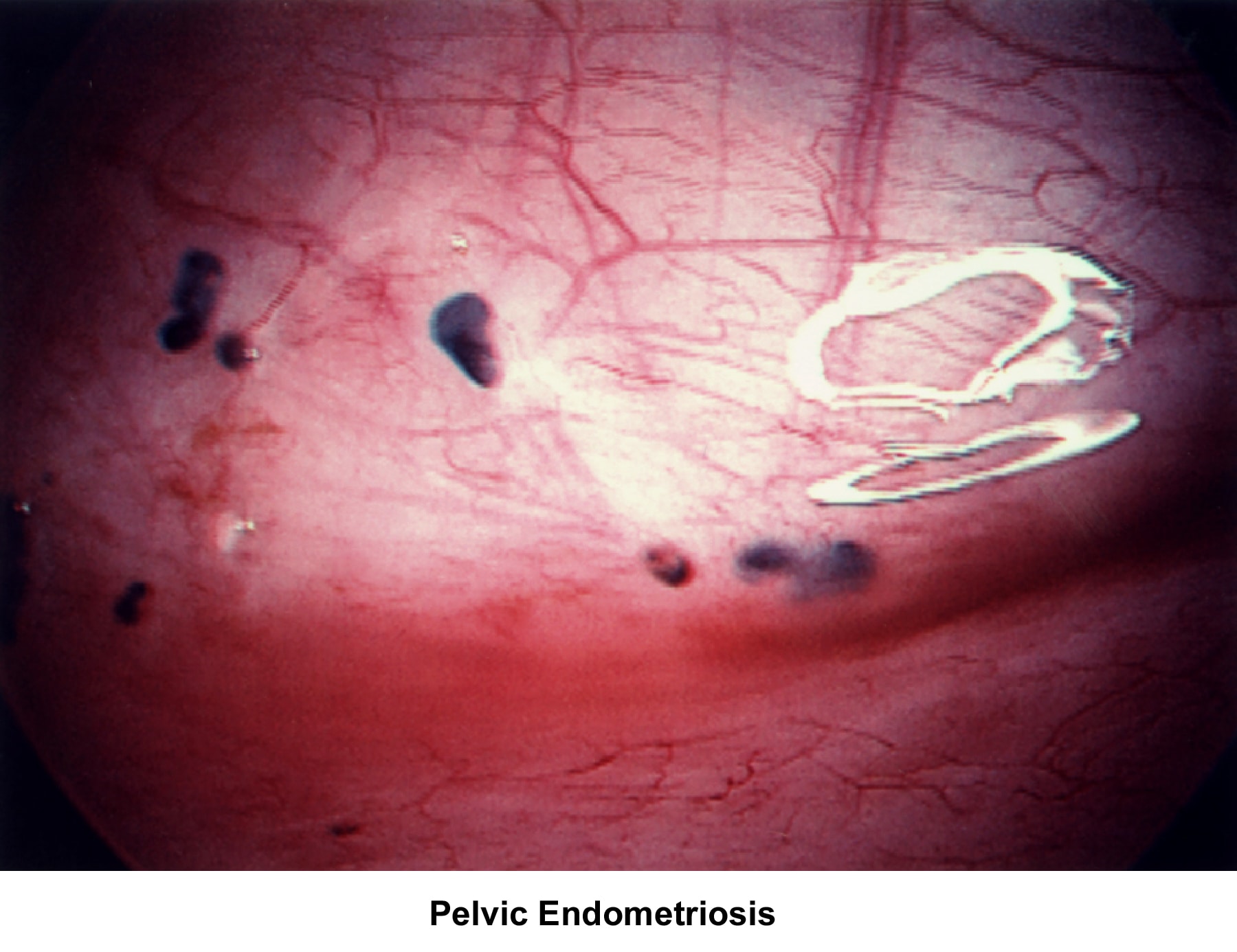
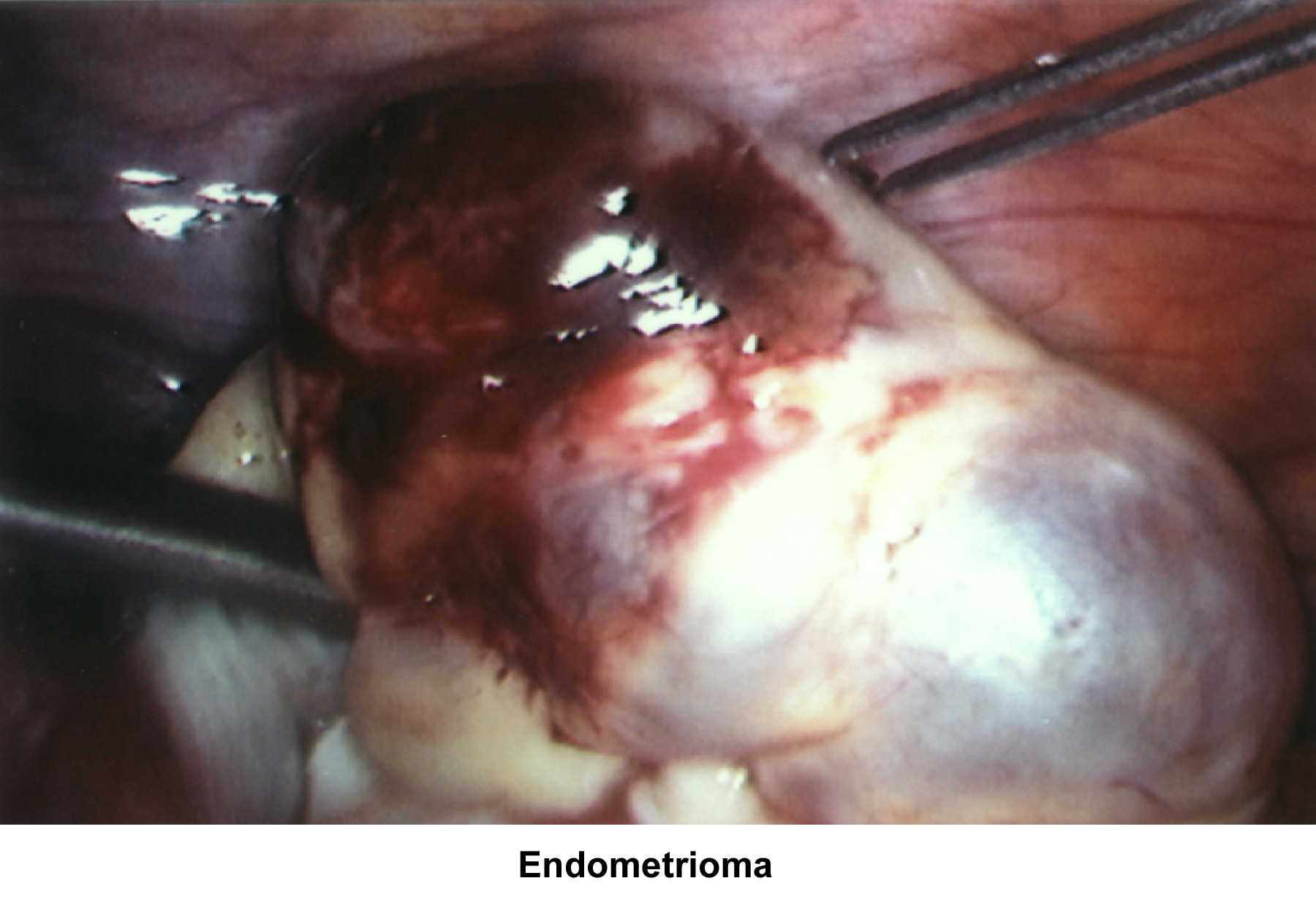
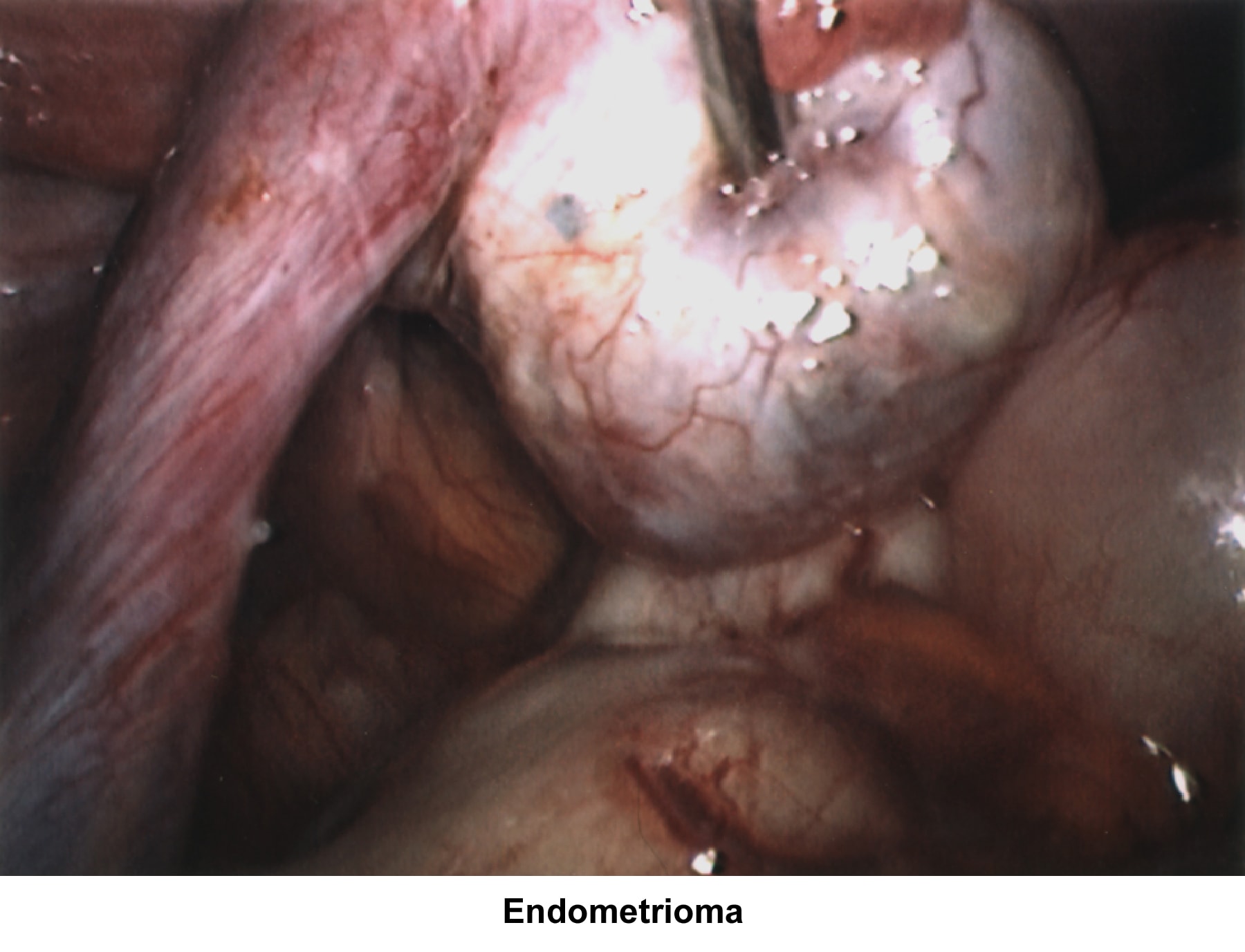
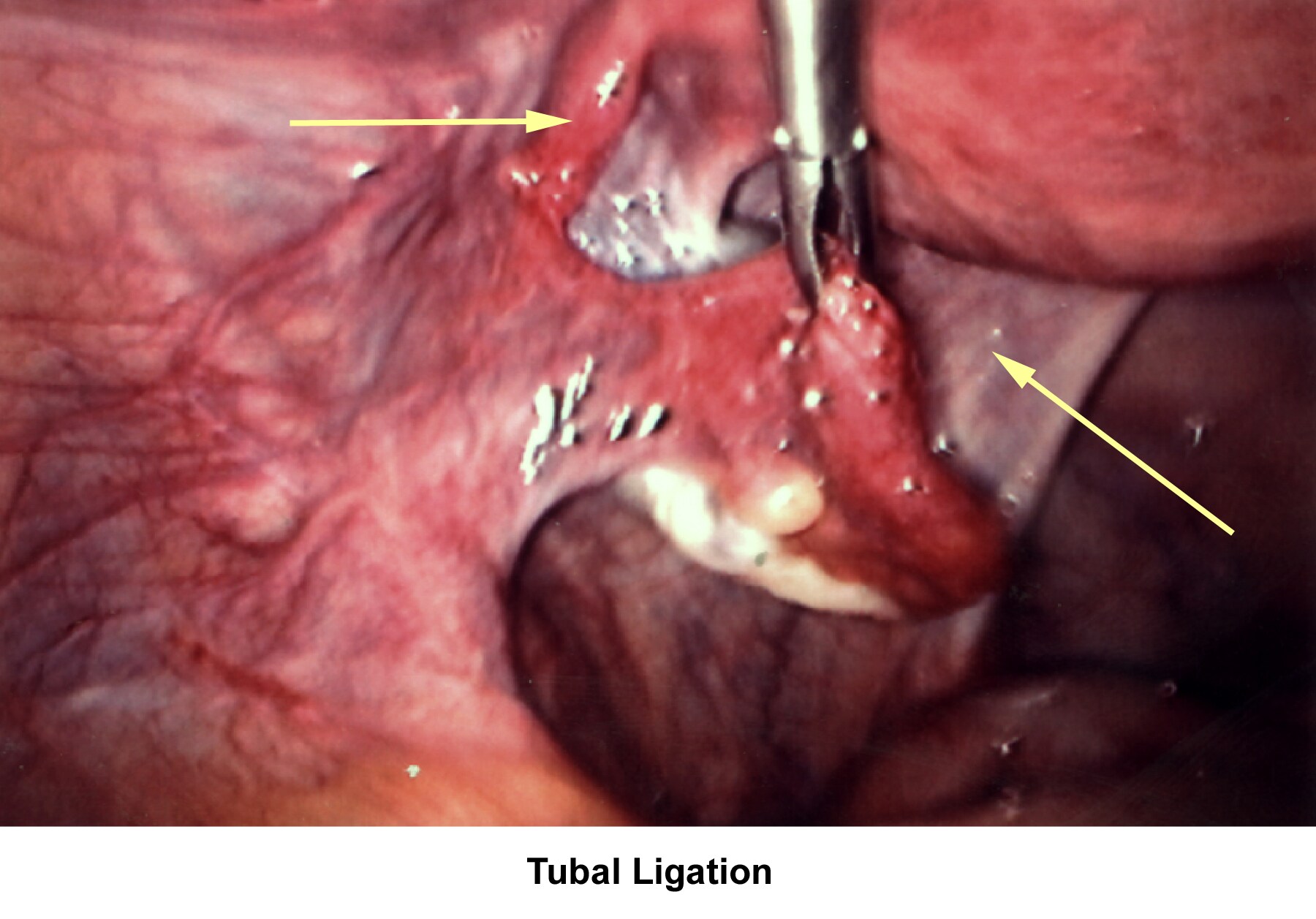
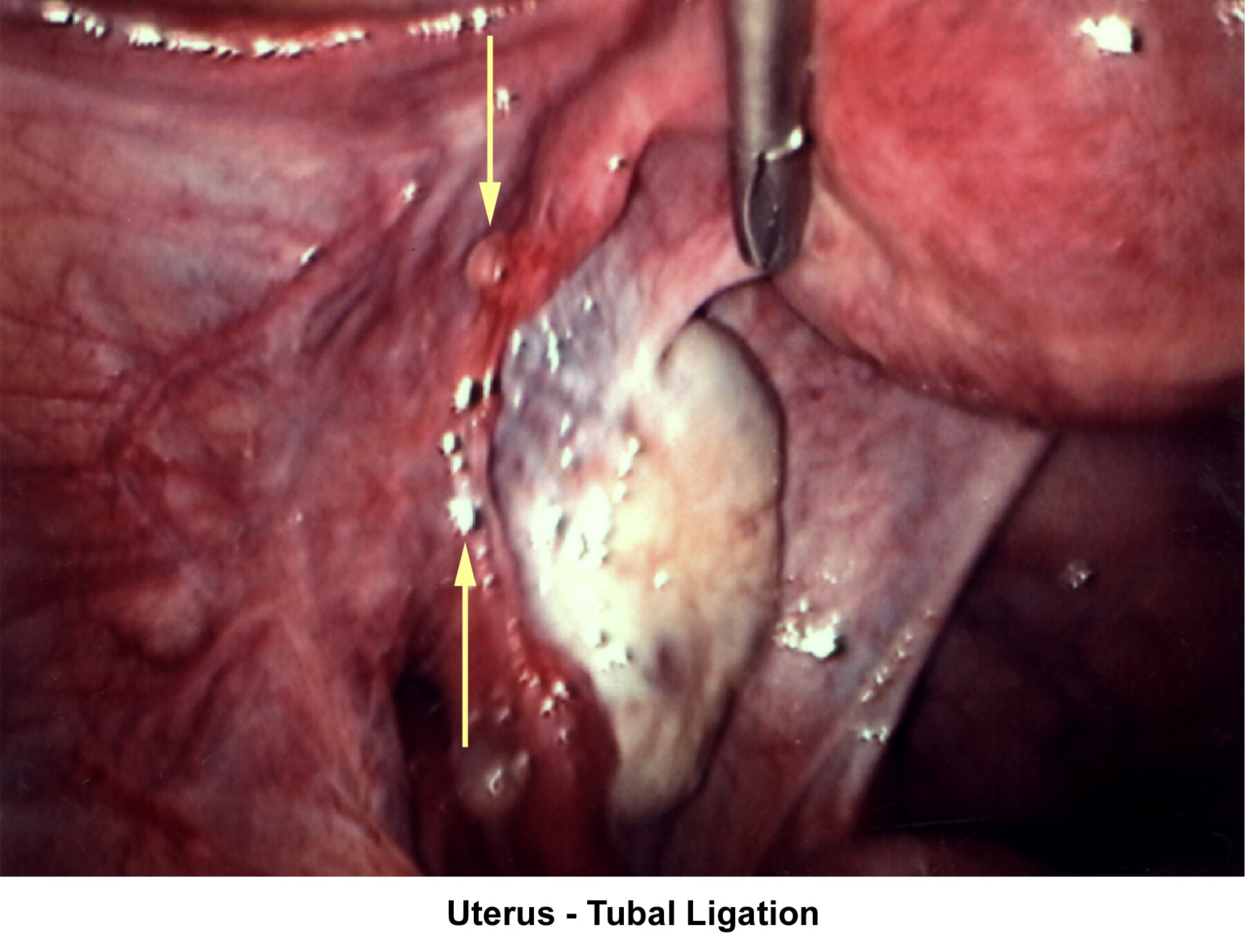
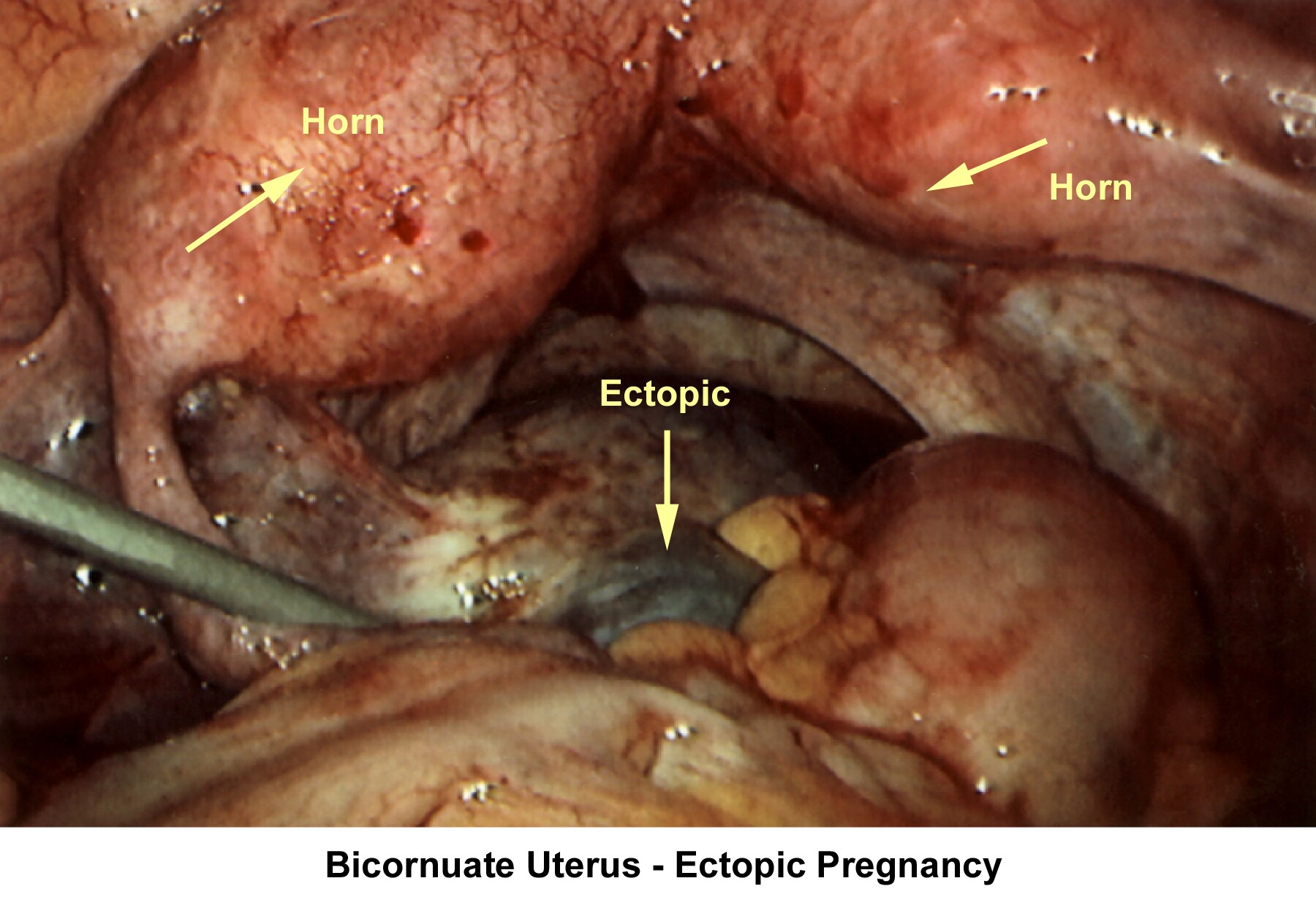
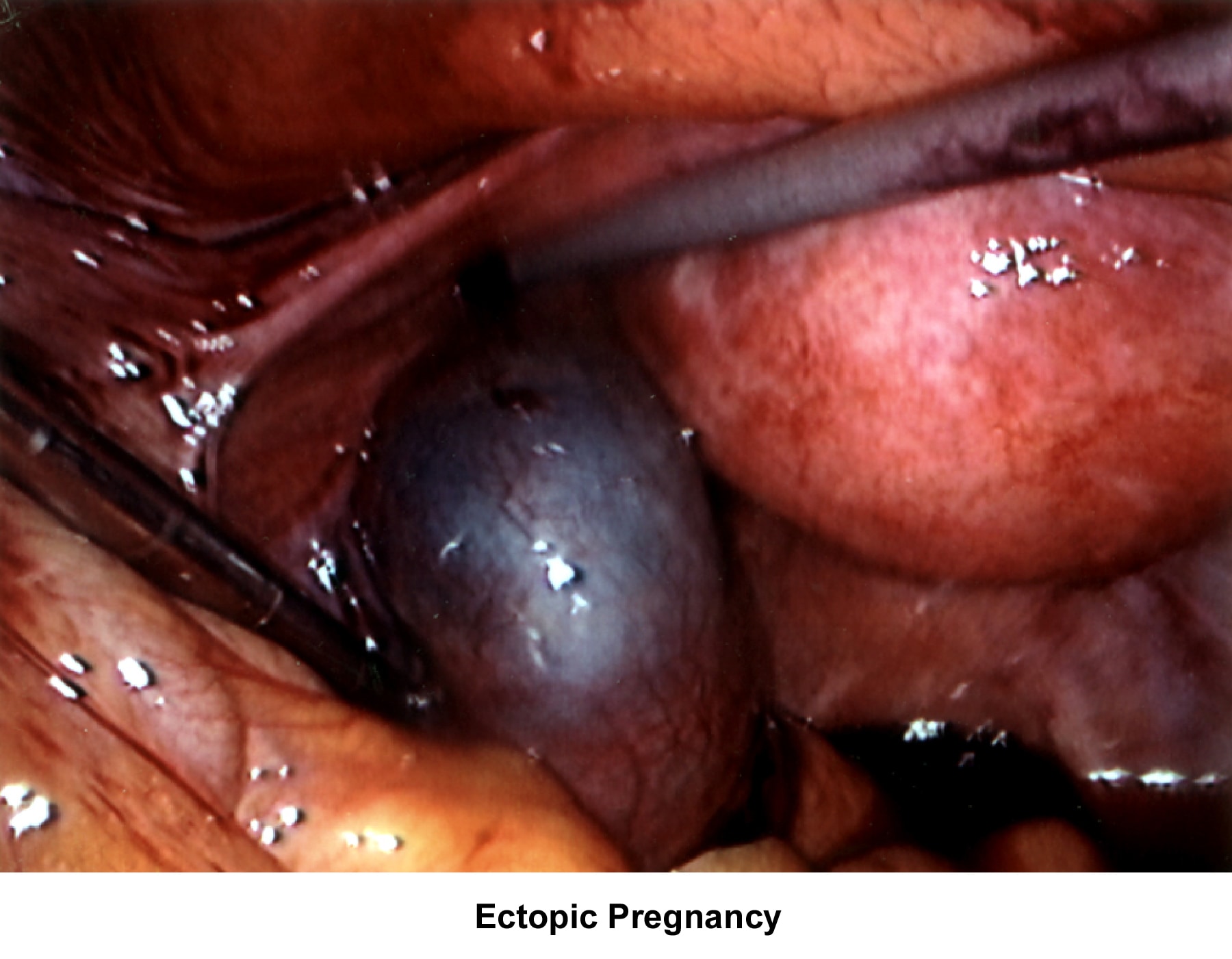
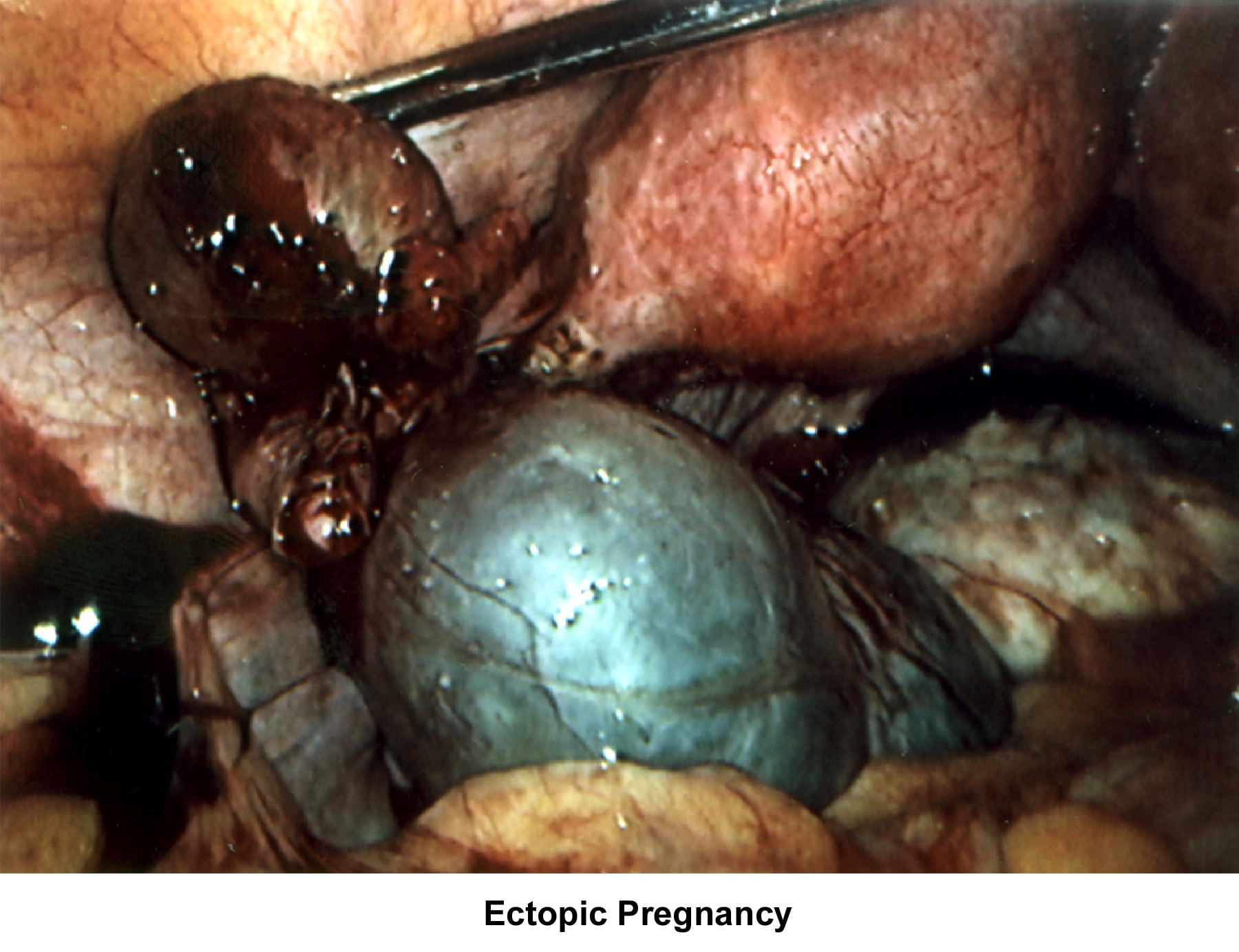
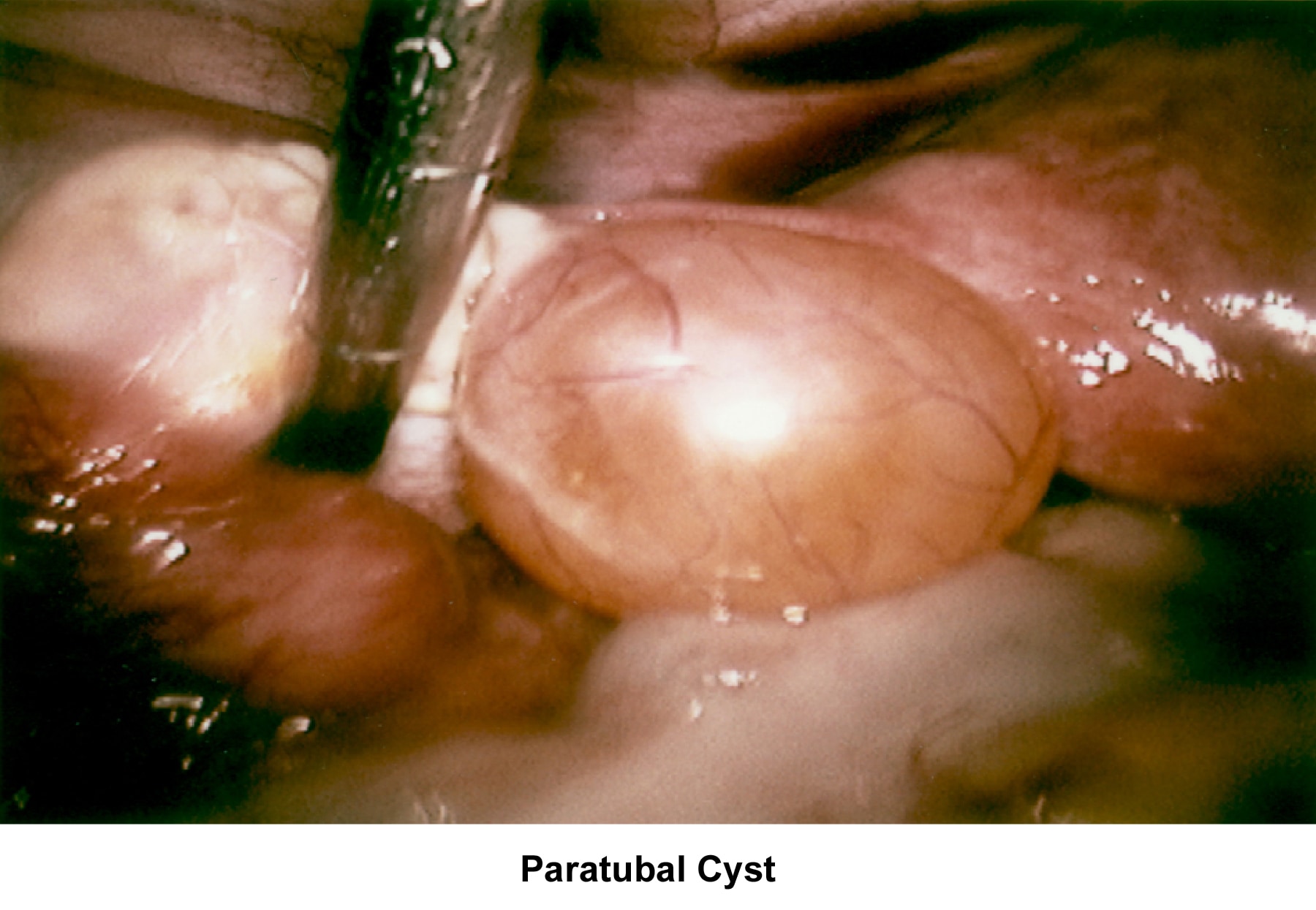
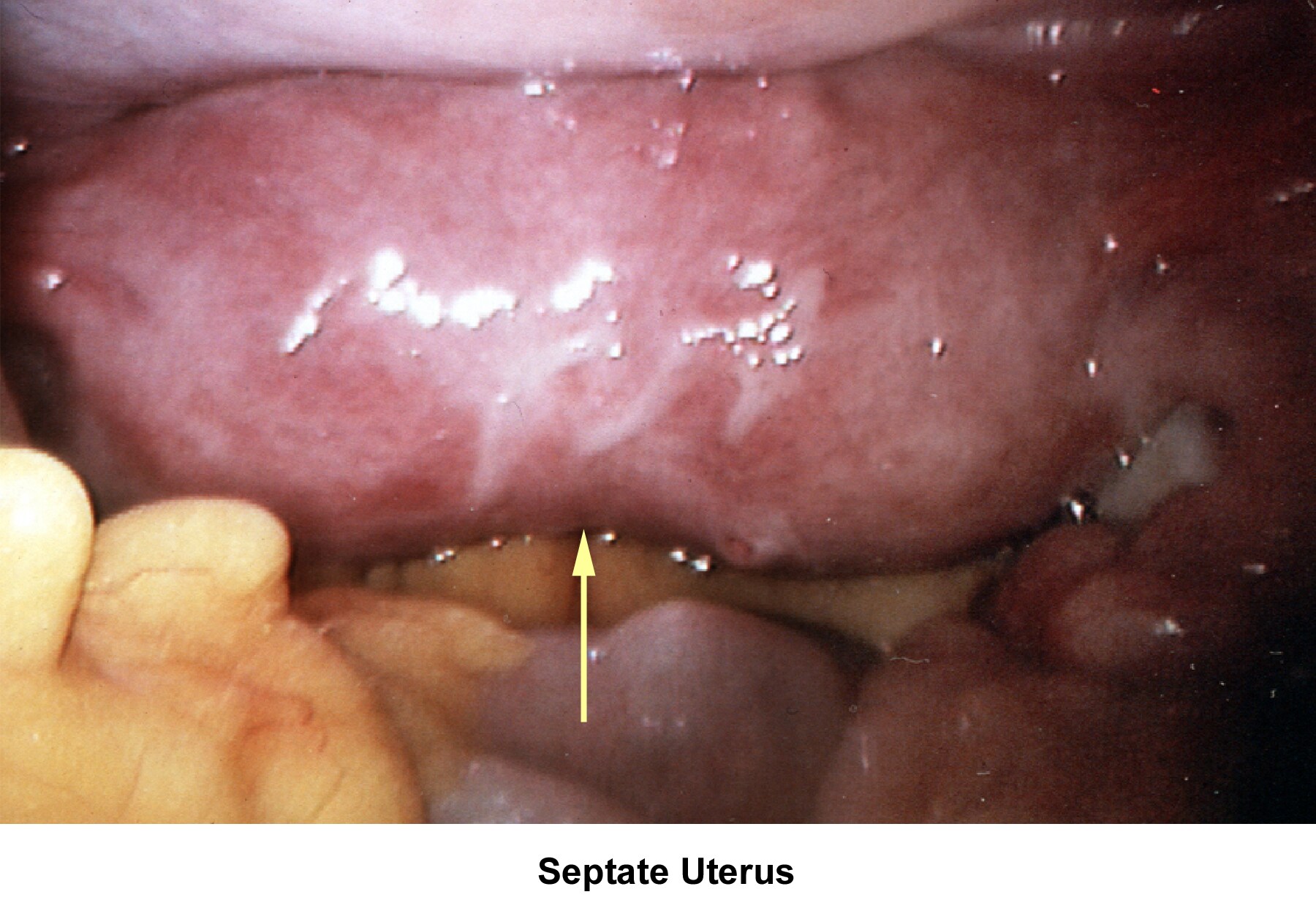
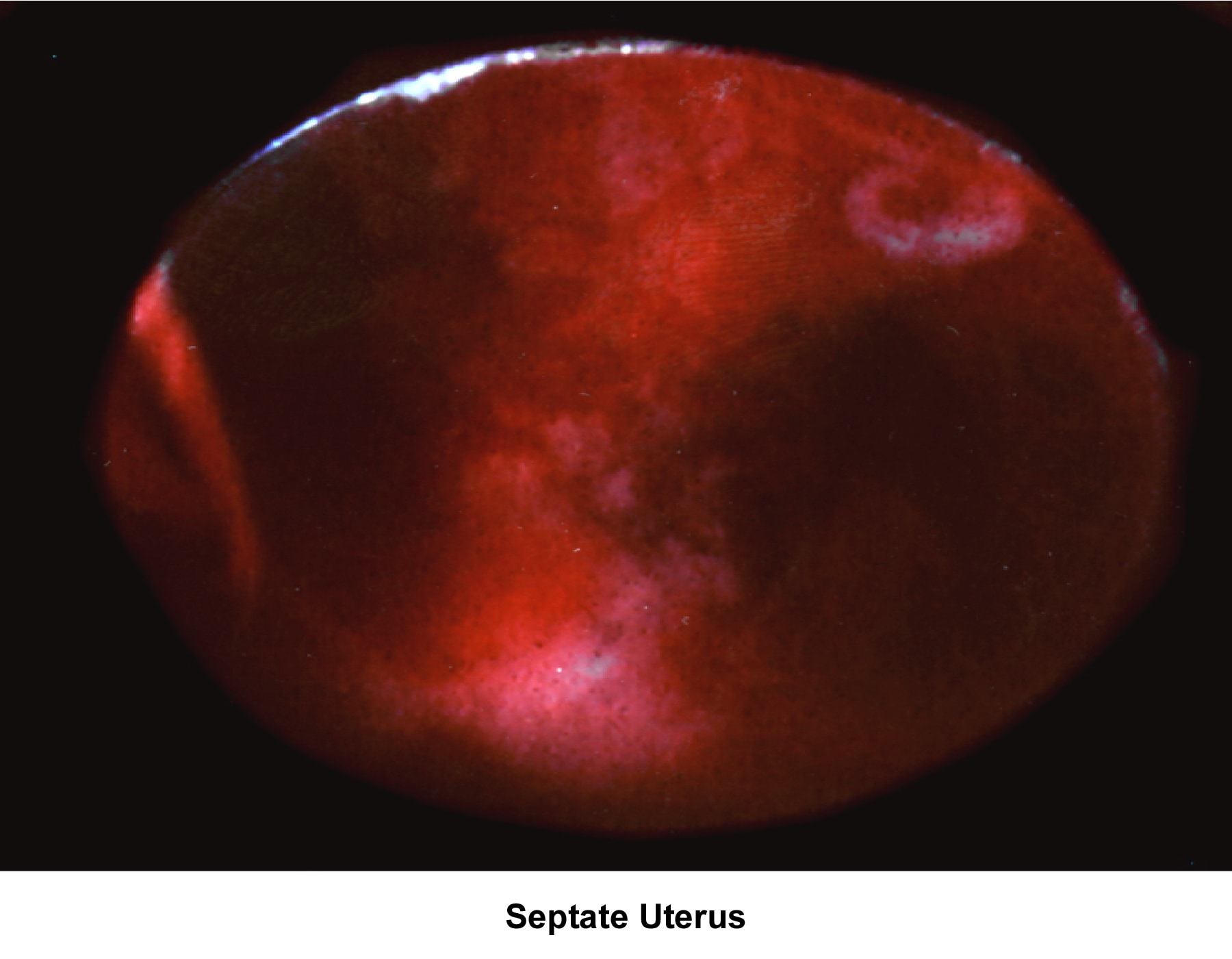
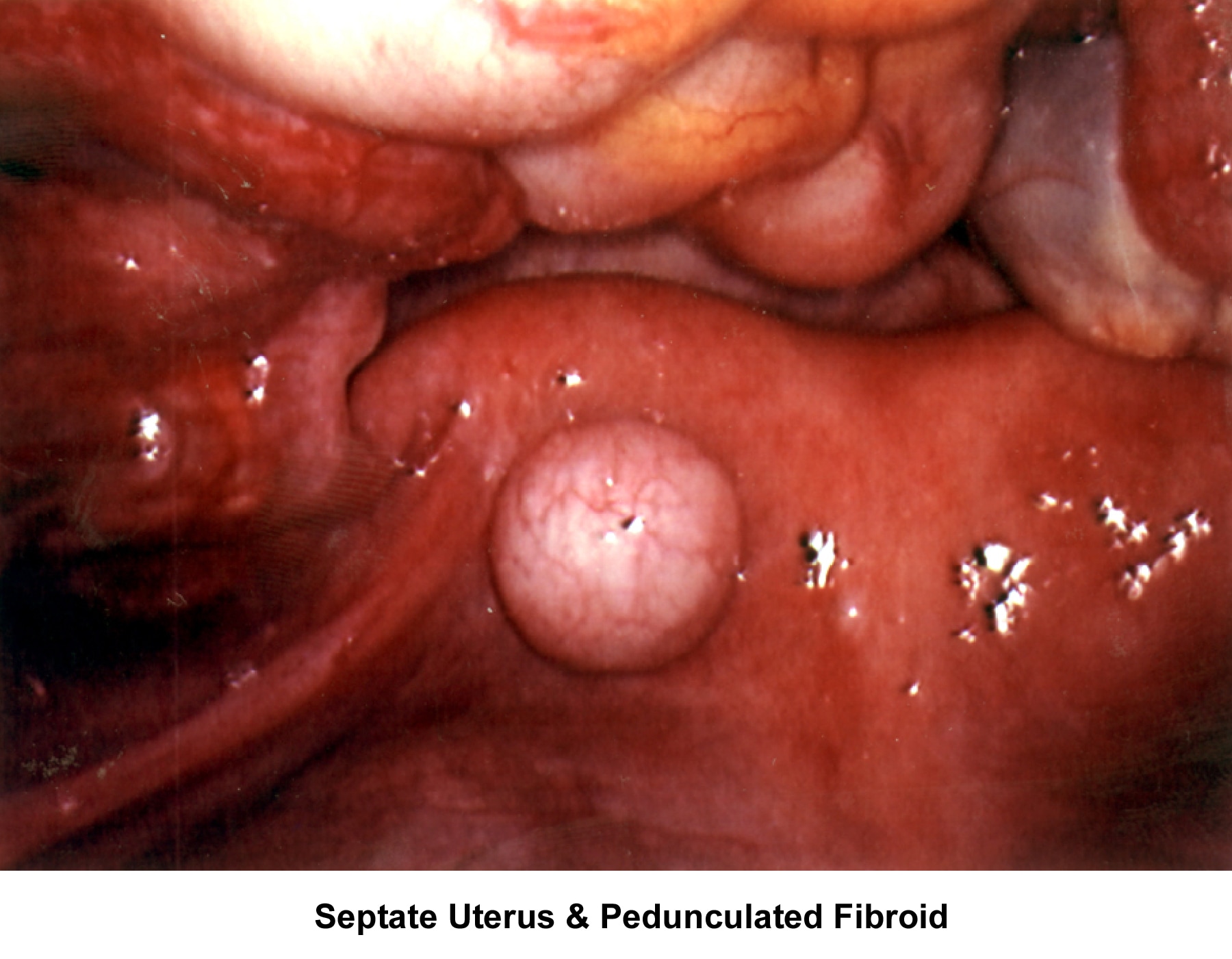
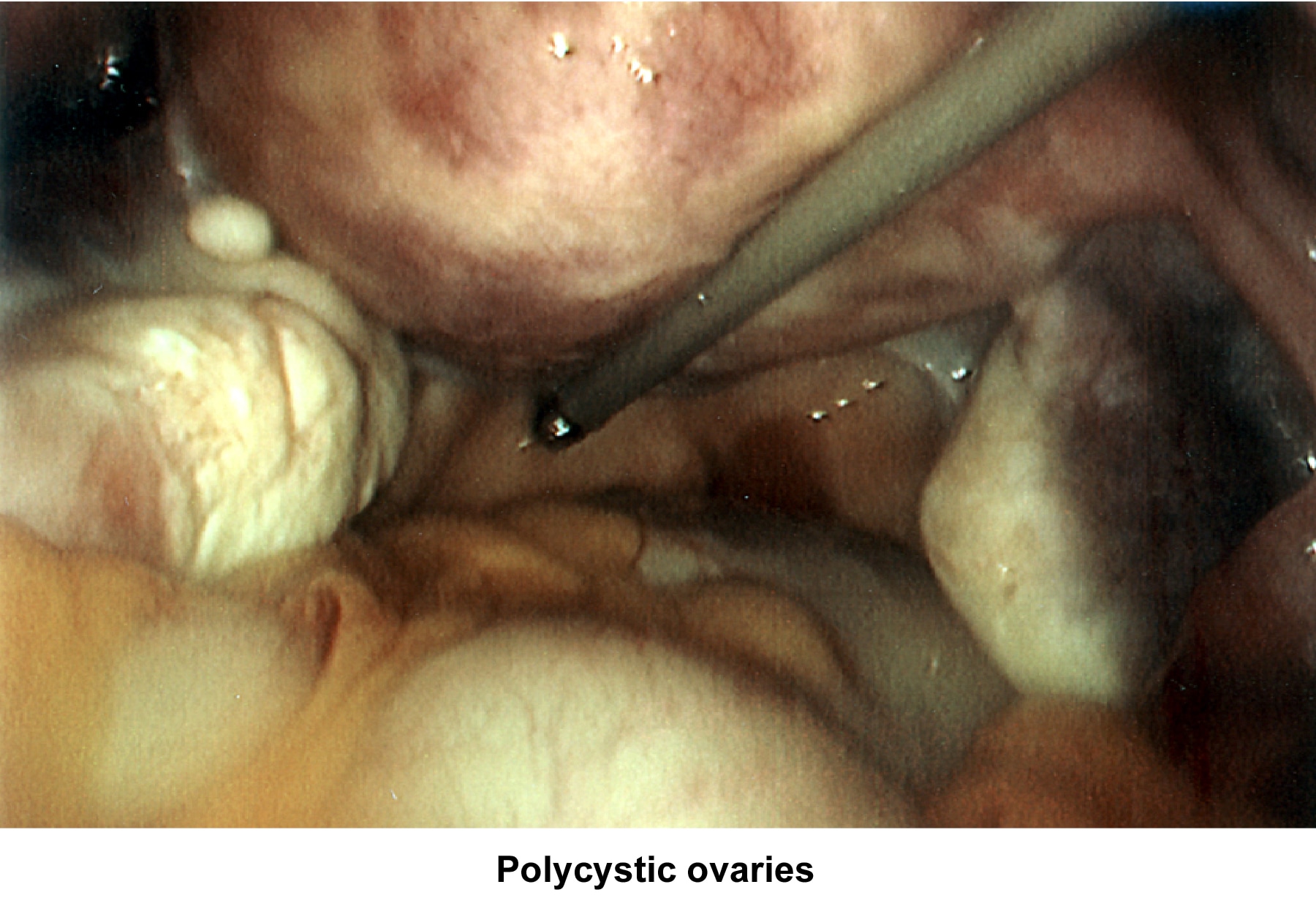
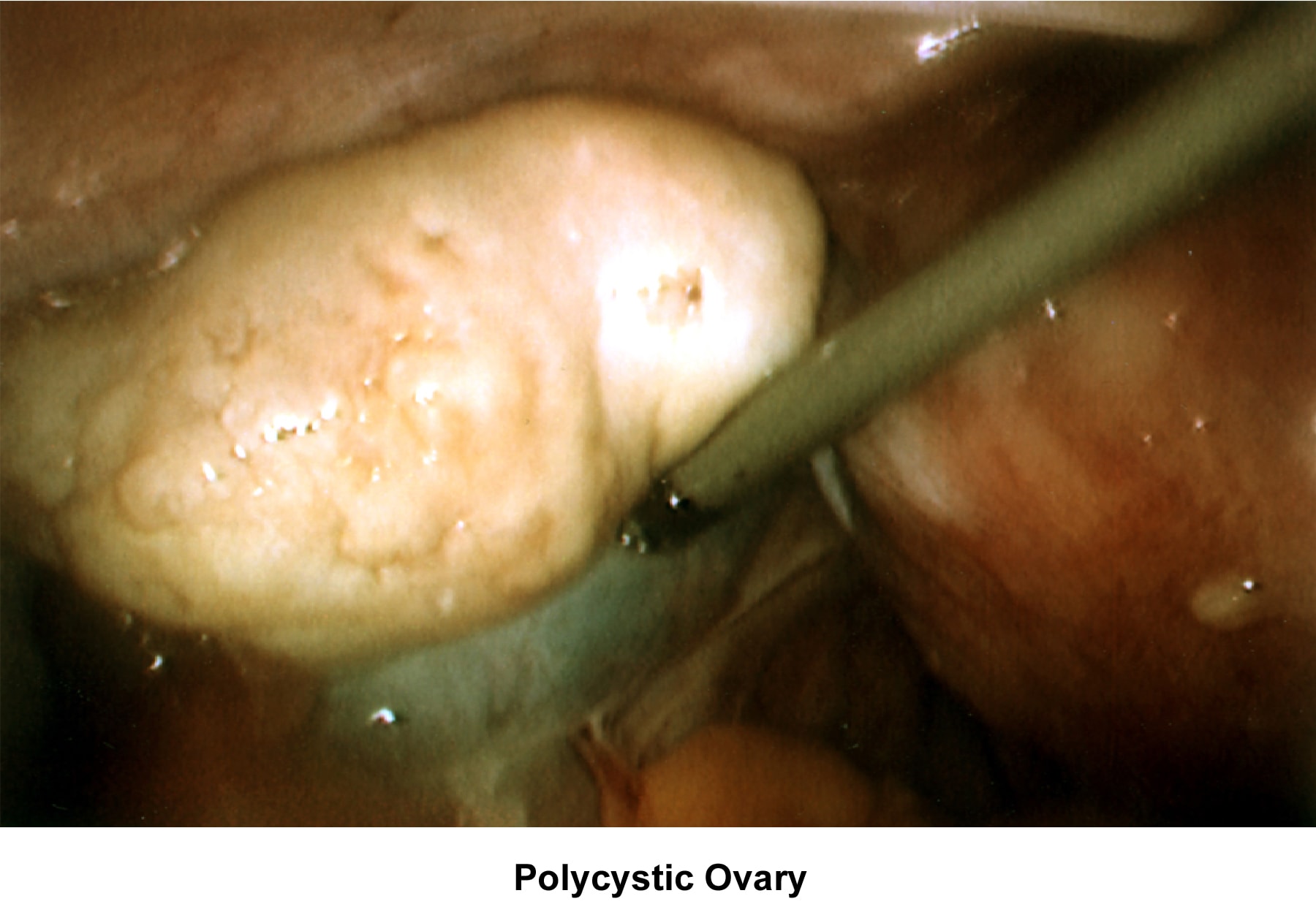
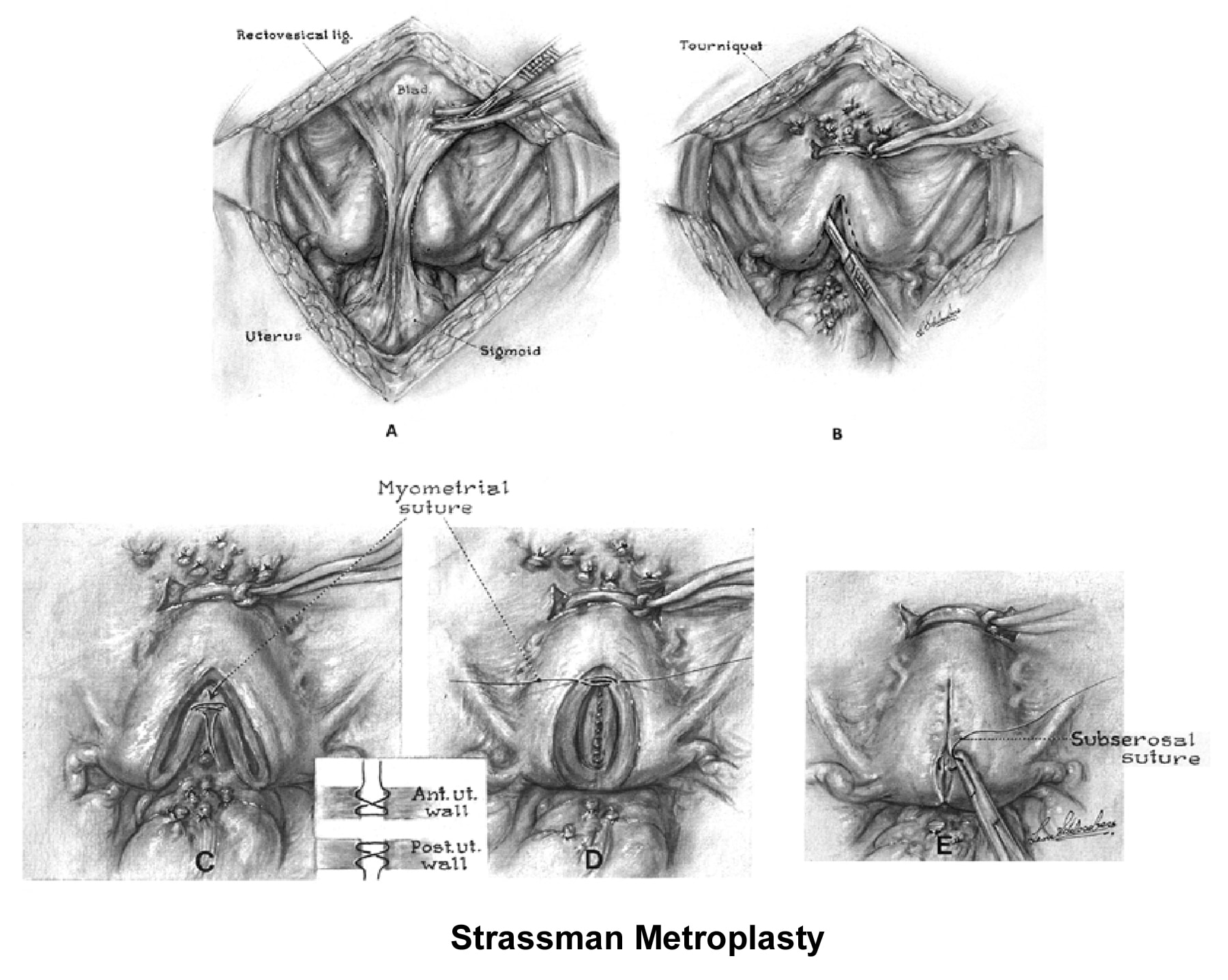
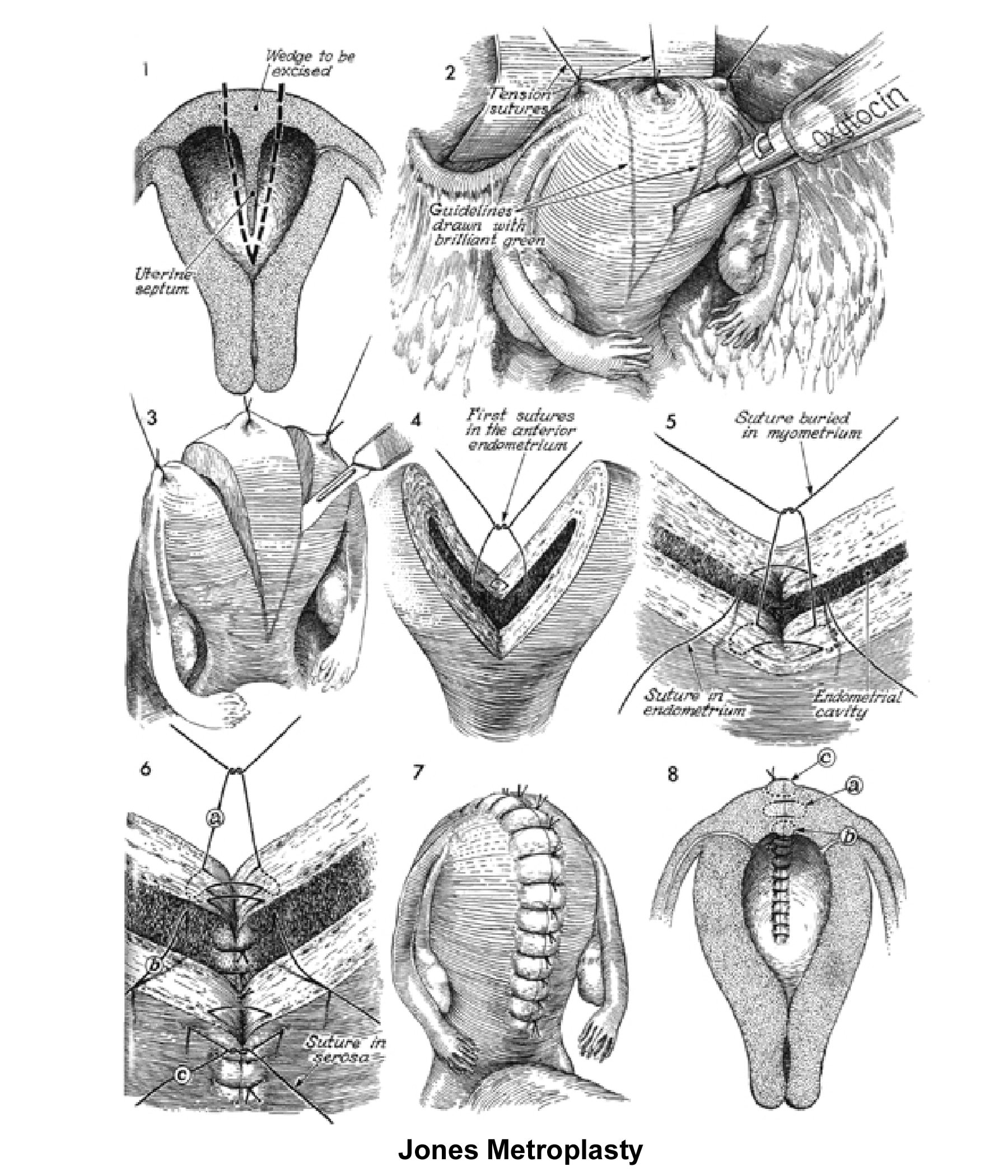
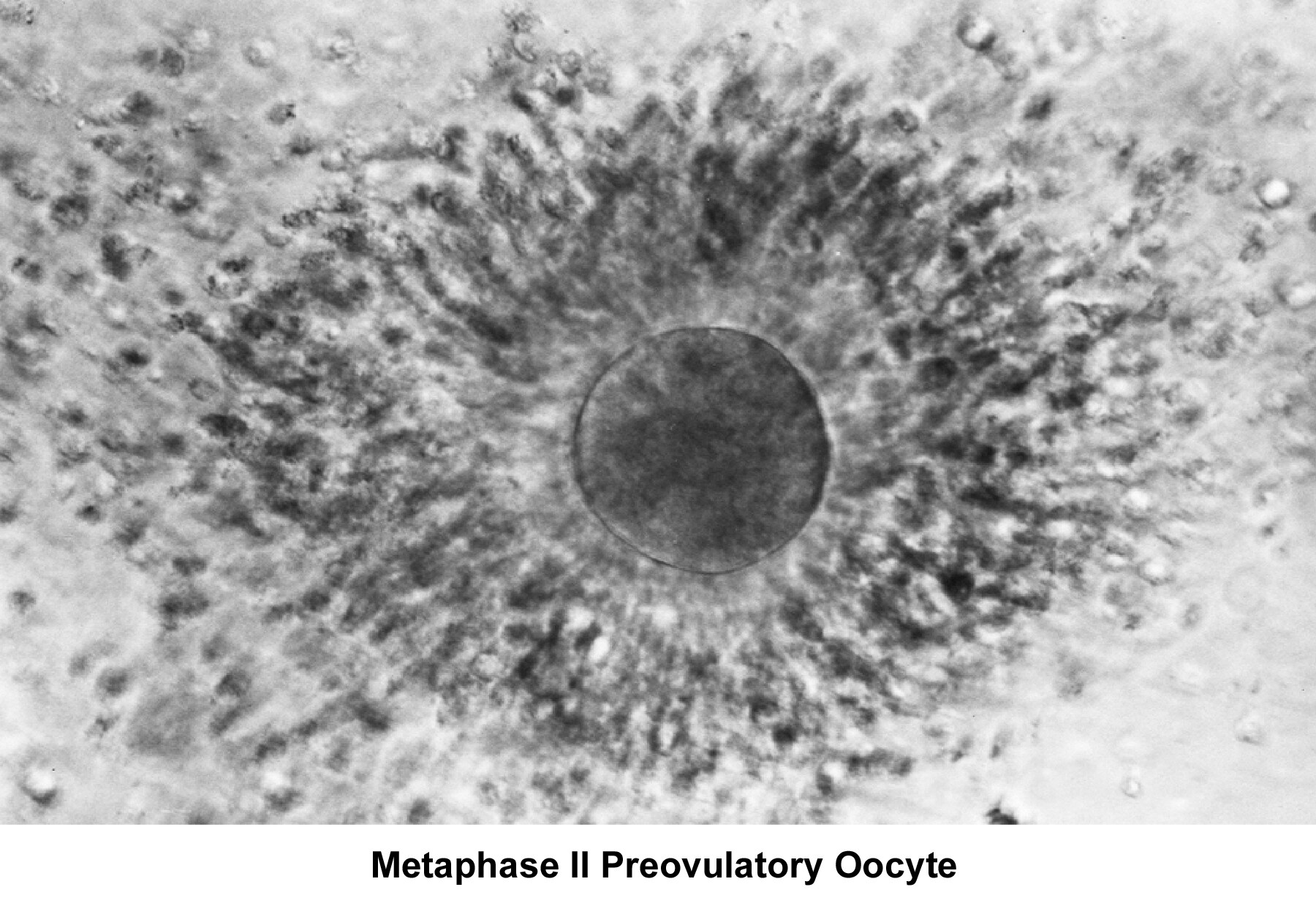
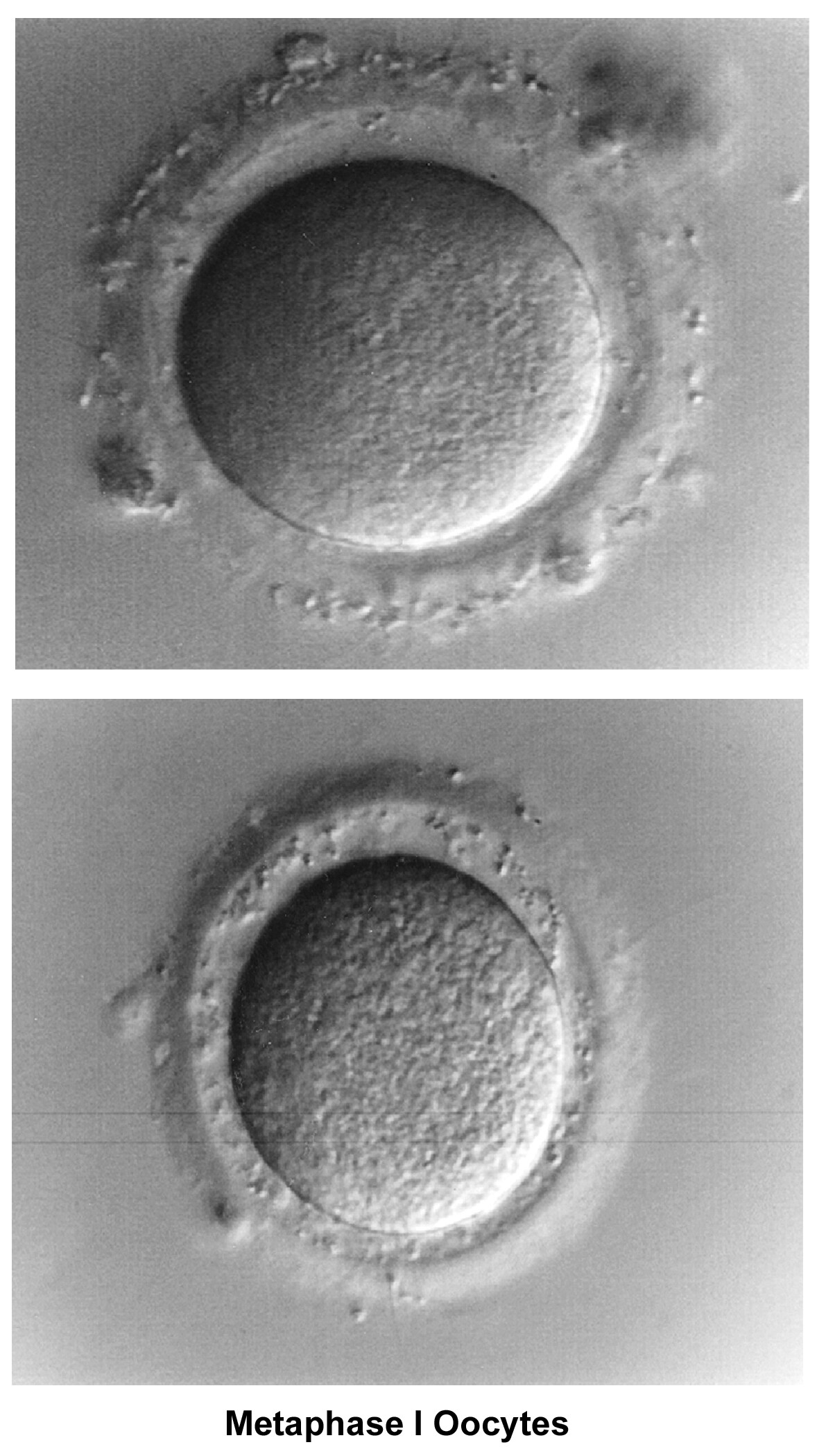
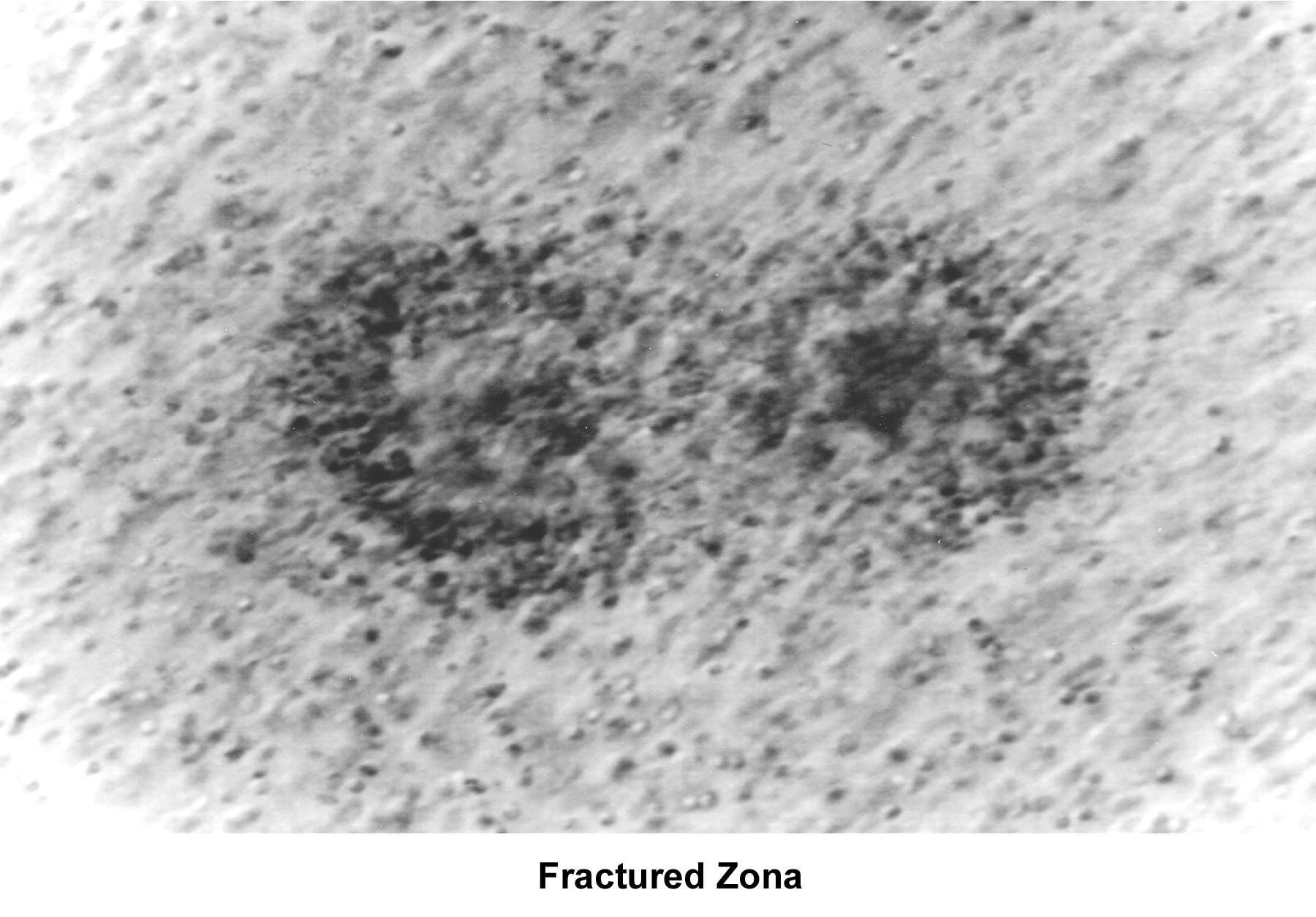
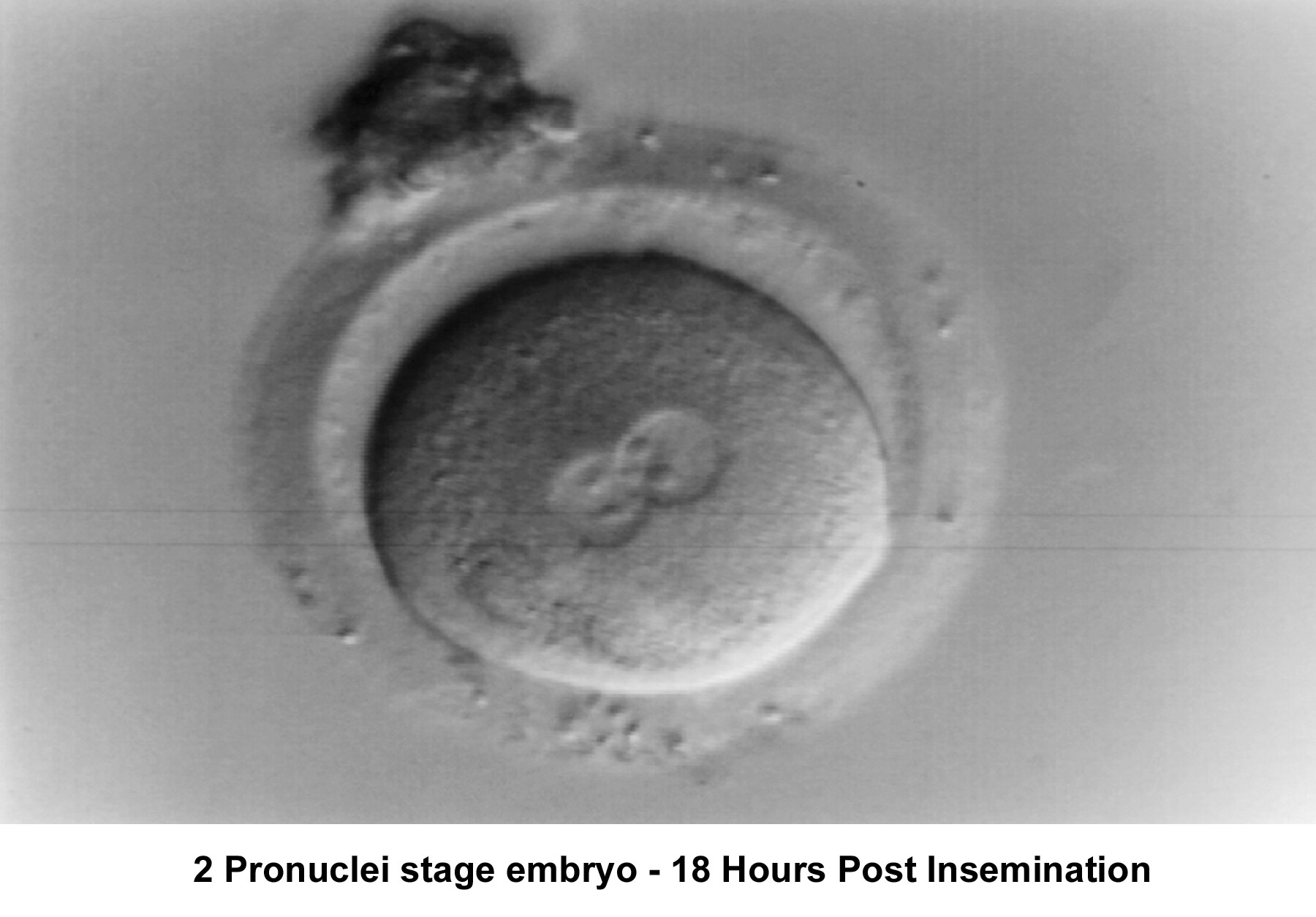
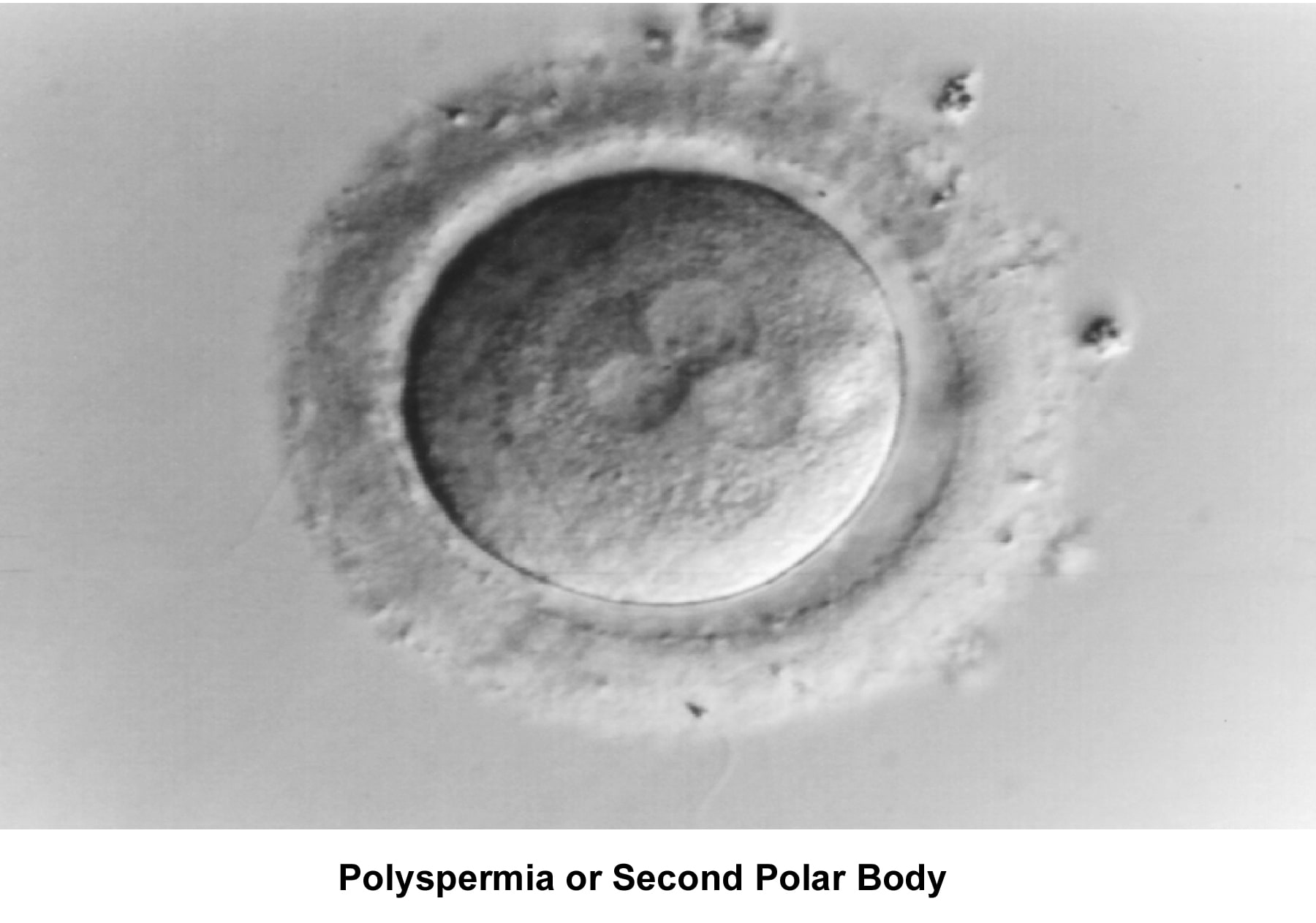
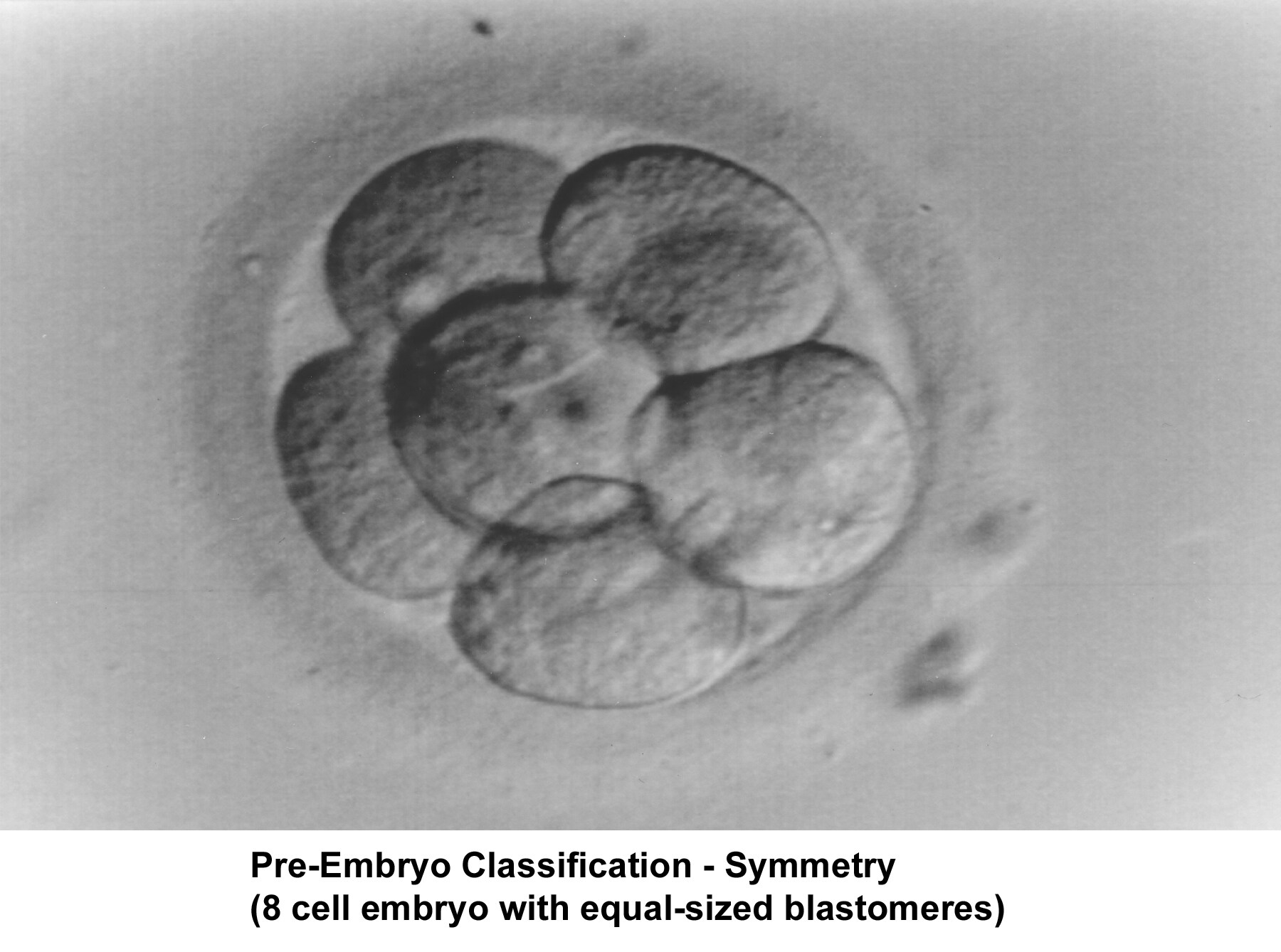
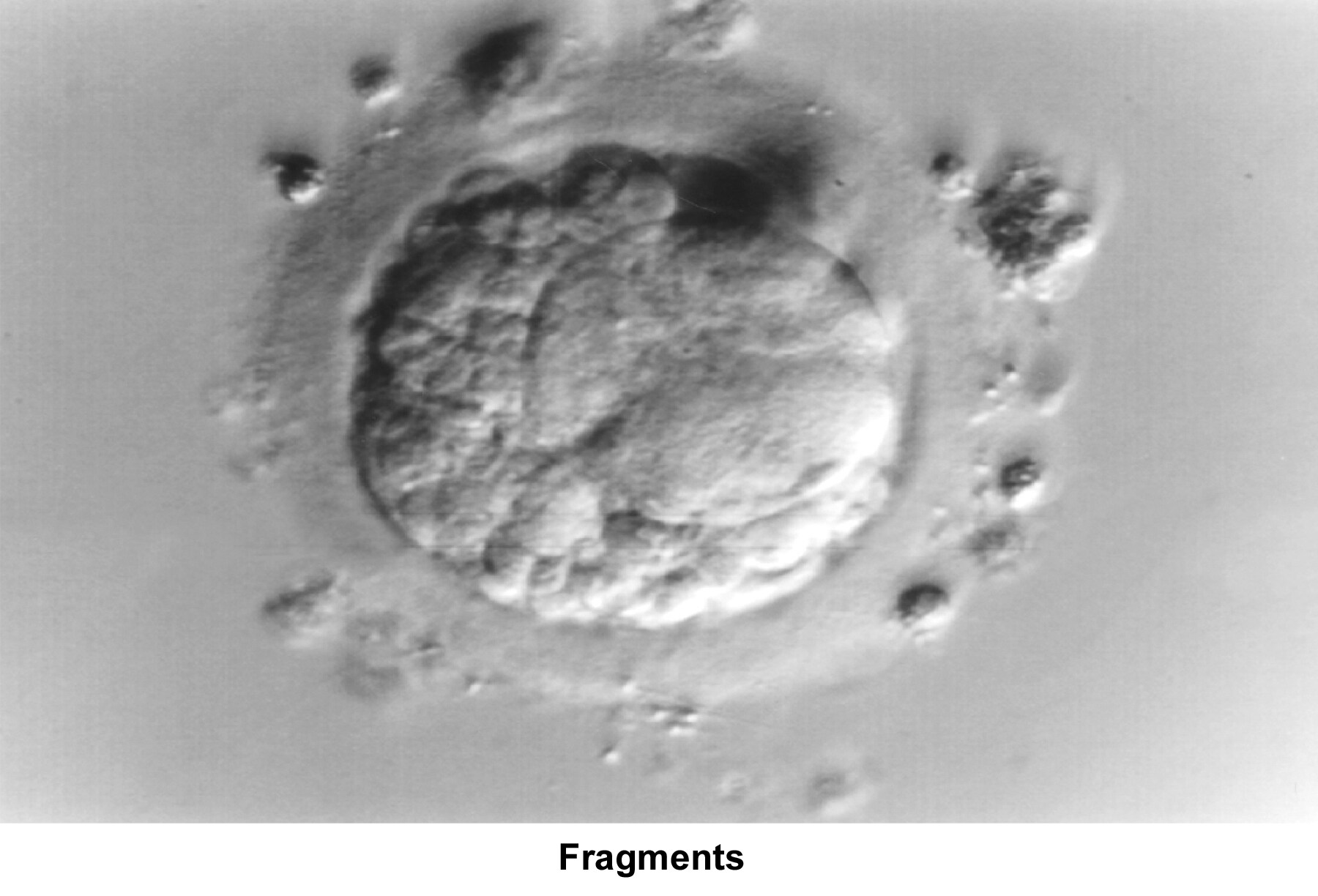
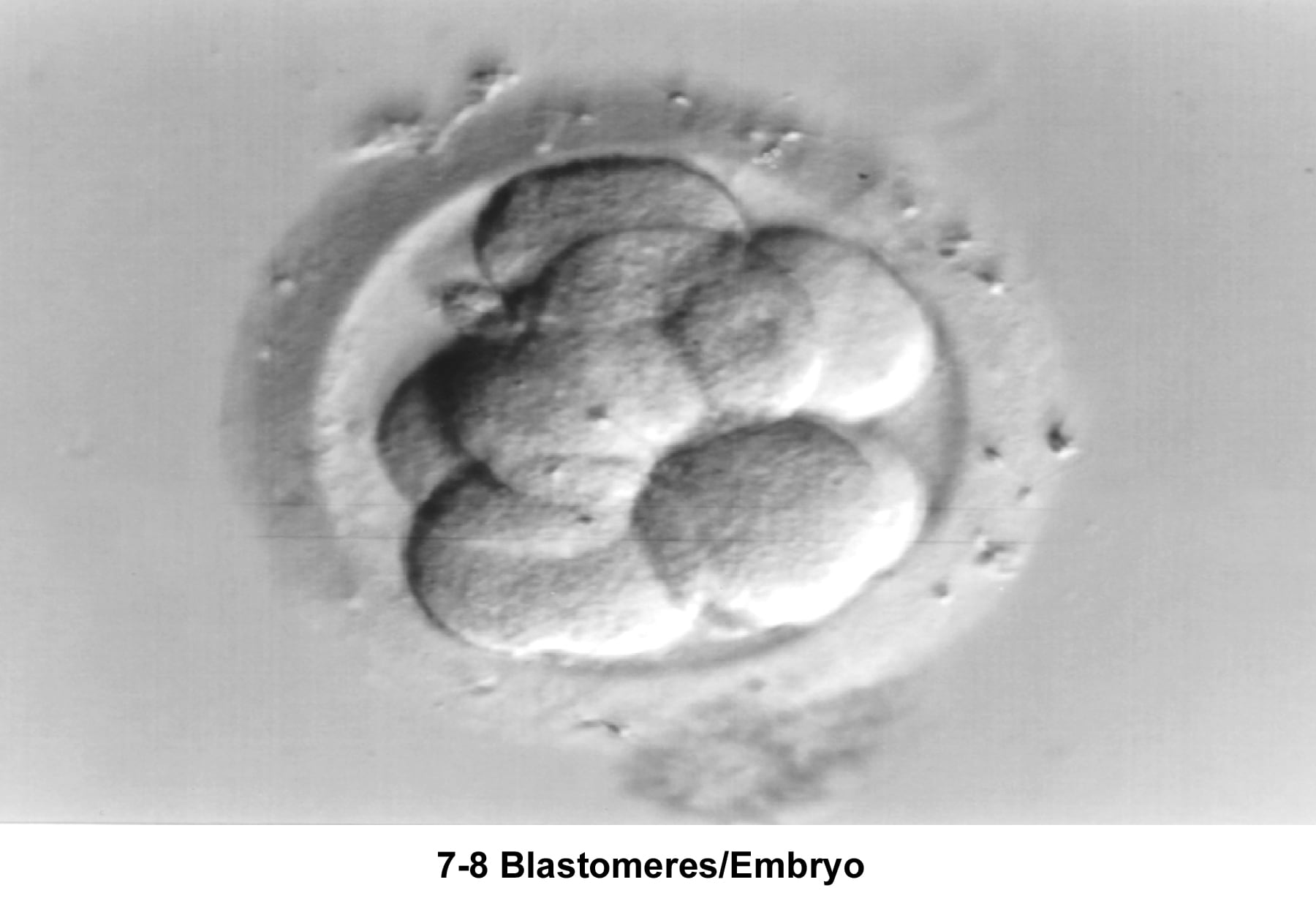
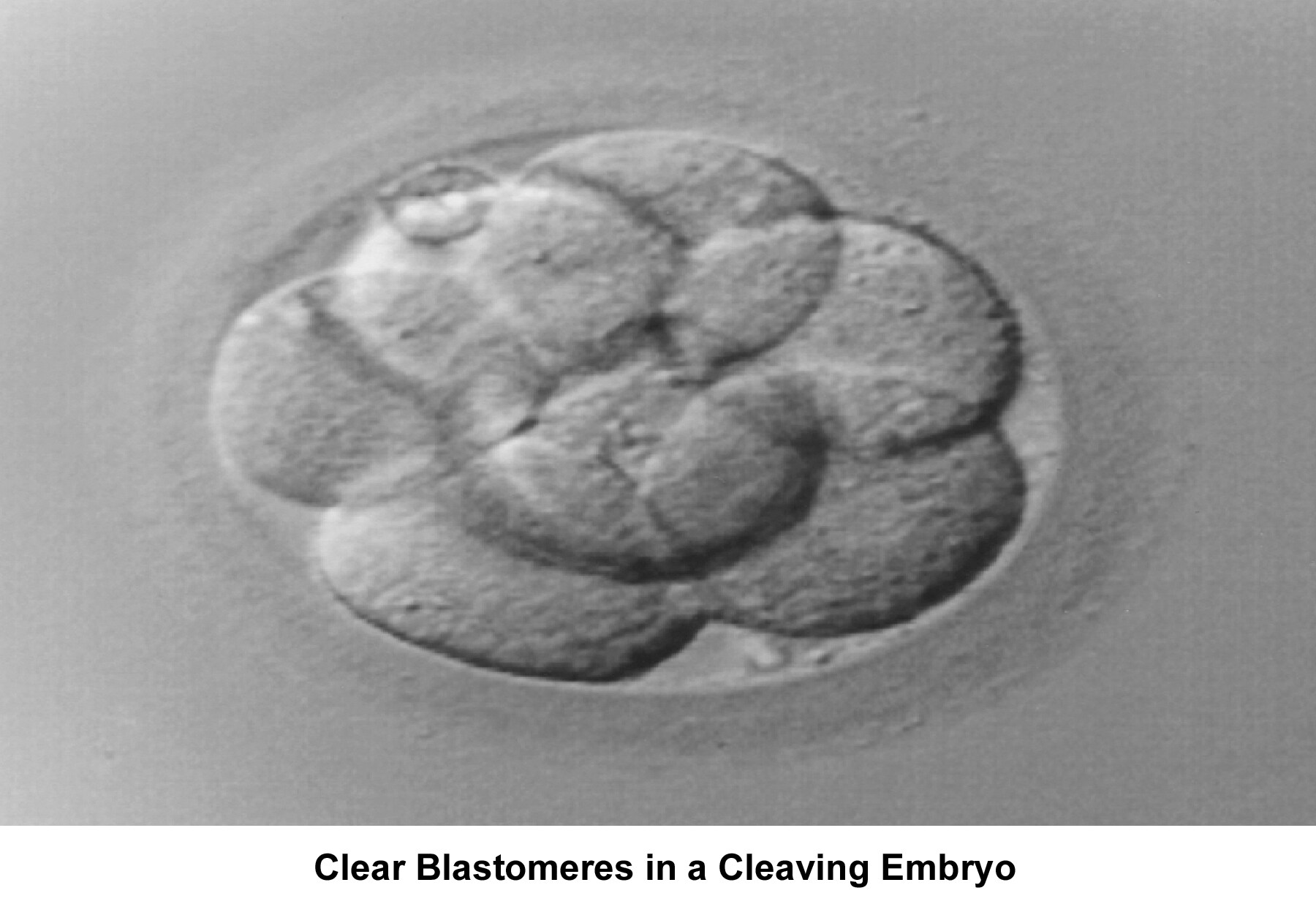
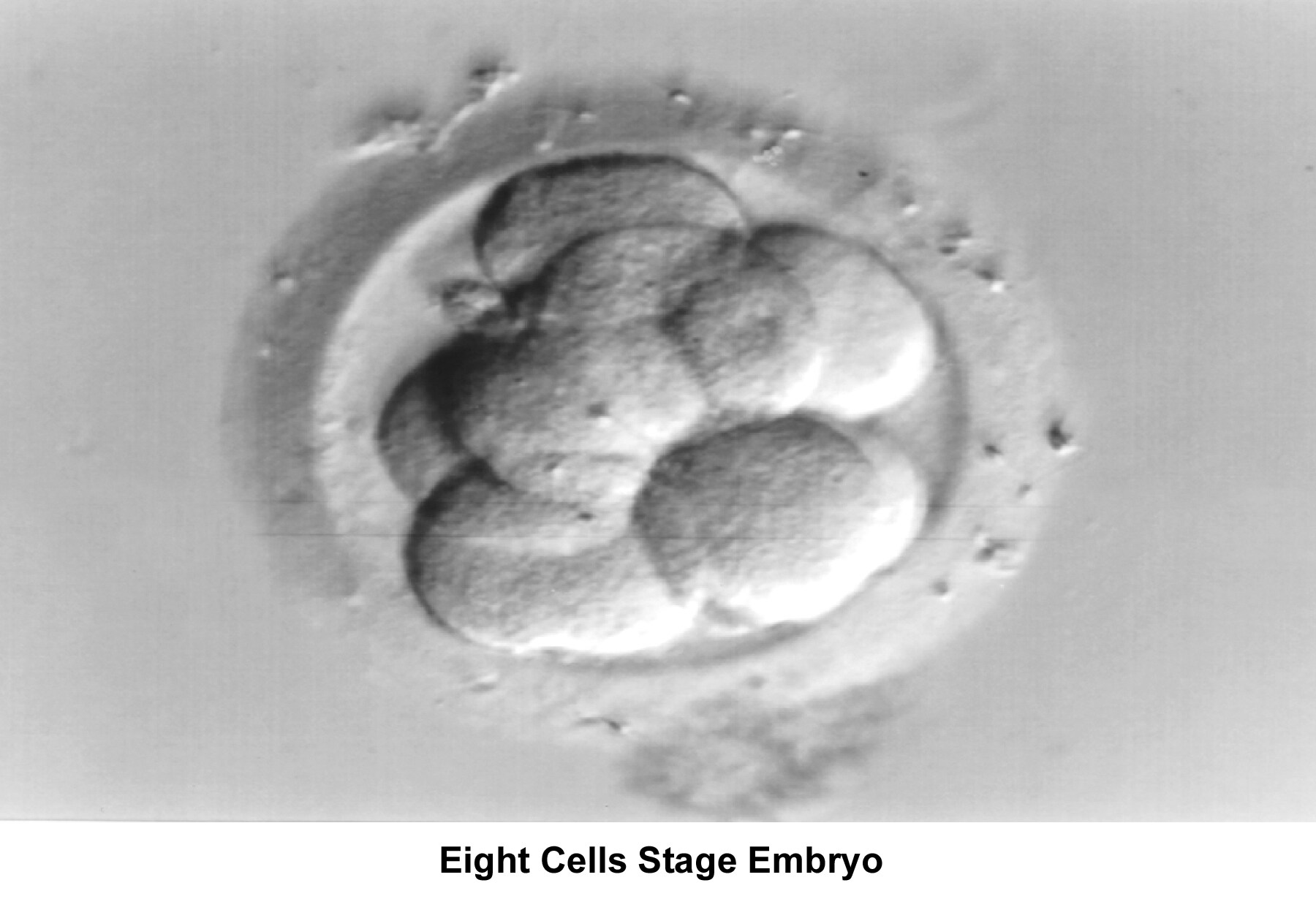
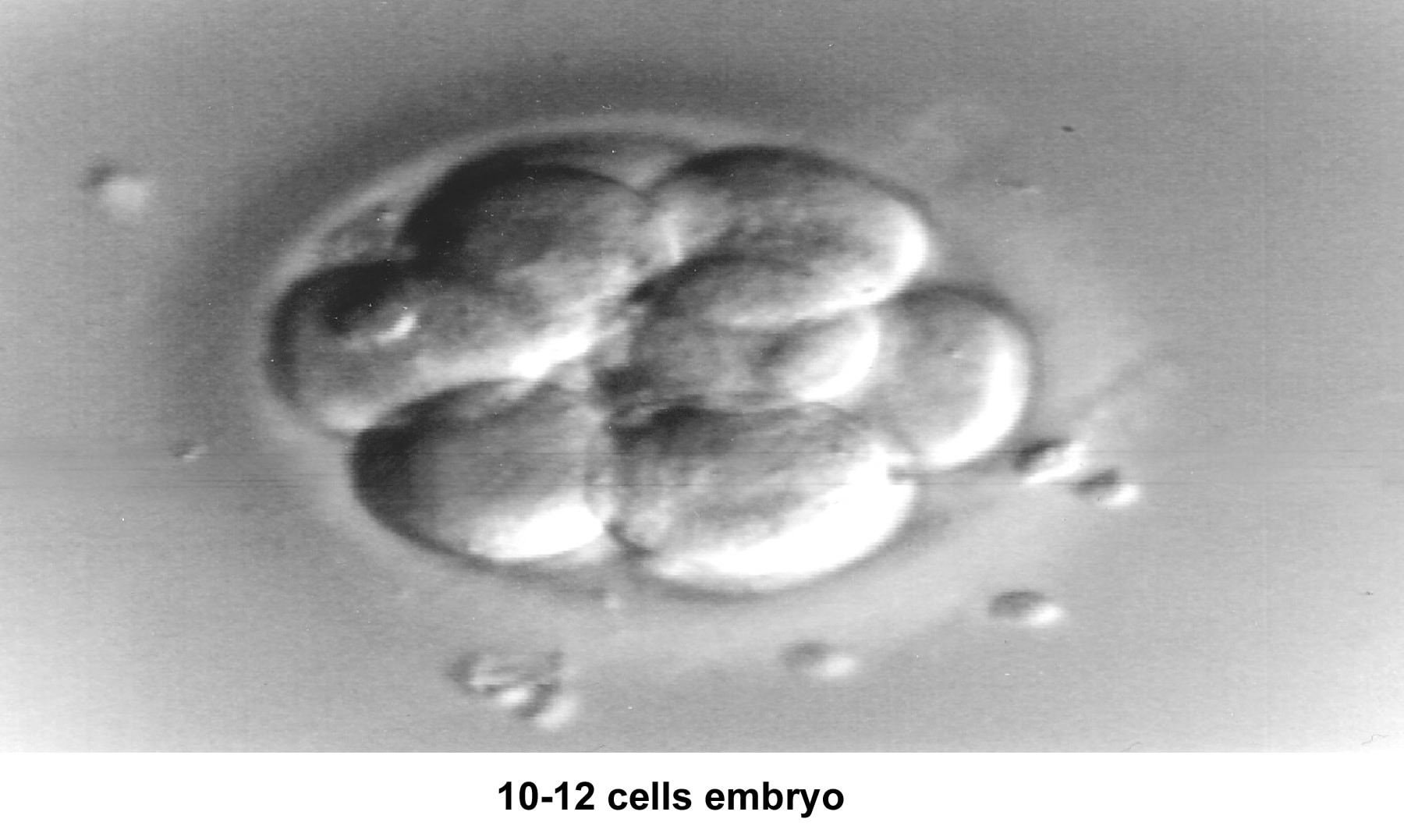
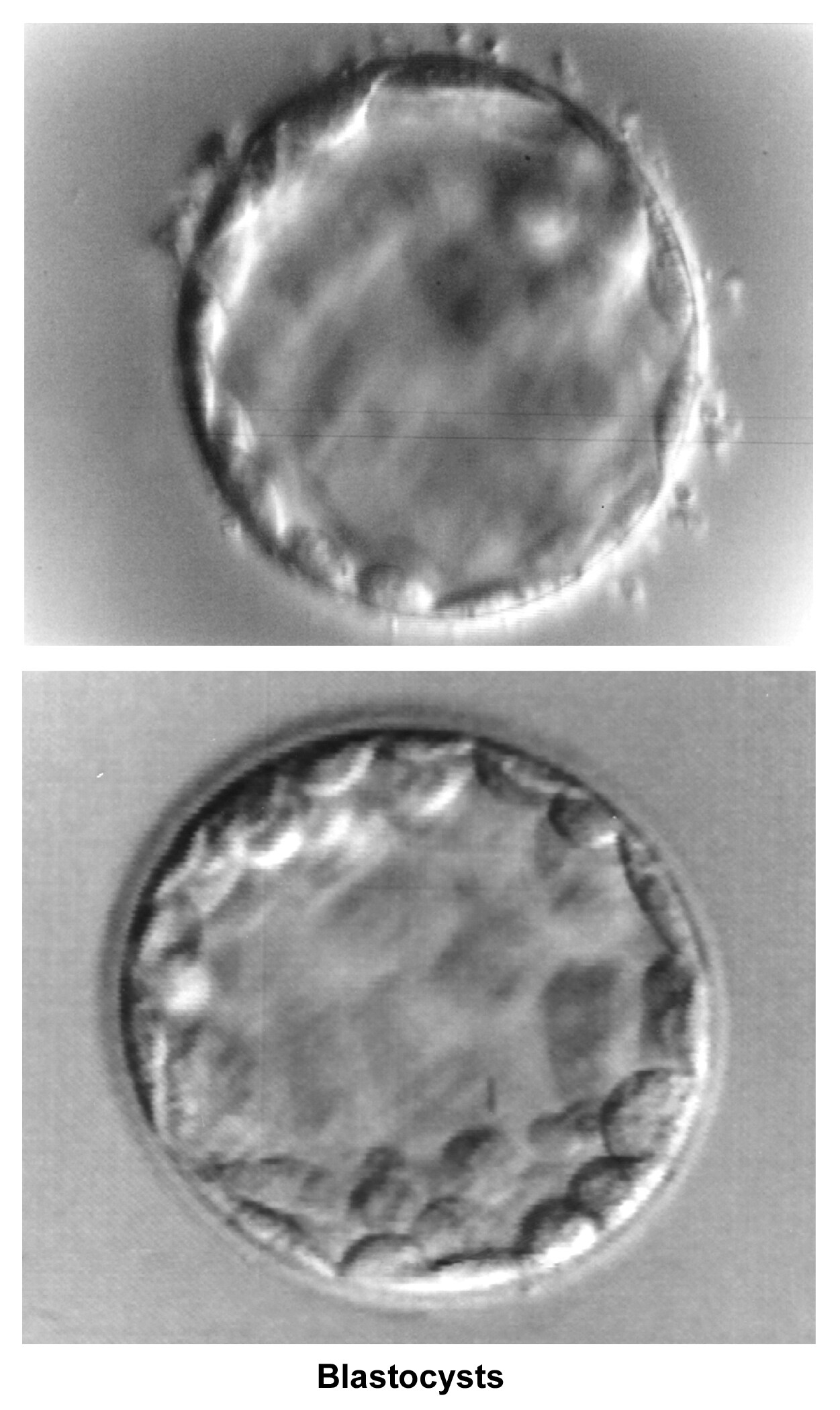
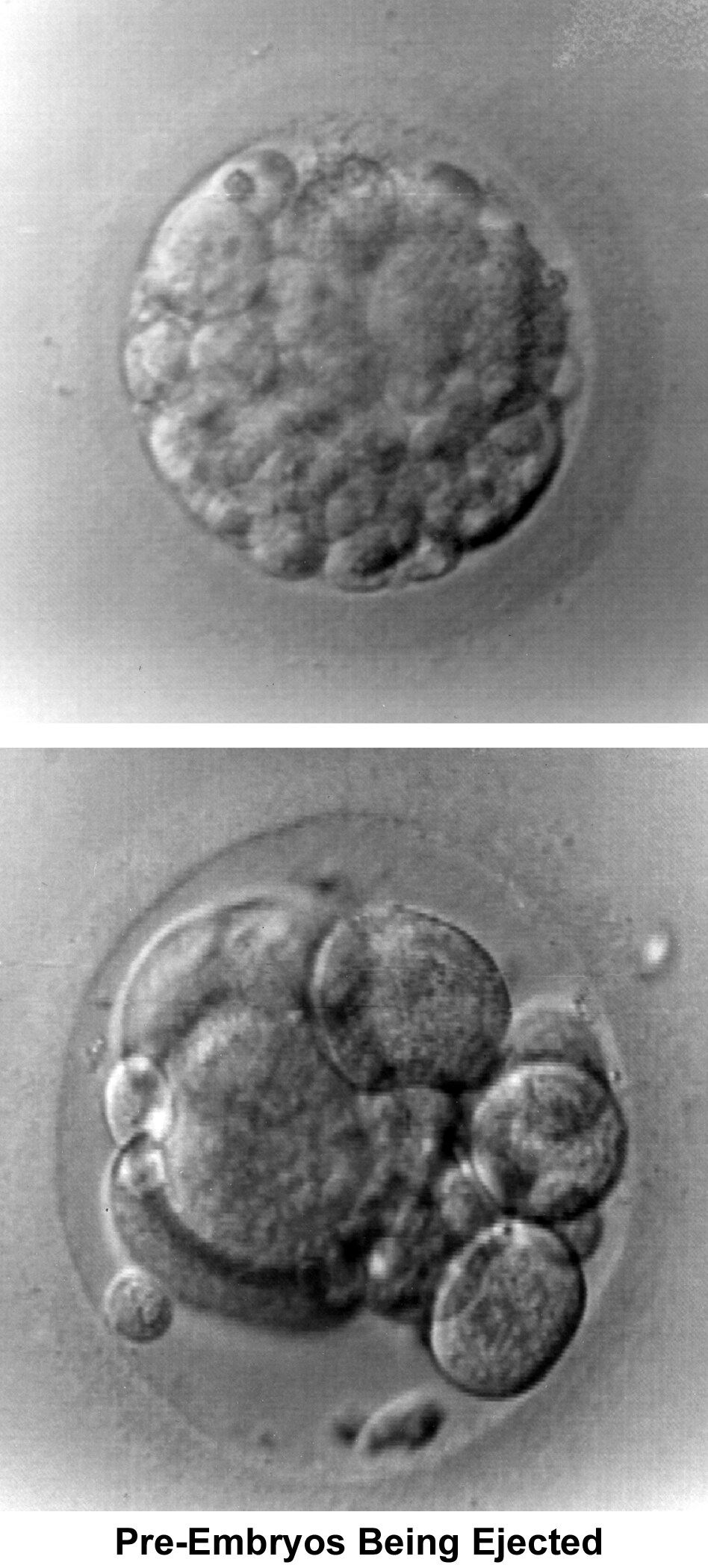
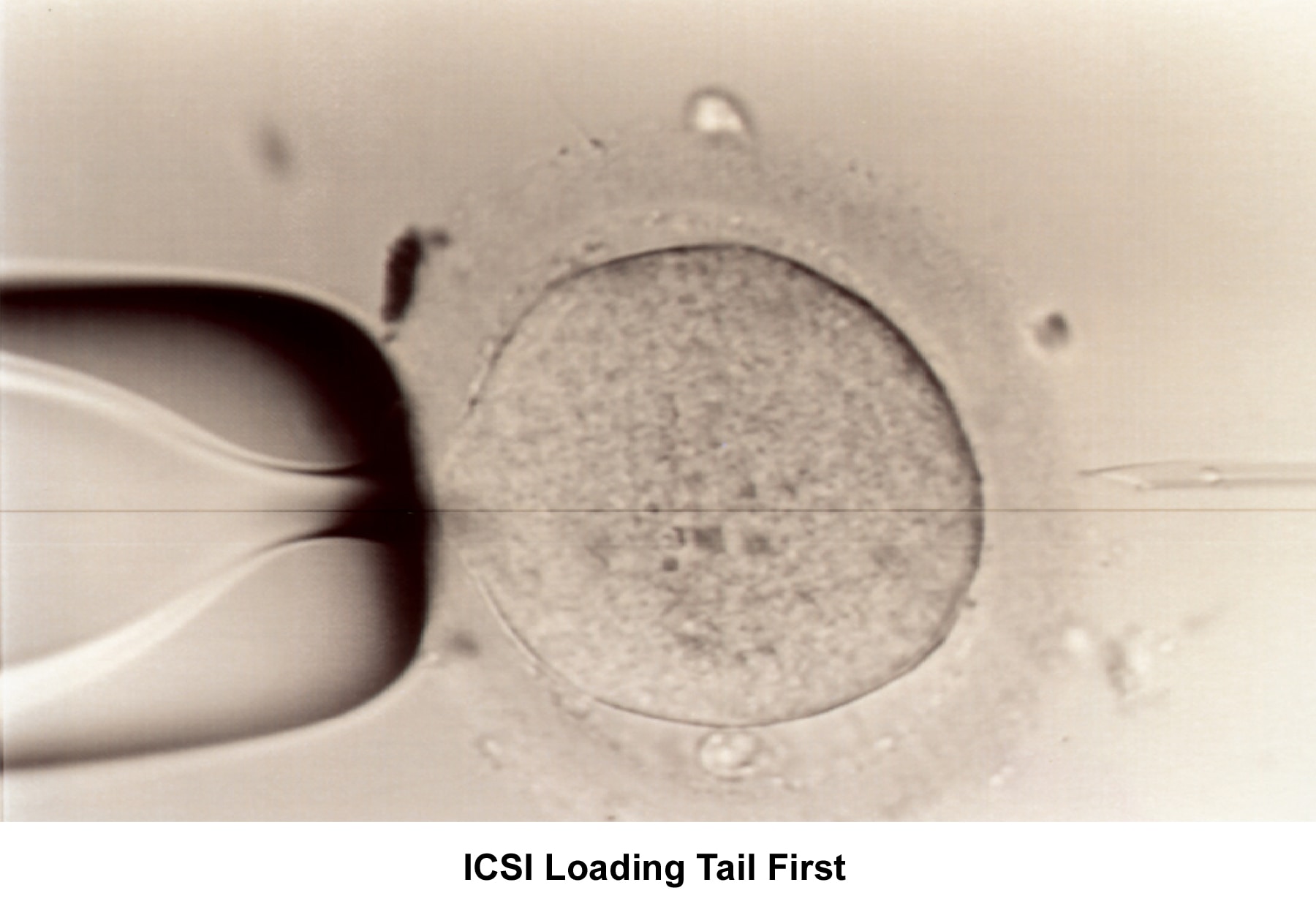
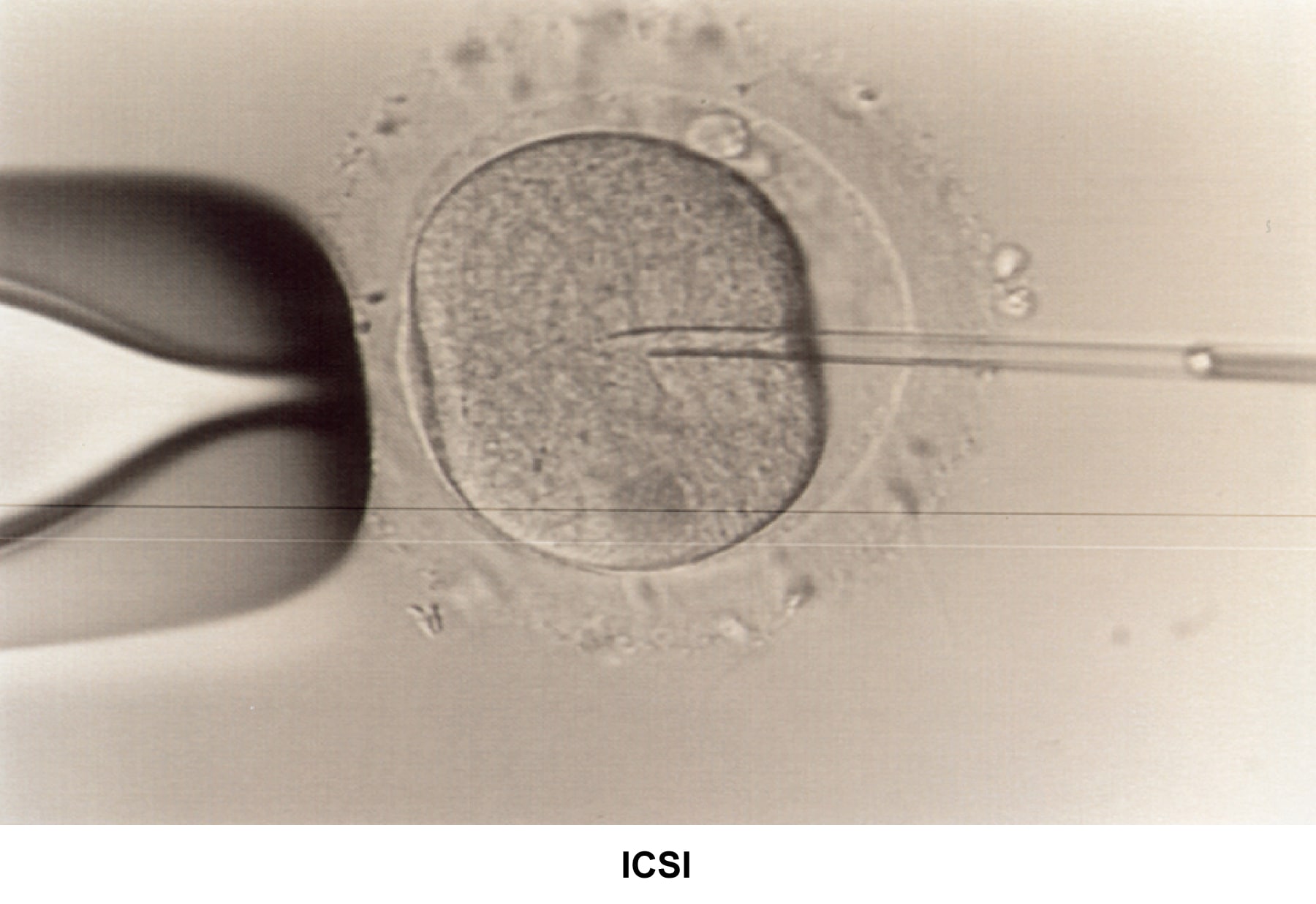
0 Comments:
Post a Comment
<< Home